Abstract
Despite their importance to successful meiosis and various evolutionary processes, meiotic recombination rates sometimes vary within species or between closely related species. For example, humans and chimpanzees share virtually no recombination hotspot locations in the surveyed portion of the genomes. However, conservation of recombination rates between closely related species has also been documented, raising an apparent contradiction. Here, we evaluate how and why conflicting patterns of recombination rate conservation and divergence may be observed, with particular emphasis on features that affect recombination, and the scale and method with which recombination is surveyed. Additionally, we review recent studies identifying features influencing fine-scale and broad-scale recombination patterns and informing how quickly recombination rates evolve, how changes in recombination impact selection and evolution in natural populations, and more broadly, which forces influence genome evolution.
Similar content being viewed by others
Introduction
One of the main goals of evolutionary genetics is to understand how variation is created and maintained within and between species. Homologous meiotic recombination affects variation in the genome: differences in the rate of recombination determine the amount of genetic variation within populations and the rate at which new combinations of alleles are introduced into populations (Brooks and Marks, 1986; Coop and Przeworski, 2007). Indeed, in flies and humans, variability in recombination rate explains more than 50% of the variation in nucleotide heterozygosity across the genome (Nachman, 2002), and recombination may shape features of the genomic landscape such as codon bias, base composition and the distribution of repetitive elements and polymorphisms (Charlesworth et al., 1994; Comeron et al., 1999; Duret and Arndt, 2008; see section ‘Molecular evolutionary consequences of recombination rate variation’). Recombination rates are also expected to mediate the effectiveness of natural and sexual selection on genome evolution because breakdown of linkage between nucleotide sites allows the sites to behave independently, permitting selection to act efficiently, eliminate mutations, reduce genetic hitchhiking and facilitate adaptive evolution by reducing interference between sites (Hill and Robertson, 1966).
Examining variation in recombination rates among closely related species may provide clues as to the evolutionary forces affecting recombination, and therefore offer insight into the forces shaping the genome over time. Studies across virtually all eukaryotic kingdoms have determined that the distribution of recombination events is non-uniform. Indeed, there is variation in recombination rate across the genome; extreme rates are known as ‘hotspots’ and ‘coldspots’ in yeast and mammals, and reflect more of a quantitative change in other organisms. Hotspots are conventionally defined as a significant increase in recombination rate from the background recombination rate, usually ranging on orders of magnitude, that takes place in a small percentage of the genome. In humans this translates to about 80% of the recombination taking place in less than 15% of the sequence (Myers et al., 2006). Although other organisms have not been assayed at the same resolution as yeast, mouse and humans, many studies observe regions with a several-fold increase above the background recombination rate. For example, in Drosophila miranda, several regions show recombination rates between 25 and 30 cM Mb−1, several times the chromosomal average of ∼5 cM Mb−1 (unpublished data; see also Cirulli et al., 2007; Stevison and Noor, 2010).
Recombination rates are variable between individuals, populations and species; however, the causative factors underlying this variation are largely unknown. Through comparison of fine-scale genetic maps of closely related species, it may be possible to identify features influencing fine-scale and broad-scale recombination patterns, as well as features that predict shifts in recombination landscapes between species. Such linkage map comparisons have the potential to answer questions such as how fast recombination rates change and how changes in recombination impact selection and evolution in natural populations. Theoretical work on the evolution of recombination and recombination modifiers has greatly contributed to these efforts, but this review will primarily focus on empirical work (for a review of theoretical considerations, see Charlesworth, 1990; Barton, 1995, 2010; Feldman et al., 1996; Lenormand and Otto, 2000; Martin et al., 2006).
The past few years have seen remarkable progress in the development of fine-scale maps and in revealing novel modifiers of recombination rate. However, some comparisons of recombination maps show seemingly contradictory results, particularly in relation to conservation and divergence of recombination rates. Therefore, in this review, we discuss conservation and divergence in homologous meiotic recombination rate between closely related species. We begin by briefly considering sequence and epigenetic features known to affect recombination and the molecular evolutionary consequences of recombination rate variation. We proceed to discuss several issues surrounding the generation and analysis of recombination maps to understand why we would see conservation or divergence between some species and not others. Similarly, we evaluate how constraints and regulating features could produce conflicting patterns of conservation and divergence when surveyed at different scales. Overall, we hope to highlight important questions pertaining to how recombination shapes genome evolution, and how studying closely related species can help answer them. Because the recombination literature is skewed toward humans, mice and yeast, this review places an emphasis on these taxa, but attempts to incorporate other organisms whenever possible.
Determinants and correlates
It is difficult to rigorously evaluate studies of recombination without considering various factors that may influence recombination rate. Control of recombination rate appears to be multifaceted, with molecular, environmental and demographic factors all having a role (for example, see Wilfert et al., 2007; see also section ‘Why would we see conservation of recombination rates between some species and not others?’ below). Attempts to elucidate the determinants of recombination have identified numerous factors of influence, many of which appear to be conserved across eukaryotes (Lichten and Goldman, 1995; Roeder, 1997; Hassold et al., 2000; Keeney, 2001; Petes, 2001; Page and Hawley, 2003). New hypotheses have emerged, for example, that variation in epigenetic features could explain the variation in the rates of recombination between closely related species (Myers et al., 2005, 2008; Ptak et al., 2005; Winckler et al., 2005). The continued study of recombination between closely related species has the potential to illuminate more decisive determinants and how they change over time. Here, we focus on molecular patterns and features that appear to be associated with or impact recombination.
At the chromosome level, the prevailing pattern across organisms is that a minimum number of crossovers must be achieved for proper segregation. In humans, the number of crossovers is strongly correlated with the number of chromosomes, where one crossover per chromosome ensures proper segregation at meiosis (Fledel-Alon et al., 2009). In other organisms, crossover rates range from one crossover per chromosome arm to as many as five per chromosome (Beye et al., 2006). Animals with numerous shorter chromosomes, such as the ‘microchromosomes’ found in many birds, tend to have higher recombination rates, again, likely as a result of ensuring proper disjunction (Groenen et al., 2009). Along chromosomes in many organisms, rates of recombination tend to be higher toward the distal portions of the chromosome and low around the centromere, perhaps because repression of meiotic recombination by the centromeric heterochromatin is also critical in proper segregation (Ellermeier et al., 2010).
