Abstract
The spontaneous autoimmune peripheral polyneuropathy (SAPP) model in B7-2 knockout non-obese diabetic mice shares clinical and histological features with chronic inflammatory demyelinating polyradiculoneuropathy (CIDP). Secondary axonal loss is prominent in the progressive phase of this neuropathy. Neurotrophin 3 (NT-3) is an important autocrine factor supporting Schwann cell survival and differentiation and stimulates neurite outgrowth and myelination. The anti-inflammatory and immunomodulatory effects of NT-3 raised considerations of potential efficacy in the SAPP model that could be applicable to CIDP. For this study, scAAV1.tMCK.NT-3 was delivered to the gastrocnemius muscle of 25-week-old SAPP mice. Measurable NT-3 levels were found in the serum at 7-week postgene delivery. The outcome measures included functional, electrophysiological and histological assessments. At week 32, NT-3-treated mice showed increased hind limb grip strength that correlated with improved compound muscle action potential amplitude. Myelinated fiber density was 1.9 times higher in the NT-3-treated group compared with controls and the number of demyelinated axons was significantly lower. The remyelinated nerve fiber population was significantly increased. These improved histopathological parameters from scAAV1.tMCK.NT-3 treatment occurred in the setting of reduced sciatic nerve inflammation. Collectively, these findings suggest a translational application to CIDP.
Similar content being viewed by others
Introduction
Chronic inflammatory demyelinating polyradiculoneuropathy (CIDP) is an acquired immune-mediated disorder affecting the peripheral nerves, with a prevalence of 9/100 000.1 The clinical features of the disease include weakness, sensory loss, imbalance and impaired ambulation, which may lead to substantial disability. The first line of the treatment consists of corticosteroids, plasma exchange and intravenous immunoglobulin, all of which have been proven to be beneficial in randomized controlled trials2, 3, 4, 5 but their efficacy differs in terms of cost, availability and adverse effects. Only 50–80% of patients with CIDP respond satisfactorily to the established first-line immunomodulatory treatments.6, 7 Refractory forms of CIDP require immune suppressants, such as azathioprine, cyclosporine, cyclophosphamide, interferons, methotrexate, mycophenolate mofetil and rituximab. Unfortunately, these drugs are not free from serious side effects and are only beneficial in a small percentage of patients. CIDP is also commonly associated with long-term dependence on treatment. Studies have shown that 55% of responders continue taking immunoglobulins, 23% remain on plasmapheresis and 18% continue on prednisone.8 These limitations in treatment provide compelling reasons to identify new therapies for CIDP. The most appealing approaches would provide neuroprotection and immunomodulation with the goal of normalizing nerve function. Minimizing secondary axonal loss following the primary demyelinating insult and promoting nerve regeneration and remyelination are important to the recovery process.
The spontaneous autoimmune peripheral polyneuropathy (SAPP) mouse develops an autoimmune neuropathy with auto-reactive T cells directed against myelin proteins, especially affecting female mice around 20 weeks of age. The model represents a progressive, unremitting chronic inflammatory demyelinating polyneuropathy with axonal loss resulting in severe weakness by 25 weeks of age.9, 10 This rodent disease is further characterized by a reduction in T regulatory cells (Tregs) (CD4+CD25+Foxp3+) and has significant peripheral nerve infiltration of immune cells, such as CD11c+ dendritic cells (DCs), CD4+ and CD8+ T lymphocytes.11, 12 In CIDP patients, a similar pathogenic role was identified for CD4+ and CD8+ T cells with an accompanying reduction in circulating Tregs.13, 14, 15 Previous studies showed that DC-mediated delivery of vasoactive intestinal peptide into SAPP mice attenuated the disease-protecting myelinated fibers as well as showing a sustained immunomodulatory effect.16 A more aggressive treatment would anticipate a reversal of the natural history of SAPP mice with applicability for resolution of CIDP. In order to achieve clinical resolution, the following requirements must be fulfilled: protection against nerve fiber loss, promotion of remyelination of regenerating axons, and a robust ongoing anti-inflammatory capability. NT-3 has the potential to succeed in all of these prerequisites because of demonstrated efficacy in preclinical and clinical peripheral nerve trials. In the tremblerJ (TrJ) mouse model of demyelinating neuropathy and in Charcot–Marie–Tooth type 1 A (CMT1A) patients, subcutaneous delivery of NT-3 resulted in improved peripheral nerve histopathology, regeneration and function.17 However, the need for repeated NT-3 injections, a very short half-life and a limited commercial supply led to our initial study using intramuscular delivery of scAAV1.NT-3 to the gastrocnemius muscle of TrJ mice.18 In this study, we measured sustained levels of NT-3 in the serum and a corresponding improvement in motor function, histopathology and compound muscle action potential (CMAP) amplitude of peripheral nerves. The specificity and safety of vector delivery was further assured through the use of a triple muscle-specific creatine kinase promoter (tMCK) with added potency delivered through the application of a self-complementary (sc) AAV1. These studies have now provided the impetus for testing the scAAV1.tMCK.NT-3 in non-inherited neuropathies. SAPP neuropathy is an obvious target with implications for CIDP. The potential for treatment of an inflammatory neuropathy is further boosted because of the selective ligand binding of NT-3 to the tyrosine-kinase receptor (TrkC) paving the necessary path for immune modulation through the production of interleukin (IL)-4 by TrkC-expressing T helper cell 2 (Th2) cells.19 The anti-inflammatory effect of NT-3 was recently shown in the experimental encephalomyelitis model through secretion of IL-10 by NT-3-transduced stem cells.20, 21 Success in neuroprotection and immunomodulation by NT-3 potentially fulfills the requirements necessary for the treatment of SAPP and its clinical correlate CIDP.
