Abstract
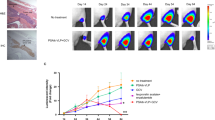
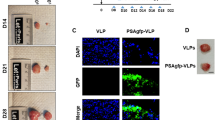
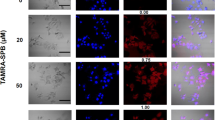
The sodium iodide symporter (NIS) directs the uptake and concentration of iodide in thyroid cells. We have extended the use of NIS-mediated radioiodine therapy to other types of cancer, we transferred and expressed the NIS gene into prostate, colon and breast cancer cells using adenoviral vectors. To improve vector efficiency we have developed a conditionally replicating adenovirus (CRAd) in which the E1a gene is driven by the prostate-specific promoter, Probasin and the cassette RSV promoter human NIScDNA-bGH polyA replaces the E3 region (CRAd Ad5PB_RSV-NIS). In vitro infection of the prostate cancer cell line LnCaP resulted in virus replication, cytolysis and release of infective viral particles. Conversely, the prostate cancer cell line PC-3 (androgen receptor negative) and the pancreatic cancer cell line Panc-1 were refractory to the viral cytopathic effect and did not support viral replication. Radioiodine uptake was readily measurable in LnCaP cells infected with Ad5PB_RSV-NIS 24 h post-infection, confirming NIS expression. In vivo, LnCaP tumor xenografts in nude-mice injected intratumorally with Ad5PB_RSV_NIS CRAd expressed NIS actively as evidenced by 99Tc uptake and imaging. Administration of therapeutic 131I after virus injection significantly increased survival probability in mice carrying xenografted LnCaP tumors compared with virotherapy alone. These data indicate that Ad5PB_RSV_NIS replication is stringently restricted to androgen-positive prostate cancer cells and results in effective NIS expression and uptake of radioiodine. This construct may allow multimodal therapy, combining cytolytic virotherapy with radioiodine treatment, to be developed as a novel treatment for prostate cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Ruijter E, van de Kaa C, Miller G, Ruiter D, Debruyne F, Schalken J et al. Molecular genetics and epidemiology of prostate carcinoma. Endocr Rev 1999; 20: 22–45.
Chiocca EA, Broaddus WC, Gillies GT, Visted T, Lamfers ML . Neurosurgical delivery of chemotherapeutics, targeted toxins, genetic and viral therapies in neuro-oncology. J Neuro-Oncol 2004; 69: 101–117.
McCormick F . Cancer gene therapy: fringe or cutting edge? Nat Rev Cancer 2001; 1: 130–141.
Yamamoto M, Curiel DT . Cancer gene therapy. Technol Cancer Res Treat 2005; 4: 315–330.
Carrasco N . Iodide transport in the thyroid gland. Biochimica et Biophysica Acta 1993; 1154: 65–82.
Jhiang SM, Cho JY, Ryu KY, DeYoung BR, Smanik PA, McGaughy VR et al. An immunohistochemical study of Na+/I- symporter in human thyroid tissues and salivary gland tissues. Endocrinology 1998; 139: 4416–4419.
Van Nostrand D, Wartofsky L . Radioiodine in the treatment of thyroid cancer. Endocrinol Metabol Clinics North Am 2007; 36: 807–822.
ELM. Carcinoma of follicular epithelium: Radioiodine and other treatments and outcomes. In: Braverman LE, Utiger RD (eds). The Thyroid: A Fundamental and Clinical Text, 7th ed. Lippincott: Raven, Philadelphia, 1996: 922–945.
Scholz IV, Cengic N, Baker CH, Harrington KJ, Maletz K, Bergert ER et al. Radioiodine therapy of colon cancer following tissue-specific sodium iodide symporter gene transfer. Gene Therapy 2005; 12: 272–280.
Dwyer RM, Bergert ER, O’Connor MK, Gendler SJ, Morris JC . In vivo radioiodide imaging and treatment of breast cancer xenografts after MUC1-driven expression of the sodium iodide symporter. Clin Cancer Res 2005; 11: 1483–1489.