At the genomic level, high recombination rates are positively and nearly ubiquitously associated with GC content, gene density, simple repeats, transposable elements and a number of different sequence motifs (Thuriaux, 1977; Gerton et al., 2000; Marais et al., 2001; Marais, 2003; Jensen-Seaman et al., 2004; Meunier and Duret, 2004; Myers et al., 2005; Groenen et al., 2009; Wong et al., 2010). In particular, it appears that a 13-mer degenerate motif may be responsible for recruiting recombination events in at least 40% of human hotspots (Myers et al., 2008). This motif binds to the zinc-finger protein PRDM9 in humans, and allelic variation controls hotspot activity in both humans and mice (Baudat et al., 2010; Berg et al., 2010). Relatedly, the Drosophila zinc-finger protein, Trade Embargo (trem), initiates double-strand breaks (DSBs) and is necessary for localization of the protein Mei-P22 to discrete foci on meiotic chromosomes, some or all of which are thought to mark sites for future DSBs (Lake et al., 2011). Thus, zinc-finger proteins and sequence motifs may be major determinants of high-recombination-rate locations and recombination rate intensities at these locations.
Observations of divergent hotspot locations and usage among human individuals, and between humans and chimpanzees, has sparked a rigorous analysis of how epigenetics is involved in meiotic hotspot determination (Myers et al., 2005; Ptak et al., 2005; Winckler et al., 2005; Neumann and Jeffreys, 2006). Subsequent studies show correlations between recombination hotspots and open chromatin, numerous histone modification patterns and DNA methylation in yeast, mice and humans (Berchowitz et al., 2009; Buard et al., 2009; Sigurdsson et al., 2009). Of particular note is the presence of a SET-methyltransferase domain in the Prdm9 gene, which is responsible for the common chromatin feature trimethylation of lysine-4 of histone H3, or H3K4me3 (Baudat et al., 2010). H3K4me3 in yeast seems to be a prominent and pre-existing mark of active recombination sites (Borde et al., 2009), potentially creating a link between sequence and epigenetic features affecting recombination.
Continued analysis between individuals and species will surely lead to a greater understanding of existing features and the discovery of novel ones. For example, the analysis of Prdm9 across species has already produced fascinating results. Chimpanzee PRDM9 has dramatically different predicted binding sequence than human PRDM9, and seems to be the most divergent of all orthologous zinc-finger proteins (Myers et al., 2010). Furthermore, Prdm9 in other mammals shows rapid evolution, with variation in zinc-finger number and patterns of substitution suggestive of complex repeat shuffling (Oliver et al., 2009; Myers et al., 2010). Although there is no direct evidence that these changes have generated recombination rate differences, it is surely an intriguing area to be researched. If proven, this may provide an explanation as to how recombination hotspots are created and how they change over time (see section ‘Why would we see recombination rate conservation at some scales and not others?’ below). Closely related species present the unique opportunity to study the evolution of features regulating and influencing recombination rate, and should be central in future studies of this basic biological process.
The molecular evolutionary consequences of recombination rate variation
Recombination rate variation within and between closely related species allows evolutionary biologists to make conclusions as to whether selective or neutral forces are governing genomic landscapes. First postulated in a groundbreaking study, Begun and Aquadro (1992) found that recombination rate was positively correlated with nucleotide diversity in Drosophila melanogaster, but did not observe an association between recombination and D. melanogaster–D. simulans divergence. This pattern is interpreted to mean that natural selection, in particular selective sweeps and/or background selection, eliminates nucleotide variability in regions of low recombination (Smith and Haigh, 1974; Charlesworth et al., 1993), and is supported by studies in several organisms (see Table 1). However, a similar association between recombination rate and nucleotide diversity may be predicted if recombination is mutagenic, but fewer studies have detected a correlation between recombination and nucleotide divergence between species and so have not met the prediction of the mutagenic hypothesis (but, see empirical studies by Brown and Jiricny, 1987; Brown et al., 1989; Strathern et al., 1995; Papavasiliou and Schatz, 2000).
Additionally, the interpretations are complicated by conflicting results in several organisms surveyed (Payseur and Nachman, 2000; Baudry et al., 2001; Nachman, 2001; Huang et al., 2004; Yi et al., 2004; Yi and Li, 2005; Noor, 2008b; Tsai et al., 2010). Confounding factors that may lead to conflicting results are listed in Table 1, but there are several we would like to highlight here. First, conflicting results may simply reflect taxon-specific mutagenicity, but this hypothesis requires more empirical work. Second, correlations of diversity or divergence to recombination rate may change according to the scale with which recombination is assayed (Bussell et al., 2006; Spencer et al., 2006; Kulathinal et al., 2008; Noor, 2008a; Stevison and Noor, 2010), making it a priority to assess these measures using fine-scale recombination over varying magnitudes. Third, and most relevant to the primary topic of this review, many studies up to this point have only assayed recombination in one species of interest, assuming recombination rates are conserved. It remains unresolved as to whether the selection or mutagenic hypothesis primarily accounts for the observed pattern, but perhaps with increasing amounts of recombination and sequence data, we will be able to make firmer conclusions.
Conservation and divergence of recombination
Following the progressive discoveries in diverse species that recombination events are non-random across the genome, one of the most exciting and surprising findings has been the realization that recombination rates sometimes change, even within species or between closely related species. The fact that recombination rates are variable and heritable implies that recombination itself can evolve in response to natural selection (Chinnici, 1971; Charlesworth and Charlesworth, 1985; Otto and Michalakis, 1998). Furthermore, evidence from human recombination hotspots seems to show that this change can occur quickly on an evolutionary timescale, with hotspots emerging and disappearing in as little as 120 000 years, and certainly within the six million years human divergence from chimpanzee (Ptak et al., 2005; Winckler et al., 2005; Jeffreys and Neumann, 2009). However, conservation of recombination between closely related species has also been detected at varying scales (see Table 2), raising many questions: (1) How does the methodology by which recombination is measured affect estimates of recombination rate? (2) Why would we see conservation of recombination rates between some species and not others? (3) Why would we see conservation at some scales and not others? (4) Finally, and perhaps most fundamentally, should we expect to see conservation between closely related species? Here we comprehensively review empirical studies that compare recombination rates between closely related species, and speculate on the answers to these questions.
How does the methodology by which recombination is measured affect estimates of recombination rate?
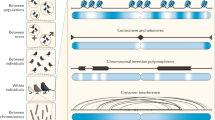
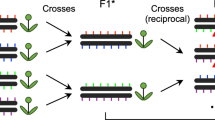
The construction of a recombination map can dramatically affect the estimate of recombination rate depending on the methodology used (and associated biases). Three methods are commonly used for estimating recombination rate: linkage disequilibrium (LD) mapping, sperm typing and direct mapping using polymorphic markers (see Table 2 for examples of recombination maps generated with these approaches, and Table 3 for potential strengths and weaknesses). The first two methods are used primarily with human data (but see Guillon and de Massy, 2002; Ptak et al., 2004; Kim et al., 2007; Arguello et al., 2010), with the labor- and resource-intensive direct mapping applied more in other model organisms. Fundamentally, the major differences between these measures are (1) whether the recombination rates measured are current versus historical, and (2) whether the recombination rates measured reflect a population average or focus on a particular individual or set of individuals.
