Abstract
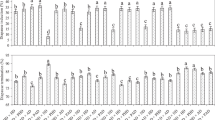
Sexual reproduction in cyclically parthenogenetic Daphnia species is a critical life history component. To investigate the nature of variation of the intensity of sexual reproduction (ISR) in Daphnia, and to measure the relative importance of different aspects of the variation, 30 random clones from a Daphnia pulicaria population were tested in a factorial design involving five photoperiods and two food concentrations. The results revealed that the ISR, indexed by the number of resting eggs produced, is influenced significantly by both the environmental conditions employed (photoperiod, food) and genetic factors (clonal effects within single environments and clone-photo-food interaction across environments). At the population level, a critical photoperiod exists (14 h light/day) for sexual reproduction to be initiated. There are significant differences among the estimates of the genetic variation for the ISR across some environments, signifying that the measurement of genetic variability in one environment has limited value for inferring the magnitude of genetic variability in other environments. There is highly significant genotype-environment (G × E) interaction, accounting for about 37.5 per cent of the total variance of the ISR. The results are discussed in the context of the population's adaptation to the ecological surroundings and the maintenance of genetic variability for the ISR in a particular environment.
Similar content being viewed by others
Article PDF
References
Banta, A M. 1939. General characterization of Cladocera. In: Banta, A. M. (ed.) Studies on the Physiology, Genetics and Evolution of Some Cladocera, pp. 2–12. Carnegie Institute, Washington,DC.
Banta, A M, and Brown, L A. 1939. Control of male and sexual egg reproduction. In: Banta, A. M. (ed.) Studies on the Physiology, Genetics and Evolution of Some Cladocera, pp. 106–129. Carnegie Institute, Washington DC.
Bell, G. 1990. The ecology and genetics of fitness in Chlamydomonas. I. Genotype-by-environment interaction among pure strains. Proc R Soc B, 240, 295–321.
Berg, K. 1934. Cyclic reproduction, sex determination and depression in the Cladocera. Biol Rev, 9, 139–174.
Brooks, J L. 1957. The Systematics of North American Daphnia. The Academy, New Haven, CT.
Bulmer, M G. 1980. The Mathematical Theory of Quantitative Genetics. Oxford University Press, New York.
Carvalho, G R, and Hughes, R H. 1983. The effects of food availability, female culture density, and photoperiod on ephippia production in Daphnia magna Straus (Crustacea: Cladocera). Freshwater Biol, 13, 37–46.
Carvalho, G R, and Wolf, H G. 1989. Resting eggs of lake-Daphnia. I. Distribution, abundance and hatching of eggs collected from various depths in lake sediments. Freshwater Biol, 2, 459–470.
Crease, T J, Lynch, M, and Spitze, K. 1990. Hierarchical analysis of population genetic variation in mitochondrial and nuclear genes of Daphnia pulex. Mol Biol Evol, 7, 444–485.
D'Ambramo, L R. 1980. Ingestion rate decrease as the stimulus for sexuality in populations of Moina macrocopa. Limnol Oceanogr, 25, 422–429.
De Meester, L. 1993. Inbreeding and outbreeding depression in Daphnia. Oecologia, 96, 80–84.
Falconer, D S. 1952. The problem of environment and selection. Am Nat, 86, 293–298.
Falconer, D S. 1981. Introduction to Quantitative Genetics, 2nd edn. Longman, New York.
Ferrari, D C, and Hebert, P D N. 1982. The induction of sexual reproduction in Daphnia magna: genetic differences between arctic and temperate populations. Can J Zool, 60, 2143–2148.
Fry, J D. 1992. The mixed-model analysis of variance applied to quantitative genetics: biological meaning of parameters. Evolution, 46, 540–550.
Gillespie, J H, and Turelli, M. 1989. Genotype-environmental interactions and maintenance of polygenic variation. Genetics, 121, 129–138.
Gupta, A P, and Lewontin, R C. 1982. A study of reaction norms in natural populations of Drosophila pseudoobscura. Evolution, 36, 934–938.
Hairston, N G, and Dillon, T A. 1990. Fluctuating selection and response in a population of freshwater copepods. Evolution, 44, 1796–1805.
Hebert, P D N. 1987. Genetics of Daphnia. In: Peters, R. H. and de Bernardi, R. (eds) Daphnia Memorie Istituto Italiano Di Idrobiologia, 45, 439–460.
Hebert, P D N, and Beaton, M J. 1989. Methodologies for Allozyme Analysis Using Cellulose Acetate Electrophoresis: a Practical Handbook. Helena Laboratories, Beaumont, TX.
Hebert, P D N, Ward, R D, and Weider, L J. 1988. Clonal-diversity patterns and breeding-system variation in Daphnia pulex, an asexual-sexual complex. Evolution, 42, 147–159.
Hebert, P D N, Beaton, M J, and Schwartz, S S. 1989. Polyphyletic origin of asexuality in Daphnia pulex. I. Breeding system variation and levels of clonal diversity. Evolution, 43, 1004–1015.
