Abstract
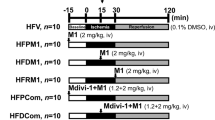
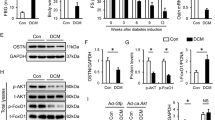
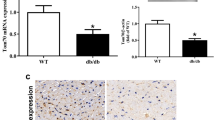
Recent studies suggest that glutaredoxin-1 (Glrx-1) may serve as therapeutic target for diabetic hearts. As the level of reactive oxygen species (ROS) is increased in the pathologic hearts including ischemia/reperfusion (I/R) and diabetes, we assumed that upregulation of Glrx-1 could reduce the cardiac risk factors associated with I/R and/or diabetes. Diabetes was induced in mice by i.p. injection of streptozotocin (150 mg kg−1). Eight days after when the blood glucose was elevated to 400 mg per 100 ml, the animals were randomly assigned to one of the following three groups, which received either empty vector, or LacZ or Glrx-1 adenoviral construct. Four days later, isolated working hearts were subjected to 30 min ischemia followed by 2 h reperfusion. Glrx-1 gene therapy significantly enhanced the Glrx-1 level, which prevented I/R-mediated reduction of ventricular recovery, increased myocardial infarct size and cardiomyocyte apoptosis in diabetic myocardium. In concert, Glrx-1 prevented diabetes and ischemia–reperfusion induced reduction of cardioprotective proteins including Akt, FoxO-1, and hemeoxygenase-1, and abolished the death signal triggered by Jnk, p38 mitogen-activated protein kinase, and c-Src. Glrx-1 gene therapy seems to prevent cardiac complications in diabetic heart due to the I/R by switching the death signal into survival signal by activating Akt-FoxO-signaling network.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Holmgren A . Thioredoxin and glutaredoxin system. J Biol Chem 1989; 264: 13963–13966.
Berndt C, Lillig CH, Holmgren A . Thiol-based mechanisms of the thioredoxin and glutaredoxin systems: implications for diseases in the cardiovascular system. Am J Physiol Heart Circ Physiol 2007; 292: H1227–H1236.
Malik G, Nagy N, Ho Y-S, Maulik N, Das DK . Role of glutaredoxin-1 in cardioprotection: an insight with Glrx-1 transgenic and knockout animals. J Mol Cell Cardiol 2008; 44: 261–269.
Maulik N, Das DK . Emerging potential of thioredoxin and thioredoxin interacting proteins in various disease conditions. Biochim Biophys Acta 2008; 1780: 1368–1382.
Di Simplicio P, de Glorgio LA, Cardaioli E, Lecis R, Micell M, Rossi R et al. Glutathione, glutathione utilizing enzymes and thioltransferase in platelets of insulin-dependent diabetic patients: relation with platelet aggregation and with microangiopatic complications. Eur J Clin Invest 1995; 25: 665–669.
Takagi Y, Nakamura T, Nishiyama A, Nozaki K, Tanaka T, Hashimoto N et al. Localization of glutaredoxin (Thioltransferase) in the rat brain and possible functional implications during focal ischemia. Biochem Biophys Res Commun 1999; 258: 390–394.
Okuda M, Inoue N, Azumi H, Seno T, Sumi Y, Hirata KI et al. Expression of glutaredoxin in human coronary arteries. Its potential role in antioxidant protection against atherosclerosis. Arterioscler Thromb Vasc Biol 2001; 21: 1483–1487.
Li X, Xu Z, Li S, Rozanski GJ . Redox regulation of Ito remodeling in diabetic rat heart. Am J Physiol Heart Circ Physiol 2005; 288: H1417–H1424.
Shelton MD, Kern TS, Mieyal JJ . Glutaredoxin regulates nuclear factor kappa-B and intracellular adhesion molecule in muller cells, model of diabetic retinopathy. J Biol Chem 2007; 282: 12467–12474.
Ivarsson R, Quintens R, Dejonghe S, Tsukamoto K, Veld P, Renstrom E et al. Rodox control of exocytosis regulatory role of NADPH, thioredoxin, and glutaredoxin. Diabetes 2005; 54: 2132–2142.
Lundstrom-Ljung J, Holmgren A . Glutaredoxin accelerates glutathione-dependent folding of reduced ribonuclease A together with protein disulfide-isomerase. J Biol Chem 1995; 270: 7822–7828.