There has been some doubt as to whether LD consistently and accurately predicts hotspots (Jeffreys et al., 2005b; Reed and Tishkoff, 2006). For instance, Coop et al. (2008) estimated that 40% of crossovers occurred outside of LD-predicted hotspots, but Khil and Camerini-Otero (2010) suspect this may be an overestimate owing to the way hotspots were measured using particular populations. Khil and Camerini-Otero also found that 26–32% of crossovers happened outside of European population LD-predicted hotspots; however, this discrepancy disappeared when hotspot locations of other populations were taken into account. There are also specific examples of LD-predicted hotspots being absent when checked with sperm typing (Kauppi et al., 2005), although such inconsistencies appear to be the exception rather than the rule (Jeffreys et al., 2000, 2001; Yauk et al., 2003).
In pedigrees and controlled crosses, increasing sample size and marker coverage changes the way we measure and perceive recombination, similar to the ‘Beavis effect’ for mapping (Beavis et al., 1994; Beavis, 1996). For example, the original honeybee linkage map used 94–142 individuals and 365 markers for a total map length of 3450 cM (Hunt and Page, 1995); the newer map used 541 markers and 283 individuals for a total length of 4061.2 cM (Solignac et al., 2004). With more accurate technologies, the honeybee genome increased in length from 178 Mb in 1974 (Jordan and Brosemer, 1974) to 262 Mb in 2006 (The Honeybee Genome Consortium), thereby decreasing the average recombination rate from 19.38 to 16 cM Mb−1 today. Similarly, in chicken, the current map used 9268 markers for a total length of 3228 cM (Groenen et al., 2009), substantially smaller than the 4200 cM previously estimated using 2261 markers (Schmid et al., 2005). However, obtaining enough markers to detect fine-scale recombination is resource-intensive, thereby producing maps that range in scale from kilobases (hereafter referred to as ‘fine scale’), to hundreds of kilobases (hereafter referred to as ‘intermediate scale’), to tens of megabases (hereafter referred to as ‘broad scale’), to whole genomes. This is relevant, as recombination rate conservation and divergence between species is scale-dependent (see below).
Other indirect quantitative approaches also exist, such as immunostaining as used by Dumont and Payseur (2011) in Murid rodents and Double Strand Break (DSB) mapping, most commonly used in yeast (Gerton et al., 2000; Buhler et al., 2007; Mancera et al., 2008). Of course, choosing an approach is constrained by the organism and the resources available, and researchers must be aware of limitations when making generalizations and conclusions.
Why would we see conservation of recombination rates between some species and not others?
There are several pertinent issues to consider when comparing recombination rates between closely related species. First, differential action of selection, or selection in changing environments, could give rise to differences between species (Chinnici, 1971; Charlesworth and Charlesworth, 1985; True et al., 1996). For example, artificial selection may have increased recombination rates in the domesticated species of chicken, honeybee and many plants (Rees and Dale, 1974; Burt and Bell, 1987; Otto and Barton, 2001; Ross-Ibarra, 2004; Wilfert et al., 2007; Groenen et al., 2009) in comparison with their wild progenitors.
Second, imprecise measures of physical distance can lead to inferred differences in recombination rate. Large insertions, deletions or inversions can affect the recombination fraction between two points in the genome because single crossover events may often lead to aneuploidy. Even if such changes do not change the actual recombination ‘rate’ (cM/Mb), if a large insertion is present in one taxon but not another, it may appear that recombination rates have diverged between species, when in fact recombination is being measured over intervals of unequal size. Moreover, chromosomal rearrangements, such as inversions, reduce the observed amount of recombination in heterokaryotypes (Hartl and Jones, 2004, pp 319–324). This repression of recombination associated with inversions extends several megabases (Mb) outside the inversion, thereby producing broader scale changes in recombination rate (Kulathinal et al., 2009). Furthermore, inversion heterozygotes show increased recombination further outside the inverted region, known as the ‘inter-chromosomal effect’ (Schultz and Redfield, 1951). Large insertions and deletions have the potential to produce comparable outcomes to inversions. In comparing the same intervals between two closely related species in the absence of a genome sequence, one runs the risk of concluding increased divergence between species when in actuality, an inversion, insertion or deletion segregating in only one species is obscuring their comparable recombination rates.
Third, recombination is variable within individuals and populations (Brooks and Marks, 1986; True et al., 1996; Carrington and Cullen, 2004; Neumann and Jeffreys, 2006; Graffelman et al., 2007; Coop et al., 2008; Paigen et al., 2008; Cheng et al., 2009; Dumont et al., 2009; Kong et al., 2010 etc). This variation may stem from actual heritable variation in recombination rates among individuals; variation within an individual among regions of its genome (as discussed above) or from environmental variation. Because of potentially dramatic within-population variation, one must use caution in making generalizations about species as a whole or presumptions that a single linkage map describes the species. Without first surveying recombination within the populations, subspecies or species of interest, it will be impossible to conclude true conservation or divergence between groups. These potential challenges in estimating recombination rate can translate into real or perceived differences between closely related species.
Why would we see recombination rate conservation at some scales and not others?
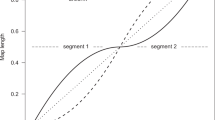
Maps produced at varying scales provide an unexpected and seemingly contradictory view of conservation and divergence between species (see Table 2). In mice and humans, conservation of recombination rate was identified at a broad scale, but divergence of recombination hotspots was identified at a fine scale, suggesting that there may be differential regulation among the scales (Myers et al., 2005; Coop and Przeworski, 2007), or perhaps, simply that we do not understand the relationship between fine-scale and broad-scale recombination patterns.
Divergence of fine-scale recombination rates observed in yeast and humans is likely caused by the rapid turnover of hotspots owing to biased gene conversion and meiotic drive. In the process of DSB repair, one pathway leads to crossover and exchange of content between homologous chromosomes, whereas the other pathway leads to gene conversion (Boulton et al., 1997; Marais, 2003). Biased gene conversion favors one allele over the other, in which case the initiating hotspot is replaced by a copy of its homolog, effectively suppressing subsequent recombination. Simulations and empirical evidence have shown the self-destructive nature of hotspots through over-transmission of recombination-suppressing alleles (meiotic drive), creating what is known as ‘the hotspot paradox’ (Boulton et al., 1997; Jeffreys and Neumann, 2002).