Results
NT-3 vector and potency
The human NT-3 cDNA (GeneBank designation NTF3, referenced in this paper as NT-3) coding sequence under the control of tMCK promoter was packaged using sc AAV1 vector (Figure 1a; similar to prior studies18). For this study, potency of the scAAV1.tMCK.NT-3 vector was tested in SAPP mice 25 weeks of age by delivering 3 × 1010 vg (vector genome) per mouse via intramuscular injection (1.5x1010 vg for each gastrocnemius muscle). At 4 and 7 weeks postinjection, NT-3 was expressed from muscle and secreted into the serum (NT-3 serum levels: 4 weeks, treated: 230.7±139 vs control: 44.3±25 pg ml−1, P<0.02; 7 weeks treated: 355±177.5 vs control: 21.5±6.9 pg ml−1, P<0.01) (Figure 1b). In all phases of this project, a cohort of phosphate-buffered saline (PBS)-injected SAPP mice served as a comparative control group.
scAAV1.tMCK.NT-3 vector and NT-3 serum levels. (a) NT-3 expression is driven by a tMCK in the sc AAV vector backbone. The diagram shows the cassette composed of a tMCK enhancer/promoter region (714 bp), the full-length NT-3 cDNA (774 bp) and the SV40 polyA tail (211 bp). (b) SAPP mice were injected with a total of 3 × 1010 vg of scAAV1.tMCK.NT-3 divided into the left and right gastrocnemius muscles (1.5 × 1010 vg in 20 μl PBS per each muscle) or PBS alone as control. Serum samples were obtained from SAPP mice via eye bleeding at 4 and 7 weeks postinjection, and NT-3 serum levels were determined by using enzyme-linked immunosorbent assay. Error bars: s.e.m. (n=8 per group). Statistical significance: two-tailed t-test (*P<0.05, **P<0.01).
Therapeutic effect of AAV1.tMCK.NT-3 in SAPP model
We tested the neuroprotective and immunomodulatory effects following intramuscular injection of scAAV1.tMCK.NT-3 in the 25-week-old SAPP mouse. Typically, mild-to-moderate weakness of fore or hind limbs is present at 21–23 weeks of age and rapidly becomes more severe by 25 weeks at which time widespread peripheral nerve inflammation is present.10, 16 We targetted this age as the baseline time point for sc.AAV1.tMCK.NT-3 injection to ensure that both cohorts were uniformly affected clinically prior to a rapidly evolving motor paralysis. At this 25 weeks of age, the clinical scores of treated and untreated/control cohorts were identical (scored around three corresponding to severe fore or hind limb weakness;10, 16 n=8 in each group).
Clinical observations and functional studies
The effect of NT-3 gene therapy on bilateral hind and fore limb grip strength were assessed weekly in the SAPP mice, at weeks 26–32. Bilateral hind limb grip strength performances showed gradual steady improvement in the treated group while the controls declined. At 29 weeks, the NT-3 cohort was clearly better and by week 32 force generation in the treated group was 67% greater than controls (AAV1.NT-3: 89.7±11.7 vs control: 29.6±4.9 g; P=0.003; Figure 2a). Fore limb grip strength performance in treated and controls increased in parallel until week 29 after which they diverged with the NT-3 cohort continuing to gain strength through 32 weeks of age (NT-3: 66.8±4.1 vs PBS control: 45±5.0 g, P<0.05; Figure 2b).
Functional improvements in SAPP mice following AAV1.NT-3 treatment. Treated group had increased hind limb (a) and fore limb (b) grip strength compared with the PBS-injected control group. Error bars represent s.e.m. Statistical significance between the AAV1.NT-3 group (n=8) and control group (n=8) was calculated by using multiple t-test; one per row was performed; *P<0.05.
Electrophysiological studies
End point sciatic nerve conduction studies at 32 weeks revealed the efficacy of scAAV1.tMCK.NT-3 treatment correlating with the functional studies. The CMAP amplitudes were 38.3% higher in the treated group compared with control SAPP mice (AAV1.NT-3: 9.65±1.3 vs control: 5.9±0.6 mV; P=0.04; Figure 3a). The mean conduction velocity from the treated SAPP mice was slighly higher than the PBS-injected controls suggestive of a trend towards improvement (Figure 3b).
Nerve conduction analysis. AAV1.NT-3 gene therapy improved the CMAPs in the SAPP mice. The end point CMAP amplitude is 38% higher in the treated group (a). Conduction velocities were not found significantly different between the two groups (b). Error bars represent s.e.m. Statistical significance between the AAV1.NT-3 and PBS control group (n=8 per group) was calculated by using two-tailed t-test; *P<0.05.
Morphological studies
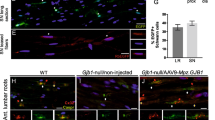
The sciatic nerve at baseline prior to treatment shows striking changes, including mononuclear leukocyte infiltration, endoneurial edema, demyelination, Wallerian degeneration and a multifocal reduction in axonal density. Thinly myelinated and solitary naked axons are commonly encountered, as well as onion bulb formation indicative of repeated attempts at remyelination.16 At 32 weeks posttreatment with scAAV1.tMCK.NT-3 gene transfer, we observed both preserved myelinated fibers and abundant thinly remyelinated nerve fibers (Figures 4a and b). This was strikingly different from the nerves of the PBS-injected SAPP mice where we found prominent lipid laden macrophages indicative of Wallerian degeneration and marked reduction in the number of myelinated nerve fibers (Figure 4c). There were also numerous demyelinated/naked axons in the PBS control cohort (Figure 4d).