Rozell B, Barcena JA, Martínez-Galisteo E, Padilla CA, Holmgren A . Immunochemical characterization and tissue distribution of glutaredoxin (thioltransferase) from calf. Eur J Cell Biol 1993; 62: 314–323.
Pai HV, Starke DW, Lesnefsky EJ, Hoppel CL, Mieyal JJ . What is the functional significance of the unique location of glutaredoxin 1 (GRx1) in the intermembrane space of mitochondria? Antioxid Redox Signal 2007; 9: 2027–2034.
Maritim AC, Sanders RA, Watkins III JB . Diabetes, oxidative stress, and antioxidants: a review. J Biochem Mol Toxico 2003; 17: 24–38.
Nakamura H, Nakamura K, Yodoi J . Redox regulation of cellular activation. Annu Rev Immunol 1997; 15: 351–369.
Song JJ, Rhee JG, Suntharalingam M, Walsh SA, Spitz DR, Lee YJ . Role of glutaredoxin in metabolic oxidative stress. glutaredoxin as a sensor of oxidative stress mediated by H2O2 . J Biol Chem 2002; 277: 46566–46577.
Song JJ, Lee YJ . Differential role of glutatredoxin and thioredoxin in metabolic oxidative stress-induced activation of apoptosis signal-regulating kinase I. Biochem J 2003; 373: 845–885.
Kim AH, Khursigara G, Sun X, Franke TF, Chao MV . Akt phosphorylates and negatively regulates apoptosis signal-regulating kinase 1. Mol Cell Biol 2001; 21: 893–901.
Satoh M, Matter CM, Ogita H, Takeshita K, Wang CY, Dorn GW et al. Inhibition of apoptosis-regulated signaling kinase-1 and prevention of congestive heart failure by estrogen. Circulation 2007; 115: 3197–3204.
Murata H, Ihara Y, Nakamura H, Yodoi J, Sumikawa K, Kondo T . Glutaredoxin exerts an antiapoptotic effect by regulating the redox state of Akt. J Biol Chem 2003; 278: 50226–50233.
Burgering BM, Kops GJ . Cell cycle and death control: long live forkheads. Trends Biochem Sci 2002; 27: 352–360.
Gurusamy N, Lekli I, Gorbunov N, Gherghiceanu M, Popescu LM, Das DK . Cardioprotection by adaptation to ischemia augments autophagy in association with BAG-1 protein. J Cell Mol Med 2008; 13: 373–387.
Juric D, Wojciechowski P, Das DK, Netticadan T . Prevention of concentric hypertrophy and diastolic impairment in aortic-banded rats treated with resveratrol. Am J Physiol Heart Circ Physiol 2007; 292: H2138–H2143.
Engelman DT, Watanabe M, Engelman RM, Rousou JA, Kisin E, Kagan VE et al. Hypoxic preconditioning preserves antioxidant reserve in the working rat heart. Cardiovasc Res 1995; 29: 133–140.
Mukherjee S, Lekli I, Das M, Azzi A, Das DK . Cardioprotection with alpha-tocopheryl phosphate: amelioration of myocardial ischemia reperfusion injury is linked with its ability to generate a survival signal through Akt activation. Biochim Biophys Acta 2008; 1782: 498–503.
Acknowledgements
This study was supported by National Heart, Lung, and Blood Institute Grants NIH HL-22559, HL-33889, HL-34360, OTKA 72315 and TAMOP-4.2.2-08/1-2008-0007.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Lekli, I., Mukherjee, S., Ray, D. et al. Functional recovery of diabetic mouse hearts by glutaredoxin-1 gene therapy: role of Akt-FoxO-signaling network. Gene Ther 17, 478–485 (2010). https://doi.org/10.1038/gt.2010.9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2010.9
Keywords
This article is cited by
-
Glutaredoxin 1 protects neurons from oxygen‐glucose deprivation/reoxygenation (OGD/R)-induced apoptosis and oxidative stress via the modulation of GSK-3β/Nrf2 signaling
Journal of Bioenergetics and Biomembranes (2021)
-
Acute fluoride exposure alters myocardial redox and inflammatory markers in rats
Molecular Biology Reports (2019)
-
Forkhead box transcription factor 1: role in the pathogenesis of diabetic cardiomyopathy
Cardiovascular Diabetology (2016)