Hobaek, A, and Larsson, P. 1990. Sex determination in Daphnia magna. Ecology, 71, 2255–2268.
Hutchinson, G E. 1967. A Treatise on Limnology, vol. 2. John Wiley&Sons, New York.
Korpelainen, H. 1986. Reproductive strategies of Daphnia magna genotypes. Hereditas, 106, 181–188.
Korpelainen, H. 1987. The effects of temperature and photoperiod on life-history parameters of Daphnia magna (Crustacea: Cladocera). Freshwater Biol, 16, 615–620.
Lande, R. 1979. Quantitative genetic analysis of multivariate evolution applied to brain:body size allometry. Evolution, 33, 402–416.
Lande, R, and Arnold, S J. 1983. The measurement of selection on correlated characters. Evolution, 37, 1210–1226.
Lynch, M. 1984. The limits of life history evolution in Daphnia. Evolution, 38, 465–482.
Lynch, M. 1985. Spontaneous mutations for life history characters in an obligate parthenogen. Evolution, 39, 804–818.
Lynch, M. 1989. The life history consequences of resource depression in Daphnia pulex. Ecology, 70, 246–256.
Lynch, M, Spitze, K, and Crease, T. 1989. The distribution of life-history variation in the Daphnia pulex complex. Evolution, 43, 1724–1736.
Mort, M A, and Wolf, H G. 1986. The genetic structure of large lake Daphnia populations. Evolution, 40, 756–766.
SAS Institute, 1990. SAS/STAT User's Guide, vols 1 and 2, Version 6, 4th edn. Cary, NC.
Searle, S R. 1971. Linear Models. Wiley, New York.
Service, P M, and Rose, M R. 1985. Genetic covariation among life-history components: the effects of novel environments. Evolution, 39, 943–945.
Sokal, R R, and Rohlf, R J. 1981. Biometry, 2nd edn. Freeman, New York.
Stearns, S C. 1992. The Evolution of Life Histories. Oxford University Press, New York.
Spitze, K. 1991. Chaoborus predation and life-history evolution in Daphnia pulex: temporal pattern of population diversity, fitness, and mean life history. Evolution, 45, 82–92.
Spitze, K, Burnson, J, and Lynch, M. 1991. The covariance structure of life-history characters in Daphnia pulex. Evolution, 45, 1081–1090.
Stross, G. 1987. Photoperiodism and phased growth in Daphnia populations: coactions in perspective. In: Peters, R. H. and de Bernardi, R. (eds) Daphnia Memorie Istituto Italiano Di Idrobiologia, 45, 367–388.
Stross, P M, and Hill, J C. 1965. Diapause induction in Daphnia requires two stimuli. Science, 150, 1462–1464.
Stross, P M, and Hill, J C. 1968. Photoperiod control of winter diapause in the freshwater crustacean, Daphnia. Biol Bull, 134, 176–198.
Tessier, A J, Young, A, and Leibold, M. 1992. Population dynamics and body size selection in Daphnia. Limnol Oceanogr, 37, 1–14.
Threlkeld, S T. 1987. Daphnia population fluctuations: patterns and mechanisms. In: Peters, R. H. and de Bernardi, R. (eds) Daphnia Memorie Istituto Italiano DiIdrobiologia, 45, 367–388.
Via, S. 1984. The quantitative genetics of polyphagy in an insect herbivore. I. Genotype-environment interaction in larval performance on different host plant species. Evolution, 38, 881–895.
Via, S, and Lande, R. 1985. Genotype-environment interaction and the evolution of phenotypic plasticity. Evolution, 39, 505–522.
Via, S, and Lande, R. 1987. Evolution of genetic variability in a spatially heterogeneous environment: effects of genotype-environment interaction. Genet Res, 49, 147–156.
Wade, M J. 1990. Genotype-environment interaction for climate and competition in a natural population of flour beetles, Tribolium castaneum. Evolution, 44, 2004–2011.
Yamada, Y. 1962. Genotype-environment interaction and genetic variation of the same trait under different environments. Jap J Genet, 37, 498–509.
Yampolsky, L YU. 1992. Genetic variation in the sexual reproduction rate within a population of a cyclical parthenogen, Daphnia magna. Evolution, 46, 833–837.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Deng, HW. Environmental and genetic control of sexual reproduction in Daphnia. Heredity 76, 449–458 (1996). https://doi.org/10.1038/hdy.1996.67
Received:
Issue Date:
DOI: https://doi.org/10.1038/hdy.1996.67
Keywords
This article is cited by
-
Transcriptome analysis of different life-history stages and screening of male-biased genes in Daphnia sinensis
BMC Genomics (2022)
-
Effect of habitat permanence on life-history: extending the Daphnia model into new climate spaces
Evolutionary Ecology (2021)
-
Sex in crowded places: population density regulates reproductive strategy
Hydrobiologia (2020)
-
Development of resistance to cyfluthrin and naphthalene among Daphnia magna
Ecotoxicology (2009)
-
Overcrowding, food and phosphorus limitation effects on ephipphia production and population dynamics in the invasive species Daphnia lumholtzi
Hydrobiologia (2009)