AAV1.NT-3 gene therapy protects myelinated fibers and enhances remyelination in sciatic nerve. One-micrometer-thick cross-sections from midsciatic nerve segments are shown. Representative pictures of relatively well preserved myelinated fibers (a) and numerous remyelinated axons with thin myelin sheath (b) from the AAV1.NT-3 injected group at 32 weeks of age are shown. PBS-injected group at the same age show lipid laden phagocytic cells and marked reduction of the myelinated fibers (c) as well as numerous demyelinated/naked axons (arrows) (d). Scale bar=10 μm. (e) Myelinated axon size distribution histograms from AAV1.NT-3 and control SAPP mice at 7 weeks postinjection. Composite histograms for myelinated fiber densities (number mm−2) were generated from n=5 mice in each group; error bars represent s.e.m. (f) Scatter plot shows g ratios of individual fibers as a function of the respective axon diameter (n=5 mice per group; a total number of 1938 treated and 926 untreated axons were analyzed that were obtained from four randomly selected images, photographed at × 100 in each mouse nerve). The AAV1.NT-3 gene therapy increased the number of remyelinated fiber population across all axon diameters but predominantly of the small and medium diameter axons. (g) G ratio percentage of distribution histogram shows a shift to left, indicating an overall increase in the number of axons with thicker myelin compared with the PBS-treated control group.
The histological outcome of NT-3 treatment was quantified by determining myelinated fiber and demyelinated axon densities. The results confirmed that myelinated fiber density was 1.9 times higher in the NT-3-treated group compared with PBS controls (9209±1913 vs 4739±800.3 mm−2, P<0.04; n=6 in each group; Table 1). This end point mean myelinated fiber density in the treated group was 15% greater than 25-week-old untreated baseline control SAPP mice (7771.8±1596 mm−2; n=11; quantified and reported previously).16 This emphasizes both an NT-3-related protective effect on myelinated fibers and increase in the small diameter myelinated fiber population that corresponds to regeneration-associated myelination as illustrated in the myelinated axon size distribution histogram (Figure 4e). The number of demyelinated/nude axons was also significantly lower in the NT-3-treated group (613.1±98.72 vs 1,120±114.5 mm−2, P<0.013), corresponding to a significant protection of myelinated fibers (Table 1). Moreover, g ratio (axon diameter/fiber diameter) determinations of the myelinated fibers showed that scAAV1.tMCK.NT-3 gene therapy significantly increased the remyelinated fiber population across all axon diameters but more notably of small and medium size axons (Figure 4f). G ratio percentage distribution histogram showed a shift to the left indicating an overall increase in the number of axons with thicker myelin (Figure 4g). Collectively, these observations are consistent with our previous studies showing NT-3-induced improvements in regeneration and remyelination in TrJ and CMT1A.17, 18 It should also be noted that these preserved/improved histopathological parameters resulting from scAAV1.tMCK.NT-3 treatment occurred in the setting of a significant reduction in inflammation quantified by CD3+ cell counts in SAPP NT-3-treated mice compared with controls (NT-3: 324±204.1 vs control: 1010±343.6 cells mm−2, P<0.05) (Table 1, Figures 5a–c).
Anti-inflammatory, immunomodulatory effect of AAV1.NT-3 treatment. Reduction of CD3+ T cells' infiltration in sciatic nerve samples of the AAV1.NT-3 group (a) compared with the control (PBS) group (b) at 32 weeks of age. Scale bar=10 μm. (c) Quantitative data for CD3+ cell counts, expressed as number mm−2 cross-sectional area of sciatic nerves (derived from four randomly photographed areas at × 40 from each nerve; obtained from eight mice per group). Expression levels of IL-1β, TNF-α, IL-10 and Foxp3 in sciatic nerves (d). Total RNA was extracted from snap-frozen sciatic nerve samples and analyzed for expression by using real-time quantitative PCR. The data was normalized to GAPDH. Error bars: s.e.m. (n=8 per group; two-tailed t-test; *P<0.05, **P<0.01), AU: arbitrary units. At 32 weeks, splenocytes were isolated and analyzed for Tregs. (e) A representative flow cytometry images of CD4+CD25+Foxp3+ Tregs gated from CD4+ T cells from the spleen. (f) Frequency of CD4+CD25+Foxp3+ Tregs in the spleen. Error bars: s.e.m. (n=8 per group). Statistical significance: two-tailed t-test (*P<0.05, **P<0.01). (g) DCs isolated from SAPP mice (n=6) were exposed to 1 μg ml−1 of LPS in the presence and absence of recombinant NT-3 (100 μg) for 24 h. Cell culture supernatants were collected and analyzed for IL-10 and TNF-α cytokines by using enzyme-linked immunosorbent assay. Statistical significance: one-way analysis of variance followed by Tukey's multiple comparison test; (*P<0.05, ***P<0.001).
Immunomodulatory effect of NT-3 in SAPP mice peripheral nerves
We further characterized this immunomodulatory effect of treatment using real-time PCR comparing the expression of pro- and anti-inflammatory cytokines. Real-time PCR studies of sciatic nerves revealed that the NT-3-treated group exhibited significantly reduced expression of tumor necrosis factor (TNF)-α and IL-1β but increased expression of IL-10 and Foxp3 (forkhead box P3) (Figure 5d). In order to see whether scAAV1.NT-3 treatment increased the percentage of CD4+CD25+Foxp3+ Tregs, known to be persistently low in SAPP mice, we analyzed the splenocytes by flow cytometry. We found a significant increase in the percentage of Tregs in the treated group, which is compatible with an immune-regulatory role of NT-3 (Figures 5e and f).