Recent evidence might provide the elusive answer as to how new hotspots are created and regulated to counteract losses due to the hotspot paradox. The zinc-finger protein PRDM9, confirmed to have a significant role in recombination in human and mouse, contains a zinc-finger-encoding region with a minisatellite structure (Baudat et al., 2010). This particular structure may confer a strong potential to generate variability by recombination or replication slippage within the array. Indeed, studies documented variability in the contact residues predicting DNA binding between human populations (Baudat et al., 2010; Berg et al., 2010; Parvanov et al., 2010), and the number of zinc fingers and their contact residues vary significantly across rodents, primates and other Metazoans (Oliver et al., 2009). The changing of contact residues could create a new family of hotspots by the binding of the protein to new sequence motifs, thereby counteracting the loss of hotspots due to biased gene conversion. Regardless of the mechanism, it is clear by the constant flux of hotspots that the precise locations seem to be unconstrained, allowing divergence at the hotspot level.
It is believed that broad-scale recombination is controlled in a different, but potentially non-mutually, exclusive manner. Above all, the broad-scale rate is defined by the necessity of one crossover per chromosome to ensure proper disjunction (Hassold et al., 2004; Fledel-Alon et al., 2009). Other chromosomal properties, including size and number, are correlated with this trend. This selective constraint is likely a large influence on the conservation of broad-scale rates between closely related species. At the intermediate scale, conclusions are more vague. Regional recombination is decidedly influenced by crossover interference, where a crossover in one location prevents another crossover from occurring close by (Foss et al., 1993; Hillers, 2004; Stahl et al., 2004; Copenhaver, 2005). Regional properties of chromosomes have an impact as well, clearly shown by the lack of crossovers in the centromeric region and typically a high number of crossovers near the telomeres. Overall, perhaps these broader scale processes are more likely to be conserved, and hence preserve recombination at this scale as a byproduct.
The connection between the different scales is also unclear. In humans, there are an estimated 60 000–80 000 hotspots across the genome (Khil and Camerini-Otero, 2010), occurring in clusters every 60–90 kb, with individual hotspots separated by 1–7 kb within each cluster (Jeffreys et al., 2001; clustering also seen in mouse, Kelmenson et al., 2005) and no region greater than 200 kb in which recombination is absent (Myers et al., 2006). It appears that non-hotspot recombination is relatively rare, with sperm genotyping studies showing very low levels of background, non-hotspot recombination, most likely located in the weaker and polymorphic hotspots (Jeffreys et al., 1998, 2001, 2005b; Jeffreys and Neumann, 2002, 2005a). It is proposed that larger scale variation may be a product of the varying density or intensity of these hotspots in different regions of the genome (Nachman, 2002; Myers et al., 2006).
There are several hypotheses as to which selective pressures influence the overall process and distribution. Hey (2004) proposed that individual hotspots may arise as a byproduct of LD between genes that are being selected. If selection favors a recombinant haplotype, then it will favor chromosomes with high recombination between the two genes, thereby producing a variation in the location and the intensity of local recombination rates that fluctuate depending on the genes under selection and the LD patterns. Kauppi et al. (2004) put forward three hypotheses: the first stating that the distribution of recombination is governed by a balance between the need for recombination to ensure proper segregation during meiosis and the need to minimize the breakdown of favorable haplotypes, and the second and the third that state, more mechanistically, that restrictions on crossover position potentially facilitate the optimal mechanical/biochemical function of chiasmata in chromosome segregation, or that restrictions on position confine DSBs to regions that are most conducive to efficient assembly of machinery and repair.
The answer is plausibly a combination of all of these propositions. The continued analysis of recombination rates assayed at different scales, combined with theory that could test selective constraints at these scales, will help to determine why we would see conservation in recombination rate at some scales and not others.
Should we expect to see conservation between closely related species?
Just as we should expect to see conservation in sequence between closely related species, we should expect to see conservation in recombination (Dumont and Payseur, 2008). Presumably, with sequence similarity comes, on average, greater sharing of features that influence recombination. Divergence should be seen as departure from the null hypothesis, but only if there is a significant increase in divergence relative to variability of recombination within species, something rarely studied.
Organisms with resolution of recombination rate at the fine scale (for example human, mouse, and yeast) show that hotspots do indeed diverge over time. Other organisms with maps at an intermediate-to-broad scale generally portray a trend of conservation (see Table 2). From the evidence we have, we can determine that hotspots are not conserved owing to their transient nature. Over long periods of time, enough changes occur on the fine scale to produce a broad-scale change. Additionally, on a broad scale, shared constraints will hold the rate more constant, so that rates are more conserved between closely related species. These processes are compounded by changes in the genome such as inversions, translocations, insertions and deletions that lead to altered recombination rates over time.
Future studies should strive to confirm both these trends, and the mechanisms behind them, to better understand the impact recombination has on the genome, and which features of the genome regulate recombination. A combination of theoretical and empirical work will be necessary. Theoretically, models and simulations can inform the role of selective and neutral processes, and the different population parameters that could lead to the conservation or divergence of recombination rates. Empirically, in chimpanzees, comparisons of human–chimpanzee recombination rates should be extended beyond isolated regions of the genome. Recombination should also be surveyed in a broader set of species, with particular attempt to obtain a resolution of less than 10 kb in order to observe the presence or absence of hotspots, albeit this is a major challenge in non-model systems. The ability to make cross-species generalizations about recombination rates will have an impact on our understanding of genome evolution, thereby implicating diverse topics such as human health, selection and neutrality in the genome, and speciation and mapping studies.
Conclusions
Several patterns emerge upon reviewing data from these recombination maps. First, labels such as conservation and divergence are somewhat misleading. No pair of species studied to date exhibits complete divergence or conservation of all studied hotspots or regions of high recombination, and furthermore, many studies report only a few intervals across the genome that can be categorized in these terms. Therefore, it is necessary to attempt to incorporate a standard, or at least explicitly defined, set of parameters when discussing divergence. Of course, the vast range of scales at which recombination has been, and continues to be, surveyed makes this difficult. Because recombination is considered a quantitative genetic trait showing variation and heritability that can be acted upon by natural selection, it shares features with other phenotypes and can be discussed in similar terms. A standard approach is to discuss sequence divergence as a percentage, and here we recommend recombination rate divergence to be treated in a similar manner. It is appropriate to express intervals conserved or diverged out of the total number surveyed, also citing the percentage of the total of the physical and recombination maps that was surveyed.
With more genome sequences becoming available, the decreasing cost of genotyping, and sophisticated software and technology at our fingertips, the detection of recombination can be completed with greater feasibility. These projects should be undertaken bearing several parameters in mind. First, recombination variation should be assayed within and across populations within species before making conclusions about differences between species. This is necessary to make conclusions regarding conservation or divergence between species, but will also provide a perspective on the speed at which recombination rate is changing. Second, genome sequences should be available for the populations or species involved, and particularly for the strains being surveyed. These data will ensure that no chromosomal rearrangements or insertions/deletions exist, and will facilitate analysis of features such as motifs, and measures of diversity and divergence. Surveying diversity and divergence will also provide a better understanding of the forces at work in the genome. Third, examining recombination at multiple scales will aid in the interpretation of different constraints influencing the distribution of recombination events, especially when compared between populations and species. Finally, in interpreting results, population history and the methodology used to infer recombination should be taken into account to avoid biases and complications.