Dwyer RM, Bergert E R, O’Connor MK, Gendler SJ, Morris JC . Sodium Iodide symporter-mediated radioiodide imaging and therapy of ovarian tumor xenografts in mice. Gene Therapy 2006; 13: 60–66.
Spitzweg C, Dietz AB, O’Connor MK, Bergert ER, Tindall DJ, Young CY et al. In vivo sodium iodide symporter gene therapy of prostate cancer. Gene Therapy 2001; 8: 1524–1531.
Dwyer RM, Schatz SM, Bergert ER, Myers RM, Harvey ME, Classic KL et al. A preclinical large animal model of adenovirus-mediated expression of the sodium-iodide symporter for radioiodide imaging and therapy of locally recurrent prostate cancer. Mol Ther: J Am Soc Gene Ther 2005; 12: 835–841.
Morris JC . 〈http://clinicaltrials.gov/ct/show/NCT00788307〉 2009.
Herrmann F . Cancer gene therapy: principles, problems, and perspectives. J Mol Med 1995; 73: 157–163.
Waehler R, Russell SJ, Curiel DT . Engineering targeted viral vectors for gene therapy. Nat Rev Genet 2007; 8: 573–587.
Bischoff JR, Kirn DH, Williams A, Heise C, Horn S, Muna M et al. An adenovirus mutant that replicates selectively in p53-deficient human tumor cells. Science 1996; 274: 373–376.
Markert JM, Malick A, Coen DM, Martuza RL . Reduction and elimination of encephalitis in an experimental glioma therapy model with attenuated herpes simplex mutants that retain susceptibility to acyclovir. Neurosurgery 1993; 32: 597–603.
Russell SJ . RNA viruses as virotherapy agents. Cancer Gene Ther 2002; 9: 961–966.
Bazan-Peregrino M, Carlisle RC, Hernandez-Alcoceba R, Iggo R, Homicsko K, Fisher KD et al. Comparison of molecular strategies for breast cancer virotherapy using oncolytic adenovirus. Hum Gene Ther 2008; 19: 873–886.
Chu RL, Post DE, Khuri FR, Van Meir EG . Use of replicating oncolytic adenoviruses in combination therapy for cancer. Clin Cancer Res 2004; 10: 5299–5312.
Dingli D, Peng KW, Harvey ME, Greipp PR, O’Connor MK, Cattaneo R et al. Image-guided radiovirotherapy for multiple myeloma using a recombinant measles virus expressing the thyroidal sodium iodide symporter. Blood 2004; 103: 1641–1646.
Zhang J, Thomas TZ, Kasper S, Matusik RJ . A small composite probasin promoter confers high levels of prostate-specific gene expression through regulation by androgens and glucocorticoids in vitro and in vivo. Endocrinology 2000; 141: 4698–4710.
Zuckier LS, Dohan O, Li Y, Chang CJ, Carrasco N, Dadachova E . Kinetics of perrhenate uptake and comparative biodistribution of perrhenate, pertechnetate, and iodide by NaI symporter-expressing tissues in vivo. J Nuclear Med 2004; 45: 500–507.
Kakinuma H, Bergert ER, Spitzweg C, Cheville JC, Lieber MM, Morris JC . Probasin promoter (ARR(2)PB)-driven, prostate-specific expression of the human sodium iodide symporter (h-NIS) for targeted radioiodine therapy of prostate cancer. Cancer Res 2003; 63: 7840–7844.
Cascallo M, Capella G, Mazo A, Alemany R . Ras-dependent oncolysis with an adenovirus VAI mutant. Cancer Res 2003; 63: 5544–5550.
Humphreys MJ, Greenhalf W, Neoptolemos JP, Ghaneh P . The potential for gene therapy in pancreatic cancer. Int J Pancreatol 1999; 26: 5–21.