DC infiltration is an important hallmark of nerve inflammation in the SAPP model.11 In our previous study, we showed that vasoactive intestinal peptide-expressing tolerogenic DCs attenuated the inflammation in the nerves of SAPP mice.16 To investigate whether NT-3 treatment causes DCs to gain a tolerogenic feature, we exposed bone marrow-derived DCs to lipopolysaccharide (LPS) in the presence or absence of recombinant human NT-3 (DC characterization: Supplementary Figure S1). At 24 h of incubation with NT-3 and LPS, DCs showed more than twofold increase of IL-10 secretion and lesser amount of TNF-α compared with DCs exposed to LPS alone (Figure 5g). Overall, these results support an immunoregulatory role of NT-3 in favor of tolerogenicity providing neuroprotection and safeguards against the inflammatory demyelinating process.
Discussion
In the present study, scAAV1.tMCK.NT-3 delivered directly to the gastrocnemius muscle of 25 week-old SAPP mice exhibited a potent anti-inflammatory, immunomodulatory and neuroprotective effects. SAPP mice at this age display an evolving motor paralysis, accompanied by reduction in CMAP amplitudes and conduction velocities. The sciatic nerve shows diffuse demyelination and axonal loss, with mononuclear cell infiltration. This mouse model is a prototypical example for CIDP; there is no treatment and paralysis continues unabated after 30 weeks of age without recovery.10 We documented in this mouse model that gene transfer to muscle leads to the secretion of NT-3 into the serum. This results in functional benefits to the SAPP mouse, including improved hind limb and preserved fore limb grip strength accompanied by improved CMAP amplitudes in the sciatic nerve. These features of neuroprotection correlated with significantly more myelinated fibers (counts included both uninjured axons with normal myelin thickness and remyelinated axons with thinner myelin for axon size), nearly twice as many as the PBS controls (9209±1913 vs 4739±800.3 mm−2, P<0.04). In addition, there was a robust increase in the smaller diameter myelinated fiber population (with axon diameter <3 μm) compatible with improved axonal regeneration and associated remyelination. G ratio determinations clearly showed a significantly increased remyelinated fiber population, with a shift to an overall increase in the number of axons with thicker myelin.
An anti-inflammatory and immunomodulatory effect was also unequivocally demonstrated following AAV1.NT-3 delivery. The number of infiltrating CD3+ T cells to the sciatic nerve was decreased, and reverse transcriptase PCR studies of sciatic nerve revealed reduced expression of TNF-α and IL-1β with an increase in the expression of IL-10 and Foxp3 in sciatic nerves. We detected significantly higher expression of IL-10, which is likely to have a major role in this anti-inflammatory effect as demonstrated previously.20, 21 Of particular importance in the treatment of this inflammatory neuropathy was the finding that CD4+CD25+Foxp3+ Tregs were increased in the spleens of treated mice, suggesting a major contribution to generation of immunoregulatory outcomes.
We also explored the possibility that NT-3 has a role in triggering tolerogenic responses in DCs following LPS exposure. A direct correlation was reported between the numbers of myeloid DCs in the cerebrospinal fluid and clinical disability in CIDP patients.22 In the experimental counterpart of the SAPP mouse model, it was observed that the time of significant DCs infiltration in the sciatic nerves corresponds to severe paralytic state.11, 12 Previously, we have shown that DCs having a tolerogenic phenotype can mitigate the inflammation in the sciatic nerves of SAPP mice.16 Based on the high levels of IL-10 and low levels of TNF-α in the sciatic nerves from the treated mice, we predicted that NT-3 was capable of inducing tolerogenic features on DCs in our model. This possibility was tested by using an in vitro assay that showed DCs exposed to recombinant NT-3 and LPS secreted more than two times higher amount of IL-10 and less amount of TNF-α in support of the premise that NT-3 may facilitate DCs to gain tolerogenic features. Further studies are needed to understand the mechanism(s) by which NT-3 effects DCs or how it is influencing the Treg population.