Fine-scale recombination maps can help to address essential questions such as how variation is created and maintained within and between species. Recombination shapes the features of the genome and creates new allelic combinations that allow increased adaptability in all sexual organisms. However, many researchers have assumed that recombination rate is invariable among individuals and between species, which we now know is inherently false. Indeed, knowledge of fine-scale variation in crossover rate is essential in modeling genome evolution, population genetics studies, genome-wide association studies and inferring evolutionary processes. Thus, results indicating how recombination rate is distributed in the genome will have implications in human health, molecular evolution and the way we study genetics. The role of recombination in genome evolution is a fundamental issue in understanding basic biological processes, and although much progress has been made, many questions remain unanswered.
References
Ardlie KG, Kruglyak L, Seielstad M (2002). Patterns of linkage disequilibrium in the human genome. Nat Rev Genet 3: 299–309.
Arguello JR, Zhang Y, Kado T, Fan CZ, Zhao RP, Innan H et al. (2010). Recombination yet inefficient selection along the Drosophila melanogaster subgroup's fourth chromosome. Mol Biol Evol 27: 848–861.
Backstrom N, Forstmeier W, Schielzeth H, Mellenius H, Nam K, Bolund E et al. (2010). The recombination landscape of the zebra finch Taeniopygia guttata genome. Genome Res 20: 485–495.
Barton NH (1995). A general-model for the evolution of recombination. Genet Res 65: 123–144.
Barton NH (2010). Mutation and the evolution of recombination. Philos Transact R Soc B Biol Sci 365: 1281–1294.
Baudat F, Buard J, Grey C, Fledel-Alon A, Ober C, Przeworski M et al. (2010). PRDM9 is a major determinant of meiotic recombination hotspots in humans and mice. Science 327: 836–840.
Baudry E, Kerdelhue C, Innan H, Stephan W (2001). Species and recombination effects on DNA variability in the tomato genus. Genetics 158: 1725–1735.
Beavis WD (1996). QTL mapping in plant breeding populations. American Statistical Association 1996 Proceedings of the Biometrics Section 13–20.
Beavis WD, Smith OS, Grant D, Fincher R (1994). Identification of quantitative trait loci using a small sample of topcrossed and F4 progeny from maize. Crop Sci 34: 882–896.
Begun DJ, Aquadro CF (1992). Levels of naturally-occurring DNA polymorphism correlate with recombination rates in Drosophila-melanogaster. Nature 356: 519–520.
Begun DJ, Holloway AK, Stevens K, Hillier LW, Poh YP, Hahn MW et al. (2007). Population genomics: whole-genome analysis of polymorphism and divergence in Drosophila simulans. PLoS Biol 5: 2534–2559.
Berchowitz LE, Hanlon SE, Lieb JD, Copenhaver GP (2009). A positive but complex association between meiotic double-strand break hotspots and open chromatin in Saccharomyces cerevisiae. Genome Res 19: 2245–2257.
Berg IL, Neumann R, Lam KWG, Sarbajna S, Odenthal-Hesse L, May CA et al. (2010). PRDM9 variation strongly influences recombination hotspot activity and meiotic instability in humans. Nat Genet 42: 1099–1103.
Beye M, Gattermeier I, Hasselmann M, Gempe T, Schioett M, Baines JF et al. (2006). Exceptionally high levels of recombination across the honey bee genome. Genome Res 16: 1339–1344.
Borde V, Robine N, Lin W, Bonfils S, Geli V, Nicolas A (2009). Histone H3 lysine 4 trimethylation marks meiotic recombination initiation sites. EMBO J 28: 99–111.
Boulton A, Myers RS, Redfield RJ (1997). The hotspot conversion paradox and the evolution of meiotic recombination. Proc Natl Acad Sci USA 94: 8058–8063.
Brooks LD, Marks RW (1986). The organization of genetic-variation for recombination in Drosophila-melanogaster. Genetics 114: 525–547.
Brown TC, Jiricny J (1987). A specific mismatch repair event protects mammalian-cells from loss of 5-methylcytosine. Cell 50: 945–950.
Brown TC, Zbinden I, Cerutti PA, Jiricny J (1989). Modified Sv40 for analysis of mismatch repair in simian and human-cells. Mut Res 220: 115–123.
Buard J, Barthes P, Grey C, de Massy B (2009). Distinct histone modifications define initiation and repair of meiotic recombination in the mouse. EMBO J 28: 2616–2624.
Buhler C, Borde V, Lichten M (2007). Mapping meiotic single-strand DNA reveals a new landscape of DNA double-strand breaks in Saccharomyces cerevisiae. PLoS Biol 5: 2797–2808.
Burt A, Bell G (1987). Mammalian chiasma frequencies as a test of 2 theories of recombination. Nature 326: 803–805.
Bussell JJ, Pearson NM, Kanda R, Filatov DA, Lahn BT (2006). Human polymorphism and human–chimpanzee divergence in pseudoautosomal region correlate with local recombination rate. Gene 368: 94–100.
Carrington M, Cullen M (2004). Justified chauvinism: advances in defining meiotic recombination through sperm typing. Trends Genet 20: 196–205.
Charlesworth B (1990). Mutation–selection balance and the evolutionary advantage of sex and recombination. Genet Res 55: 199–221.
Charlesworth B, Charlesworth D (1985). Genetic-variation in recombination in Drosophila .1. Responses to selection and preliminary genetic-analysis. Heredity 54 (Feb): 71–83.
Charlesworth B, Morgan MT, Charlesworth D (1993). The effect of deleterious mutations on neutral molecular variation. Genetics 134: 1289–1303.
Charlesworth B, Sniegowski P, Stephan W (1994). The evolutionary dynamics of repetitive DNA in eukaryotes. Nature 371: 215–220.
Cheng EY, Hunt PA, Naluai-Cecchini TA, Fligner CL, Fujimoto VY, Pasternack TL et al. (2009). Meiotic recombination in human oocytes. PLoS Genet 5: e1000661.
Chinnici JP (1971). Modification of recombination frequency in Drosophila .1. Selection for increased and decreased crossing over. Genetics 69: 71–83.
Cirulli ET, Kliman RM, Noor MAF (2007). Fine-scale crossover rate heterogeneity in Drosophila pseudoobscura. J Mol Evol 64: 129–135.