Hung MC, Hortobagyi GN, Ueno NT . Development of clinical trial of E1A gene therapy targeting HER-2/neu-overexpressing breast and ovarian cancer. Adv Exp Med Biol 2000; 465: 171–180.
Miura Y, Ohnami S, Yoshida K, Ohashi M, Nakano M, Fukuhara M et al. Intraperitoneal injection of adenovirus expressing antisense K-ras RNA suppresses peritoneal dissemination of hamster syngeneic pancreatic cancer without systemic toxicity. Cancer Lett 2005; 218: 53–62.
Young LS, Searle PF, Onion D, Mautner V . Viral gene therapy strategies: from basic science to clinical application. J Pathol 2006; 208: 299–318.
Schmidt-Wolf G, Schmidt-Wolf IG . Human cancer and gene therapy. Ann Hematol 1994; 69: 273–279.
Kaminsky SM, Levy O, Salvador C, Dai G, Carrasco N . The Na+/I- symporter of the thyroid gland. Soc Gen Physiol Ser 1993; 48: 251–262.
Hortobagyi GN, Ueno NT, Xia W, Zhang S, Wolf JK, Putnam JB et al. Cationic liposome-mediated E1A gene transfer to human breast and ovarian cancer cells and its biologic effects: a phase I clinical trial. J Clin Oncol 2001; 19: 3422–3433.
Ricciardelli C, Jackson MW, Choong CS, Stahl J, Marshall VR, Horsfall DJ et al. Elevated levels of HER-2/neu and androgen receptor in clinically localized prostate cancer identifies metastatic potential. Prostate 2008; 68: 830–838.
Qu CF, Li Y, Song YJ, Rizvi SM, Raja C, Zhang D et al. MUC1 expression in primary and metastatic pancreatic cancer cells for in vitro treatment by (213)Bi-C595radioimmunoconjugate. Br J Cancer 2004; 91: 2086–2093.
Anderson RD, Haskell R E, Xia H, Roessler BJ, Davidson BL . A simple method for the rapid generation of recombinant adenovirus vectors. Gene Therapy 2000; 7: 1034–1038.
Weiss SJ, Philp NJ, Grollman EF . Iodide transport in a continuous line of cultured cells from rat thyroid. Endocrinology 1984; 114: 1090–1098.
Spitzweg C, Dietz AB, O’Connor MK, Bergert ER, Tindall DJ, Young CY et al. In vivo sodium iodide symporter gene therapy of prostate cancer. Gene Therapy 2001; 8: 1524–1531.
Acknowledgements
We thank Azhal Ahmadi, Robert Parrish, Rachel E Carlson, Tracy Decklever and Dr Jian Qiao for technical help. This work was supported by Prostate SPORE Grant P50 CA 091956, Donald J Tindall, PI, John C Morris, Project Director.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Trujillo, M., Oneal, M., McDonough, S. et al. A probasin promoter, conditionally replicating adenovirus that expresses the sodium iodide symporter (NIS) for radiovirotherapy of prostate cancer. Gene Ther 17, 1325–1332 (2010). https://doi.org/10.1038/gt.2010.63
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2010.63
Keywords
This article is cited by
-
Preferable sites and orientations of transgene inserted in the adenovirus vector genome: The E3 site may be unfavorable for transgene position
Gene Therapy (2015)
-
Molecular imaging of oncolytic viral therapy
Molecular Therapy - Oncolytics (2014)
-
Viral dose, radioiodide uptake, and delayed efflux in adenovirus-mediated NIS radiovirotherapy correlates with treatment efficacy
Gene Therapy (2013)
-
Sodium iodide symporter (NIS)-mediated radiovirotherapy of hepatocellular cancer using a conditionally replicating adenovirus
Gene Therapy (2013)
-
Effect of increased viral replication and infectivity enhancement on radioiodide uptake and oncolytic activity of adenovirus vectors expressing the sodium iodide symporter
Cancer Gene Therapy (2013)