It should be noted that these significantly better preserved/improved histopathological parameters occurred in the setting of decreased inflammation compared with untreated controls emphasizing the anti-inflammatory function of NT-3 in addition to its traditionally known biological effects of improved regeneration and associated remyelination. NT-3 belongs to the neurotrophin family of growth factors and widely expressed throughout the nervous system, including dorsal ganglia and spinal motor neurons as well as both peripheral nerve system and central nervous system glia, Schwann cells and oligodendrocytes.23, 24, 25, 26, 27 Although NT-3, similar to other neurotrophins, was initially identified because of its essential function in the nervous system development, myelination, growth and regeneration control, a much broader effect is now increasingly being recognized extending to a wide range of cell types, including immune cells and participation in inflammatory responses. Studies suggest that NT-3 has a critical role in the regulation or maintenance of the Th1/Th2 balance through interaction with its receptor, TrkC, expressed by Th2 cells.19 NT-3 acts as a trigger for cytokine production in TrkC-expressing Th2 cells but not in Th1 cells. Thus the NT-3/TrkC system is thought to be involved in the induction or maintenance of Th2-dependent immunity. Moreover, specifically Th2 cells, which express TrkC, are required for NT-3-induced axonal sprouting in the injured nervous system.28, 29, 30 Th2 cells are known to express the neuroprotective cytokines IL-4, IL-5, IL-10 and IL-1329 as well as the neurotrophic factors brain-derived neurotrophic factor and NT-331, 32 that was proposed to act directly on the injured axons to allow sprouting.30
The outcomes in this translational study of inflammatory neuropathy in the SAPP mouse have application to CIDP, a human disease with similar features. NT-3 gene therapy could potentially circumvent the use of chronic corticosteroid treatment and/or eliminate long-term intravenous immunoglobulin usage to maintain clinical stability.33 Also relevant to the findings in the present study are our previous observations demonstrating biological effects of NT-3 in both TrJ mice (experimentally) and in humans with CMT1A (clinically). In these earlier studies in both species, NT-3 was delivered subcutaneously three times per week.17 This approach is impractical requiring chronic, sustained delivery to maintain efficacy using a product with short half-life and limited availability, but the findings in the TrJ mouse showed translational application that predicted efficacy in a human clinical trial for CMT1A patients treated for 6 months resulting in an increase myelinated fiber density and improved myelination of regenerating axons.17 Our follow-up studies in TrJ mice using intramuscular delivery of scAAV1.NT-3 established the therapeutic dose and a preferential muscle-specific promoter to achieve sustained NT-3 levels to serve as a template for future CMT1A clinical trials with a potential to extend treatment to other nerve diseases.18
Further advantages of NT-3 are bestowed by the anti-inflammatory benefits provided by a secreted pool of immunomodulatory cytokines. We clearly documented this following AAV1.NT-3 gene delivery to the inflamed peripheral nerve of the SAPP mouse. This finding in our peripheral nerve system study is reinforced in recent reports showing that delivery of NT-3 has an equally robust effect in the central nervous system combating experimental encephalomyelitis whether delivered by genetically engineered embryonic stem cell-derived microglia20 or by bone marrow-derived neural stem cells.21 Comparable to what we observed in an inflammatory peripheral nervous system disorder, experimental encephalomyelitis mice treated with NT-3 showed a similar neuroprotective outcome characterized by neuronal sprouting, less axonal injury and reduced demyelination.
The translational prospects of the potential use of NT-3 delivery whether by virus or stem cells, and whether used for central nervous system or peripheral nerve system disease, are clearly illustrated by these preclinical studies that pave a potential path forward for clinical trial. The applicability for peripheral nerve disease is particularly attractive given that we can achieve efficacy by an easily accessible route of intramuscular injection. Of particular interest as a disease target for treatment are cases of CMT1A neuropathy with superimposed inflammation (atypical CIDP)34, 35 where the therapeutic effect of this neurotrophin could facilitate both nerve regeneration, remyelination and, at the same time, combat the secondary inflammation that leads to even greater debilitation than the inherited disease alone.
Materials and methods
Generation of scAAV.1.tMCK.NT-3 vectors
The design of scAAV1.tMCK.NT-3 expression cassette was previously published by our group.18 AAV1 vector production and purification was carried out by our viral vector core at Nationwide Children’s Hospital, and the final titer (vg ml−1) was determined by quantitative reverse transcriptase PCR using the specific primers and probes for tMCK promoter utilizing a Prism 7500 Real-time detector system (PE Applied Biosystems, Grand Island, NY, USA). Aliquoted viruses were kept in −80 °C until use.
Enzyme-linked immunosorbent assays
Blood sample were collected from treated and non-treated mice by performing eye bleeding under anesthesia at 4 and 7 weeks postinjection. The sera of mice were separated by centrifugation, and NT-3 levels were detected by using the Anti-NT-3 ELISA Kit (Abcam, Cambridge, MA, USA) following the manufacturer’s instructions.
Animals, treatment and monitoring
A breeding pair of SAPP mice was obtained from the Baylor College of Medicine. All mice were maintained at our animal facility of Nationwide Children's Hospital, and the experiments were performed according to the guidelines approved by the Research Institute at Nationwide Children’s Hospital Animal Care and Use Committee. Clinical scoring of SAPP mice were performed as previously published.10 To avoid the clinical variability among the mice, we selected 25 weeks of age as the time point for AAV1.tMCK.NT-3 injection at which time both cohorts were clinically uniformly affected prior to a rapidly evolving motor paralysis. The clinical scores of treated and untreated/control cohorts were identical (scored around three corresponding to severe fore or hind limb weakness; n=8 in each group) at this baseline time point. The experimental group was injected with a total of 3 × 1010 vg of scAAV1.tMCK.NT-3, divided into the left and right gastrocnemius muscles (1.5x1010 vg in 20 μl PBS per muscle); control group received PBS alone. Functional tests included bilateral fore and hind grip strength test and were performed weekly as previously described.36 All outcome evaluations were done in a blinded manner. Animal identification was obtained after the data collection was completed.
Isolation of DCs and splenocytes
DCs were isolated as previously described.37 Briefly, the bone marrow cells flushed from femur and tibiae of 4-week-old male SAPP mice and cultured in 150 mm petri dishes with RPMI medium containing 10% fetal bovine serum, glutamax, 20 ng ml−1 recombinant mouse granulocyte macrophages colony-stimulating factor, 1% Penicillin–Streptomycin Amphotericin B and 50 μM 2-mercaptoethanol. The medium was replaced every other day, and the cells were analyzed for the DC markers on days 8–9. Splenocytes were isolated at 32 weeks by taking the spleens and mashing them in a sterile strainer with 40-μm pore size in a 10-cm petri dish until getting a single-cell suspension in RPMI medium with 10% fetal bovine serum and 1% Penicillin–Streptomycin Amphotericin B. The cells were centrifuged and resuspended in ammonium chloride–potassium buffer (Life Sciences Cambrex, Walkersville, MD, USA) and incubated for 5 min to get rid of erythrocytes. Then the cells were washed by PBS, counted and analyzed for Tregs by using flow cytometry.