Clark AG, Wang X, Matise T (2010). Contrasting methods of quantifying fine structure of human recombination. Annu Rev Genomics Hum Genet 11: 45–64.
Comeron JM, Kreitman M, Aguade M (1999). Natural selection on synonymous sites is correlated with gene length and recombination in Drosophila. Genetics 151: 239–249.
Coop G, Przeworski M (2007). An evolutionary view of human recombination. Nat Rev Genet 8: 23–34.
Coop G, Wen XQ, Ober C, Pritchard JK, Przeworski M (2008). High-resolution mapping of crossovers reveals extensive variation in fine-scale recombination patterns among humans. Science 319: 1395–1398.
Copenhaver GP (2005). Plant genetics: when not to interfere. Curr Biol 15: R290–R291.
Dawson DA, Akesson M, Burke T, Pemberton JM, Slate J, Hansson B (2007). Gene order and recombination rate in homologous chromosome regions of the chicken and a passerine bird. Mol Biol Evol 24: 1537–1552.
Dumont BL, Broman KW, Payseur BA (2009). Variation in genomic recombination rates among heterogeneous stock mice. Genetics 182: 1345–1349.
Dumont BL, Payseur BA (2008). Evolution of the genomic rate of recombination in mammals. Evolution 62: 276–294.
Dumont BL, Payseur BA (2011). Evolution of the genomic recombination rate in Murid rodents. Genetics 187: 643–657.
Dumont BL, White MA, Steffy B, Wiltshire T, Payseur BA (2011). Extensive recombination rate variation in the house mouse species complex inferred from genetic linkage maps. Genome Res 21: 114–125.
Duret L, Arndt PF (2008). The impact of recombination on nucleotide substitutions in the human genome. PLoS Genet 4: e1000071.
Ellermeier C, Higuchi EC, Phadnis N, Holm L, Geelhood JL, Thon G et al. (2010). RNAi and heterochromatin repress centromeric meiotic recombination. Proc Natl Acad Sci USA 107: 8701–8705.
Feldman MW, Otto SP, Christiansen FB (1996). Population genetic perspectives on the evolution of recombination. Annu Rev Genet 30: 261–295.
Fledel-Alon A, Wilson DJ, Broman K, Wen XQ, Ober C, Coop G et al. (2009). Broad-scale recombination patterns underlying proper disjunction in humans. PLoS Genet 5: e1000658.
Foss E, Lande R, Stahl FW, Steinberg CM (1993). Chiasma interference as a function of genetic-distance. Genetics 133: 681–691.
Gerton JL, DeRisi J, Shroff R, Lichten M, Brown PO, Petes TD (2000). Global mapping of meiotic recombination hotspots and coldspots in the yeast Saccharomyces cerevisiae. Proc Natl Acad Sci USA 97: 11383–11390.
Graffelman J, Balding DJ, Gonzalez-Neira A, Bertranpetit J (2007). Variation in estimated recombination rates across human populations. Hum Genet 122: 301–310.
Groenen MAM, Wahlberg P, Foglio M, Cheng HH, Megens HJ, Crooijmans RPMA et al. (2009). A high-density SNP-based linkage map of the chicken genome reveals sequence features correlated with recombination rate. Genome Res 19: 510–519.
Guillon H, de Massy B (2002). An initiation site for meiotic crossing-over and gene conversion in the mouse. Nat Genet 32: 296–299.
Hartl DL, Jones EW (2004). Genetics: Analysis of Genes and Genomes, 6th edn. Jones and Bartlett Publishers: Sudbury, Massachusetts.
Hassold T, Judis L, Chan ER, Schwartz S, Seftel A, Lynn A (2004). Cytological studies of meiotic recombination in human males. Cytogenet Genome Res 107: 249–255.
Hassold T, Sherman S, Hunt P (2000). Counting cross-overs: characterizing meiotic recombination in mammals. Hum Mol Genet 9: 2409–2419.
Hellmann I, Ebersberger I, Ptak SE, Paabo S, Przeworski M (2003). A neutral explanation for the correlation of diversity with recombination rates in humans. Am J Hum Genet 72: 1527–1535.
Hey J (2004). What's so hot about recombination hotspots? PLoS Biol 2: 730–733.
Hill WG, Robertson A (1966). Effect of linkage on limits to artificial selection. Genet Res 8: 269–294.
Hillers KJ (2004). Crossover interference. Curr Biol 14: R1036–R1037.
Huang SW, Friedman R, Yu N, Yu A, Li WH (2004). How strong is the mutagenicity of recombination in mammals? Mol Biol Evol 22: 426–431.
Hunt GJ, Page RE (1995). Linkage map of the honey-bee, Apis mellifera, based on RAPD markers. Genetics 139: 1371–1382.
Jeffreys AJ, Kauppi L, Neumann R (2001). Intensely punctate meiotic recombination in the class II region of the major histocompatibility complex. Nat Genet 29: 217–222.
Jeffreys AJ, Murray J, Neumann R (1998). High-resolution mapping of crossovers in human sperm defines a minisatellite-associated recombination hotspot. Mol Cell 2: 267–273.
Jeffreys AJ, Neumann R (2002). Reciprocal crossover asymmetry and meiotic drive in a human recombination hot spot. Nat Genet 31: 267–271.
Jeffreys AJ, Neumann R (2005a). Factors influencing recombination frequency and distribution in a human meiotic crossover hotspot. Hum Mol Genet 14: 2277–2287.
Jeffreys AJ, Neumann R (2009). The rise and fall of a human recombination hot spot. Nat Genet 41: 625–629.
Jeffreys AJ, Neumann R, Panayi M, Myers S, Donnelly P (2005b). Human recombination hot spots hidden in regions of strong marker association. Nat Genet 37: 601–606.
Jeffreys AJ, Ritchie A, Neumann R (2000). High resolution analysis of haplotype diversity and meiotic crossover in the human TAP2 recombination hotspot. Hum Mol Genet 9: 725–733.
Jensen-Seaman MI, Furey TS, Payseur BA, Lu YT, Roskin KM, Chen CF et al. (2004). Comparative recombination rates in the rat, mouse, and human genomes. Genome Res 14: 528–538.
Jordan RA, Brosemer RW (1974). Characterization of DNA from 3 bee species. J Insect Physiol 20: 2513–2520.
Kauppi L, Jeffreys AJ, Keeney S (2004). Where the crossovers are: recombination distributions in mammals. Nat Rev Genet 5: 413–424.
Kauppi L, Stumpf MPH, Jeffreys AJ (2005). Localized breakdown in linkage disequilibrium does not always predict sperm crossover hot spots in the human MHC class II region. Genomics 86: 13–24.