Flow cytometry analysis
Monoclonal anti CD3-FITC, CD4-PE and CD25-APC antibodies were purchased from BD Pharmingen (San Diego, CA, USA). Anti-Foxp3-FITC were purchased from eBioscience (San Diego, CA, USA). For Treg analysis, 5x106cells from the spleens of each mouse were first fixed with 1% paraformaldehyde and incubated with anti-Foxp3 antibody in 0.05% Tween-20 in PBS overnight at 4 °C followed by washing and incubating with anti-CD4 PE and anti CD25 APC antibodies in 100 μl PBS for 45 min. The excess amount of antibodies was washed twice with 1 ml PBS followed by centrifugation and re-suspending the cells in 200 μl PBS for analysis. All flow cytometry analyses were done by using FACSCalibur flow cytometer (BD Biosciences, Mountain View, CA, USA) and were computed by the Flow Jo software (Tree Star, Ashland, OR, USA).
Quantitative real-time PCR analysis
Total RNA was extracted from snap-frozen sciatic nerve samples of treated and control mice for the expression analysis of cytokines (IL-1β, TNF-α, IL-10 and Foxp3) by using Trizol (Life Sciences Cambrex). cDNAs were synthesized using the Transcriptor High Fidelity cDNA Synthesis Kit (Roche, GmbH, Mannheim, Germany). All quantitative PCR analysis was performed using iTaq Universal SYBR Green Supermix (Bio-Rad, (Hercules, CA, USA) according to the manufacturer's instructions. The primer sequences for cytokines, housekeeping gene GAPDH (glyceraldehyde 3-phosphate dehydrogenase) and Foxp3 were obtained from the literature.38, 39 Real-time PCR analyses were performed by using ABI 7500 real-time PCR system (Applied Biosystems, Foster City, CA, USA) and the data were computed by using the DataAssist Software (Life Sciences Cambrex).
Histological analysis
Mice were killed quickly by an over-dosage of xylazine/ketamine anesthesia at 32 weeks of age, 7 weeks post-AAV1.NT-3 injection. Both sciatic nerves were removed; one side was snap frozen for quantitative PCR analysis. The proximal half of the other side in its in situ length was fixed in 3% glutaraldehyde in 0.1m phosphate buffer and further processed for plastic embedding according to established methods in our laboratory.36 The distal half was fixed in 4% paraformaldehyde overnight, cryoprotected and frozen in isopentane, cooled in liquid nitrogen. In 12-μm thick cross-sections from frozen sciatic nerves, CD3+ T-cell infiltration was assessed with immunohistochemistry using standard protocols. At least four randomly selected frames photographed at × 40 objective were used for counting of CD3+ cells, and the data were expressed as the number per mm2.
Myelinated fiber and demyelinated axon density determinations
Quantitative analysis at the light microscopic level was performed on 1-μm-thick cross-sections from mid-sciatic nerves. In four randomly selected and digitally stored images photographed at × 100 objective, the number of myelinated fibers and demyelinated/nude axons were determined using the Axiovision software v.4.8.2.0 (Carl Zeiss MicroImaging, Munich, Germany) and expressed as the number per mm2 of endoneurial area. Axons undergoing Wallerian degeneration were excluded. Four randomly selected areas from six mice were analyzed in each group.
G ratio of the myelinated fibers
The g ratio refers to the ratio of axonal diameter/fiber diameter and lower g ratios represent axons with thicker myelin.40 For g ratio determinations, digitally stored four cross-sectional images of mid-sciatic nerves, captured at × 100 magnification from five mice in each group were utilized; the shortest axial lengths as axon diameters and fiber diameters were recorded with a calibrated micrometer, using the AxioVision 4.2 software (Carl Zeiss MicroImaging) as we described previously.36 The g ratio of individual fibers was plotted as function of the respective axon diameter. In addition, the g ratio distribution histograms were generated as the percentage of total fibers analyzed. Composites of myelinated fiber axon size distribution histograms were also generated from data obtained for the AAV1.NT-3 and PBS-injected groups.
Nerve conduction studies
Sciatic motor nerve conduction studies were performed bilaterally on each SAPP mouse under isoflurane anesthesia using a portable electrodiagnostic system (Synergy N2 EMG and nerve conduction study machine, Natus, Middleton, WI, USA) and techniques similar to that which we have previously described.41, 42 Briefly, the sciatic motor nerve conduction responses were recorded using two fine ring electrodes (Alpine Biomed, Skovlunde, Denmark) used as the active (E1) and reference (E2) electrodes. The active recording electrode was placed over the proximal portion of the gastrocnemius muscle and the reference electrode over the mid-metatarsal region of the foot. Using an irrigating syringe (Covidien, Mansfield, MA, USA), the skin under the ring electrodes was precisely coated with electrode gel (Spectra 360 by Parker laboratories, Fairfield, NJ, USA) to reduce skin impedance. A pair of 28-gauge monopolar needle electromyography electrodes (Teca, Oxford Instruments Medical, New York, NY, USA) was used to supramaximally stimulate the sciatic nerve 5 mm proximal to the E1 electrode and then at the sciatic notch. The parameters measured included CMAP amplitude, distal latency and conduction velocity.
Statistical analysis
GraphPad Prism software (La Jolla, CA, USA) was used for all statistical analyses. Statistical difference of two groups was calculated by using two-tailed t-test or one-way analysis of variance followed by Tukey's multiple comparison test when applicable.
References
Laughlin RS, Dyck PJ, Melton LJ 3rd, Leibson C, Ransom J, Dyck PJ . Incidence and prevalence of CIDP and the association of diabetes mellitus. Neurology 2009; 73: 39–45.
Mehndiratta MM, Hughes RA . Corticosteroids for chronic inflammatory demyelinating polyradiculoneuropathy. Cochrane Database Syst Rev 2012; 8: CD002062.
Mehndiratta MM, Hughes RA, Agarwal P . Plasma exchange for chronic inflammatory demyelinating polyradiculoneuropathy. Cochrane Database Syst Rev 2012; 9: CD003906.
Mendell JR, Barohn RJ, Freimer ML, Kissel JT, King W, Nagaraja HN et al. Randomized controlled trial of IVIg in untreated chronic inflammatory demyelinating polyradiculoneuropathy. Neurology 2001; 56: 445–449.
Eftimov F, Winer JB, Vermeulen M, de Haan R, van Schaik IN . Intravenous immunoglobulin for chronic inflammatory demyelinating polyradiculoneuropathy. Cochrane Database Syst Rev 2009; (1): CD001797.
Gorson KC . An update on the management of chronic inflammatory demyelinating polyneuropathy. Ther Adv Neurol Disord 2012; 5: 359–373.
Dalakas MC . Medscape. Advances in the diagnosis, pathogenesis and treatment of CIDP. Nat Rev Neurol 2011; 7: 507–517.
Viala K, Maisonobe T, Stojkovic T, Koutlidis R, Ayrignac X, Musset L et al. A current view of the diagnosis, clinical variants, response to treatment and prognosis of chronic inflammatory demyelinating polyradiculoneuropathy. J Peripher Nerv Syst 2010; 15: 50–56.
Kim HJ, Jung CG, Jensen MA, Dukala D, Soliven B . Targeting of myelin protein zero in a spontaneous autoimmune polyneuropathy. J Immunol 2008; 181: 8753–8760.
Ubogu EE, Yosef N, Xia RH, Sheikh KA . Behavioral, electrophysiological, and histopathological characterization of a severe murine chronic demyelinating polyneuritis model. J Peripher Nerv Syst 2012; 17: 53–61.
Salomon B, Rhee L, Bour-Jordan H, Hsin H, Montag A, Soliven B et al. Development of spontaneous autoimmune peripheral polyneuropathy in B7-2-deficient NOD mice. J Exp Med 2001; 194: 677–684.
Bour-Jordan H, Thompson HL, Bluestone JA . Distinct effector mechanisms in the development of autoimmune neuropathy versus diabetes in nonobese diabetic mice. J Immunol 2005; 175: 5649–5655.
Van den Berg LH, Mollee I, Wokke JH, Logtenberg T . Increased frequencies of HPRT mutant T lymphocytes in patients with Guillain-Barre syndrome and chronic inflammatory demyelinating polyneuropathy: further evidence for a role of T cells in the etiopathogenesis of peripheral demyelinating diseases. J Neuroimmunol 1995; 58: 37–42.
Schneider-Hohendorf T, Schwab N, Uceyler N, Gobel K, Sommer C, Wiendl H . CD8+ T-cell immunity in chronic inflammatory demyelinating polyradiculoneuropathy. Neurology 2012; 78: 402–408.
Chi LJ, Wang HB, Wang WZ . Impairment of circulating CD4+CD25+ regulatory T cells in patients with chronic inflammatory demyelinating polyradiculoneuropathy. J Peripher Nerv Syst 2008; 13: 54–63.
Yalvac ME, Arnold WD, Hussain SR, Braganza C, Shontz KM, Clark KR et al. VIP-expressing dendritic cells protect against spontaneous autoimmune peripheral polyneuropathy. Mol Ther 2014; 22: 1353–1363.
Sahenk Z, Nagaraja HN, McCracken BS, King WM, Freimer ML, Cedarbaum JM et al. NT-3 promotes nerve regeneration and sensory improvement in CMT1A mouse models and in patients. Neurology 2005; 65: 681–689.
Sahenk Z, Galloway G, Clark KR, Malik V, Rodino-Klapac LR, Kaspar BK et al. AAV1.NT-3 gene therapy for charcot-marie-tooth neuropathy. Mol Ther 2014; 22: 511–521.
Sekimoto M, Tsuji T, Matsuzaki J, Chamoto K, Koda T, Nemoto K et al. Functional expression of the TrkC gene, encoding a high affinity receptor for NT-3, in antigen-specific T helper type 2 (Th2) cells. Immunol Lett 2003; 88: 221–226.
Beutner C, Lepperhof V, Dann A, Linnartz-Gerlach B, Litwak S, Napoli I et al. Engineered stem cell-derived microglia as therapeutic vehicle for experimental autoimmune encephalomyelitis. Gene Therapy 2013; 20: 797–806.
Yang J, Yan Y, Xia Y, Kang T, Li X, Ciric B et al. Neurotrophin 3 transduction augments remyelinating and immunomodulatory capacity of neural stem cells. Mol Ther 2014; 22: 440–450.
Press R, Nennesmo I, Kouwenhoven M, Huang YM, Link H, Pashenkov M . Dendritic cells in the cerebrospinal fluid and peripheral nerves in Guillain-Barre syndrome and chronic inflammatory demyelinating polyradiculoneuropathy. J Neuroimmunol 2005; 159: 165–176.