Keeney S (2001). Mechanism and control of meiotic recombination initiation. Curr Top Dev Biol 52: 1–53.
Kelmenson PM, Petkov P, Wang XS, Higgins DC, Paigen BJ, Paigen K (2005). A torrid zone on mouse chromosome 1 containing a cluster of recombinational hotspots. Genetics 169: 833–841.
Khil PP, Camerini-Otero RD (2010). Genetic crossovers are predicted accurately by the computed human recombination map. PLoS Genet 6: e1000831.
Kim S, Plagnol V, Hu TT, Toomajian C, Clark RM, Ossowski S et al. (2007). Recombination and linkage disequilibrium in Arabidopsis thaliana. Nat Genet 39: 1151–1155.
Kong A, Thorleifsson G, Gudbjartsson DF, Masson G, Sigurdsson A, Jonasdottir A et al. (2010). Fine-scale recombination rate differences between sexes, populations and individuals. Nature 467: 1099–1103.
Kraft T, Sall T, Magnusson-Rading I, Nilsson NO, Hallden C (1998). Positive correlation between recombination rates and levels of genetic variation in natural populations of sea beet (Beta vulgaris subsp. maritima). Genetics 150: 1239–1244.
Kulathinal RJ, Bennett SM, Fitzpatrick CL, Noor MA (2008). Fine-scale mapping of recombination rate in Drosophila refines its correlation to diversity and divergence. Proc Natl Acad Sci USA 105: 10051–10056.
Kulathinal RJ, Stevison LS, Noor MA (2009). The genomics of speciation in Drosophila: diversity, divergence, and introgression estimated using low-coverage genome sequencing. PLoS Genet 5: e1000550.
Lake CM, Nielsen RJ, Hawley RS (2011). The Drosophila zinc finger protein trade embargo is required for double strand break formation in meiosis. PLoS Genet 7: e1002005.
Lenormand T, Otto SP (2000). The evolution of recombination in a heterogeneous environment. Genetics 156: 423–438.
Lichten M, Goldman ASH (1995). Meiotic recombination hotspots. Annu Rev Genet 29: 423–444.
Mancera E, Bourgon R, Brozzi A, Huber W, Steinmetz LM (2008). High-resolution mapping of meiotic crossovers and non-crossovers in yeast. Nature 454: 479–485.
Marais G (2003). Biased gene conversion: implications for genome and sex evolution. Trends Genet 19: 330–338.
Marais G, Mouchiroud D, Duret L (2001). Does recombination improve selection on codon usage? Lessons from nematode and fly complete genomes. Proc Natl Acad Sci USA 98: 5688–5692.
Martin G, Otto SP, Lenormand T (2006). Selection for recombination in structured populations. Genetics 172: 593–609.
Meunier J, Duret L (2004). Recombination drives the evolution of GC-content in the human genome. Mol Biol Evol 21: 984–990.
Meznar ER, Gadau J, Koeniger N, Rueppell O (2010). Comparative linkage mapping suggests a high recombination rate in all honeybees. J Hered 101: S118–S126.
Myers S, Bottolo L, Freeman C, McVean G, Donnelly P (2005). A fine-scale map of recombination rates and hotspots across the human genome. Science 310: 321–324.
Myers S, Bowden R, Tumian A, Bontrop RE, Freeman C, MacFie TS et al. (2010). Drive against hotspot motifs in primates implicates the PRDM9 gene in meiotic recombination. Science 327: 876–879.
Myers S, Freeman C, Auton A, Donnelly P, McVean G (2008). A common sequence motif associated with recombination hot spots and genome instability in humans. Nat Genet 40: 1124–1129.
Myers S, Spencer CCA, Auton A, Bottolo L, Freeman C, Donnelly P et al. (2006). The distribution and causes of meiotic recombination in the human genome. Biochem Soc Transact 34: 526–530.
Nachman MW (1997). Patterns of DNA variability at X-linked loci in Mus domesticus. Genetics 147: 1303–1316.
Nachman MW (2001). Single nucleotide polymorphisms and recombination rate in humans. Trends Genet 17: 481–485.
Nachman MW (2002). Variation in recombination rate across the genome: evidence and implications. Curr Opin Genet Dev 12: 657–663.
Nachman MW, Bauer VL, Crowell SL, Aquadro CF (1998). DNA variability and recombination rates at X-linked loci in humans. Genetics 150: 1133–1141.
Neumann R, Jeffreys AJ (2006). Polymorphism in the activity of human crossover hotspots independent of local DNA sequence variation. Hum Mol Genet 15: 1401–1411.
Noor MAF (2008a). Connecting recombination, nucleotide diversity and species divergence in Drosophila. Fly 2: 255–256.
Noor MAF (2008b). Mutagenesis from meiotic recombination is not a primary driver of sequence divergence between Saccharomyces species. Mol Biol Evol 25: 2439–2444.
Oliver PL, Goodstadt L, Bayes JJ, Birtle Z, Roach KC, Phadnis N et al. (2009). Accelerated evolution of the Prdm9 speciation gene across diverse metazoan taxa. PLoS Genet 5: e1000753.
Otto SP, Barton NH (2001). Selection for recombination in small populations. Evolution 55: 1921–1931.
Otto SP, Michalakis Y (1998). The evolution of recombination in changing environments. Trends Ecol Evol 13: 145–151.
Page SL, Hawley RS (2003). Chromosome choreography: the meiotic ballet. Science 301: 785–789.
Paigen K, Szatkiewicz JP, Sawyer K, Leahy N, Parvanov ED, Ng SHS et al. (2008). The recombinational anatomy of a mouse chromosome. PLoS Genet 4: e1000119.
Papavasiliou FN, Schatz DG (2000). Cell-cycle-regulated DNA double-strand breaks in somatic hypermutation of immunoglobulin genes. Nature 408: 216–221.
Parvanov ED, Petkov PM, Paigen K (2010). Prdm9 controls activation of mammalian recombination hotspots. Science 327: 835.
Payseur BA, Nachman MW (2000). Microsatellite variation and recombination rate in the human genome. Genetics 156: 1285–1298.
Petes TD (2001). Meiotic recombination hot spots and cold spots. Nat Rev Genet 2: 360–369.
Ptak SE, Hinds DA, Koehler K, Nickel B, Patil N, Ballinger DG et al. (2005). Fine-scale recombination patterns differ between chimpanzees and humans. Nat Genet 37: 429–434.
Ptak SE, Roeder AD, Stephens M, Gilad Y, Paabo S, Przeworski M (2004). Absence of the TAP2 human recombination hotspot in chimpanzees. PLoS Biol 2: 849–855.
Reed FA, Tishkoff SA (2006). Positive selection can create false hotspots of recombination. Genetics 172: 2011–2014.