Barbacid M, Lamballe F, Pulido D, Klein R . The trk family of tyrosine protein kinase receptors. Biochim Biophys Acta 1991; 1072: 115–127.
Huang EJ, Wilkinson GA, Farinas I, Backus C, Zang K, Wong SL et al. Expression of Trk receptors in the developing mouse trigeminal ganglion: in vivo evidence for NT-3 activation of TrkA and TrkB in addition to TrkC. Development 1999; 126: 2191–2203.
Sahenk Z . Neurotrophins and peripheral neuropathies. Brain Pathol 2006; 16: 311–319.
Richner M, Ulrichsen M, Elmegaard SL, Dieu R, Pallesen LT, Vaegter CB . Peripheral nerve injury modulates neurotrophin signaling in the peripheral and central nervous system. Mol Neurobiol 2014; 50: 945–970.
Meier C, Parmantier E, Brennan A, Mirsky R, Jessen KR . Developing Schwann cells acquire the ability to survive without axons by establishing an autocrine circuit involving insulin-like growth factor, neurotrophin-3, and platelet-derived growth factor-BB. J Neurosci 1999; 19: 3847–3859.
Moalem G, Xu K, Yu L . T lymphocytes play a role in neuropathic pain following peripheral nerve injury in rats. Neuroscience 2004; 129: 767–777.
Hendrix S, Peters EM . Neuronal plasticity and neuroregeneration in the skin — the role of inflammation. J Neuroimmunol 2007; 184: 113–126.
Chen Q, Shine HD . Neuroimmune processes associated with Wallerian degeneration support neurotrophin-3-induced axonal sprouting in the injured spinal cord. J Neurosci Res 2013; 91: 1280–1291.
Besser M, Wank R . Cutting edge: clonally restricted production of the neurotrophins brain-derived neurotrophic factor and neurotrophin-3 mRNA by human immune cells and Th1/Th2-polarized expression of their receptors. J Immunol 1999; 162: 6303–6306.
Moalem G, Gdalyahu A, Shani Y, Otten U, Lazarovici P, Cohen IR et al. Production of neurotrophins by activated T cells: implications for neuroprotective autoimmunity. J Autoimmun 2000; 15: 331–345.
Van Doorn PA . Treatment of patients with chronic inflammatory demyelinating polyneuropathy. Rev Neurol 1996; 152: 383–386.
Desurkar A, Lin JP, Mills K, Al-Sarraj S, Jan W, Jungbluth H et al. Charcot-Marie-Tooth (CMT) disease 1A with superimposed inflammatory polyneuropathy in children. Neuropediatrics 2009; 40: 85–88.
Maurer M, Toyka KV, Gold R . Immune mechanisms in acquired demyelinating neuropathies: lessons from animal models. Neuromuscul Disord 2002; 12: 405–414.
Sahenk Z, Galloway G, Edwards C, Malik V, Kaspar BK, Eagle A et al. TrkB and TrkC agonist antibodies improve function, electrophysiologic and pathologic features in Trembler J mice. Exp Neurol 2010; 224: 495–506.
Jing H, Vassiliou E, Ganea D . Prostaglandin E2 inhibits production of the inflammatory chemokines CCL3 and CCL4 in dendritic cells. J Leukoc Biol 2003; 74: 868–879.
Toscano MG, Delgado M, Kong W, Martin F, Skarica M, Ganea D . Dendritic cells transduced with lentiviral vectors expressing VIP differentiate into VIP-secreting tolerogenic-like DCs. Mol Ther 2010; 18: 1035–1045.
Baratelli F, Lin Y, Zhu L, Yang SC, Heuze-Vourc'h N, Zeng G et al. Prostaglandin E2 induces FOXP3 gene expression and T regulatory cell function in human CD4+ T cells. J Immunol 2005; 175: 1483–1490.
Friede RL, Beuche W . Combined scatter diagrams of sheath thickness and fibre calibre in human sural nerves: changes with age and neuropathy. J Neurol Neurosurg Psychiatry 1985; 48: 749–756.
Xia RH, Yosef N, Ubogu EE . Dorsal caudal tail and sciatic motor nerve conduction studies in adult mice: technical aspects and normative data. Muscle Nerve 2010; 41: 850–856.
Arnold WD, Porensky PN, McGovern VL, Iyer CC, Duque S, Li X et al. Electrophysiological biomarkers in spinal muscular atrophy: preclinical proof of concept. Ann Clin Transl Neurol 2014; 1: 34–44.
Acknowledgements
This study was supported by GBS-CIDP Foundation International and Mehmet E Yalvac is a Paul D. Wellstone Muscular Dystrophy Cooperative Research Center Post-doctoral Fellow.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on Gene Therapy website
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Yalvac, M., Arnold, W., Braganza, C. et al. AAV1.NT-3 gene therapy attenuates spontaneous autoimmune peripheral polyneuropathy. Gene Ther 23, 95–102 (2016). https://doi.org/10.1038/gt.2015.67
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2015.67
This article is cited by
-
AAV1.NT-3 gene therapy for X-linked Charcot–Marie–Tooth neuropathy type 1
Gene Therapy (2022)
-
Systemic IGF-1 gene delivery by rAAV9 improves spontaneous autoimmune peripheral polyneuropathy (SAPP)
Scientific Reports (2018)
-
AAV1.NT-3 gene therapy increases muscle fiber diameter through activation of mTOR pathway and metabolic remodeling in a CMT mouse model
Gene Therapy (2018)
-
Hepatocyte Growth Factor (HGF) Promotes Peripheral Nerve Regeneration by Activating Repair Schwann Cells
Scientific Reports (2018)