Rees H, Dale PJ (1974). Chiasmata and variability in Lolium and Festuca populations. Chromosoma 47: 335–351.
Roeder GS (1997). Meiotic chromosomes: it takes two to tango. Genes Dev 11: 2600–2621.
Roselius K, Stephan W, Stadler T (2005). The relationship of nucleotide polymorphism, recombination rate and selection in wild tomato species. Genetics 171: 753–763.
Ross-Ibarra J (2004). The evolution of recombination under domestication: a test of two hypotheses. Am Nat 163: 105–112.
Schmid M, Nanda I, Hoehn H, Schartl M, Haaf T, Buerstedde JM et al. (2005). Second report on chicken genes and chromosomes 2005. Cytogenet Genome Res 109: 415–479.
Schultz J, Redfield H (1951). Interchromosomal effects on crossing over in Drosophila. Cold Spring Harb Symp Quant Biol 16: 175–197.
Shifman S, Bell JT, Copley RR, Taylor MS, Williams RW, Mott R et al. (2006). A high-resolution single nucleotide polymorphism genetic map of the mouse genome. PLoS Biol 4: 2227–2237.
Sigurdsson MI, Smith AV, Bjornsson HT, Jonsson JJ (2009). HapMap methylation-associated SNPs, markers of germline DNA methylation, positively correlate with regional levels of human meiotic recombination. Genome Res 19: 581–589.
Slatkin M (2008). Linkage disequilibrium—understanding the evolutionary past and mapping the medical future. Nat Rev Genet 9: 477–485.
Smith JM, Haigh J (1974). Hitch-hiking effect of a favorable gene. Genet Res 23: 23–35.
Solignac M, Mougel F, Vautrin D, Monnerot M, Cornuet JM (2007). A third-generation microsatellite-based linkage map of the honey bee, Apis mellifera, and its comparison with the sequence-based physical map. Genome Biol 8: R66.
Solignac M, Vautrin D, Baudry E, Mougel F, Loiseau A, Cornuet JM (2004). A microsatellite-based linkage map of the Honeybee, Apis mellifera L. Genetics 167: 253–262.
Spencer CCA, Deloukas P, Hunt S, Mullikin J, Myers S, Silverman BW et al. (2006). The influence of recombination on human genetic diversity. PLoS Genet 2: 1375–1385.
Stahl FW, Foss HM, Young LS, Borts RH, Abdullah MFF, Copenhaver GP (2004). Does crossover interference count in Saccharomyces cerevisiae? Genetics 168: 35–48.
Stephan W, Langley CH (1998). DNA polymorphism in Lycopersicon and crossing-over per physical length. Genetics 150: 1585–1593.
Stephens JC, Schneider JA, Tanguay DA, Choi J, Acharya T, Stanley SE et al. (2001). Haplotype variation and linkage disequilibrium in 313 human genes. Science 293: 489–493.
Stevison LS, Noor MAF (2010). Genetic and evolutionary correlates of fine-scale recombination rate variation in Drosophila persimilis. J Mol Evol 71: 332–345.
Strathern JN, Shafer BK, Mcgill CB (1995). DNA-synthesis errors associated with double-strand-break repair. Genetics 140: 965–972.
Takahashi A, Liu YH, Saitou N (2004). Genetic variation versus recombination rate in a structured population of mice. Mol Biol Evol 21: 404–409.
Tenaillon MI, Sawkins MC, Anderson LK, Stack SM, Doebley J, Gaut BS (2002). Patterns of diversity and recombination along chromosome 1 of maize (Zea mays ssp mays L.). Genetics 162: 1401–1413.
Tenaillon MI, Sawkins MC, Long AD, Gaut RL, Doebley JF, Gaut BS (2001). Patterns of DNA sequence polymorphism along chromosome 1 of maize (Zea mays ssp mays L. Proc Natl Acad Sci USA 98: 9161–9166.
Tenaillon MI, U'Ren J, Tenaillon O, Gaut BS (2004). Selection versus demography: a multilocus investigation of the domestication process in maize. Mol Biol Evol 21: 1214–1225.
Thuriaux P (1977). Is recombination confined to structural genes on eukaryotic genome. Nature 268: 460–462.
True JR, Mercer JM, Laurie CC (1996). Differences in crossover frequency and distribution among three sibling species of Drosophila. Genetics 142: 507–523.
Tsai IJ, Burt A, Koufopanou V (2010). Conservation of recombination hotspots in yeast. Proc Natl Acad Sci USA 107: 7847–7852.
Wilfert L, Gadau J, Schmid-Hempel P (2007). Variation in genomic recombination rates among animal taxa and the case of social insects. Heredity 98: 189–197.
Winckler W, Myers SR, Richter DJ, Onofrio RC, McDonald GJ, Bontrop RE et al. (2005). Comparison of fine-scale recombination rates in humans and chimpanzees. Science 308: 107–111.
Wong AK, Ruhe AL, Dumont BL, Robertson KR, Guerrero G, Shull SM et al. (2010). A comprehensive linkage map of the dog genome. Genetics 184: 595–605.
Yauk CL, Bois PRJ, Jeffreys AJ (2003). High-resolution sperm typing of meiotic recombination in the mouse MHC E-beta gene. EMBO J 22: 1389–1397.
Yi S, Summers TJ, Pearson NM, Li WH (2004). Recombination has little effect on the rate of sequence divergence in pseudoautosomal boundary 1 among humans and great apes. Genome Res 14: 37–43.
Yi SJ, Li WH (2005). Molecular evolution of recombination hotspots and highly recombining pseudoautosomal regions in hominoids. Mol Biol Evol 22: 1223–1230.
Acknowledgements
We thank the Noor lab group, Corbin Jones, Richard Kliman and two anonymous reviewers for helpful comments on the paper. Funding for research in the Noor laboratory was provided by NIH Grants GM076051, GM086445 and GM092501.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Smukowski, C., Noor, M. Recombination rate variation in closely related species. Heredity 107, 496–508 (2011). https://doi.org/10.1038/hdy.2011.44
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/hdy.2011.44
Keywords
This article is cited by
-
Emergence and influence of sequence bias in evolutionarily malleable, mammalian tandem arrays
BMC Biology (2023)
-
High heterogeneity in genomic differentiation between phenotypically divergent songbirds: a test of mitonuclear co-introgression
Heredity (2023)
-
Accurate recombination estimation from pooled genotyping and sequencing: a case study on barley
BMC Genomics (2022)
-
Genomic patterns of homozygosity and inbreeding depression in Murciano-Granadina goats
Journal of Animal Science and Biotechnology (2022)
-
Recombination mapping of the Brazilian stingless bee Frieseomelitta varia confirms high recombination rates in social hymenoptera
BMC Genomics (2021)