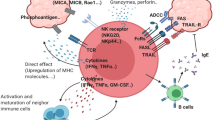
Abstract
Adoptive T-cell transfer of in vitro cultured T cells derived from cancer patients with naturally developed immune responses has met with some success as an immunotherapeutic approach, although only a limited number of patients showed spontaneous immune responses. To find alternative ways, such as cancer-specific T-cell receptor (TCR) gene transfer, in preparation for sufficient numbers of antigen-specific T cells is an important issue in the field of adoptive T-cell therapy. Given the inherent disadvantage of αβ TCR transfer to other αβ T cells, namely the possible formation of mixed TCR heterodimers with endogenous α or β TCR, we employed γδ T cells as a target for retroviral transfer of cancer-specific TCR and examined whether γδ T cells were useful as an alternative population for TCR transfer. Although retroviral transduction to γδ T cells with TCR αβ genes alone, isolated from a MAGE-A4143–151-specific αβ CD8+ cytotoxic T lymphocyte (CTL) clone, did not provide sufficient affinity to recognize major histocompatibility (MHC)–peptide complexes due to the lack of CD8 co-receptor, γδ T cells co-transduced with TCR αβ and CD8 αβ genes acquired cytotoxicity against tumor cells and produced cytokines in both αβ- and γδ-TCR-dependent manners. Furthermore, αβ TCR and CD8-transduced γδ T cells, stimulated either through αβ TCR or γδ TCR, rapidly responded to target cells compared with conventional αβ T cells, reminiscent of γδ T cells. We propose αβ TCR-transduced γδ T cells as an alternative strategy for adoptive T-cell transfer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Abbreviations
- 2M3B1PP:
-
2-Methyl-3-Butenyl-1-Pyrophosphate
- CTL:
-
Cytotoxic T lymphocytes
- IFN:
-
Interferon
- IL:
-
Interleukin
- TAP:
-
Transporter associated with antigen processing
- TCR:
-
T-cell receptor
References
Rosenberg SA, Yang JC, Restifo NP . Cancer immunotherapy: moving beyond current vaccines. Nat Med 2004; 10: 909–915.
Gattinoni L, Powell DJ Jr, Rosenberg SA, Restifo NP . Adoptive immunotherapy for cancer: building on success. Nat Rev Immunol 2006; 6: 383–393.
Morgan RA, Dudley ME, Wunderlich JR, Hughes MS, Yang JC, Sherry RM et al. Cancer regression in patients after transfer of genetically engineered lymphocytes. Science 2006; 314: 126–129.
van der Veken LT, Hagedoorn RS, van Loenen MM, Willemze R, Falkenburg JH, Heemskerk MH . αβ T-cell receptor engineered γδT cells mediate effective antileukemic reactivity. Cancer Res 2006; 66: 3331–3337.
Kato Y, Tanaka Y, Miyagawa F, Yamashita S, Minato N . Targeting of tumor cells for human γδ T cells by nonpeptide antigens. J Immunol 2001; 167: 5092–5098.
Tanaka Y . Human γδ T cells and tumor immunotherapy. J Clin Exp Hematop 2006; 46: 11–23.
Tough DF, Sprent J . Lifespan of γδ T cells. J Exp Med 1998; 187: 357–365.
Gao Y, Yang W, Pan M, Scully E, Girardi M, Augenlicht LH et al. γδ T cells provide an early source of interferon γ in tumor immunity. J Exp Med 2003; 198: 433–442.
Lafont V, Loisel S, Liautard J, Dudal S, Sable-Teychene M, Liautard JP et al. Specific signaling pathways triggered by IL-2 in human Vγ9Vδ2 T cells: an amalgamation of NK and αβ T cell signaling. J Immunol 2003; 171: 5225–5232.
Correa I, Bix M, Liao NS, Zijlstra M, Jaenisch R, Raulet D . Most γδ T cells develop normally in β2-microglobulin-deficient mice. Proc Natl Acad Sci USA 1992; 89: 653–657.
Bigby M, Markowitz JS, Bleicher PA, Grusby MJ, Simha S, Siebrecht M et al. Most γδ T cells develop normally in the absence of MHC class II molecules. J Immunol 1993; 151: 4465–4475.
Kunzmann V, Bauer E, Feurle J, Weissinger F, Tony HP, Wilhelm M . Stimulation of γδ T cells by aminobisphosphonates and induction of antiplasma cell activity in multiple myeloma. Blood 2000; 96: 384–392.
Kato Y, Tanaka Y, Tanaka H, Yamashita S, Minato N . Requirement of species-specific interactions for the activation of human γδ T cells by pamidronate. J Immunol 2003; 170: 3608–3613.
Heemskerk MH, Hagedoorn RS, van der Hoorn MA, van der Veken LT, Hoogeboom M, Kester MG et al. Efficiency of T-cell receptor expression in dual-specific T cells is controlled by the intrinsic qualities of the TCR chains within the TCR-CD3 complex. Blood 2007; 109: 235–243.
De Plaen E, De Backer O, Arnaud D, Bonjean B, Chomez P, Martelange V et al. A new family of mouse genes homologous to the human MAGE genes. Genomics 1999; 55: 176–184.
Scanlan MJ, Simpson AJ, Old LJ . The cancer/testis genes: review, standardization, and commentary. Cancer Immun 2004; 4: 1.
Miyahara Y, Naota H, Wang L, Hiasa A, Goto M, Watanabe M et al. Determination of cellularly processed HLA-A2402-restricted novel CTL epitopes derived from two cancer germ line genes, MAGE-A4 and SAGE. Clin Cancer Res 2005; 11: 5581–5589.
Hiasa A, Hirayama M, Nishikawa H, Kitano S, Nukaya I, Yu SS et al. Long-term phenotypic, functional and genetic stability of cancer-specific T-cell receptor (TCR) αβ genes transduced to CD8+ T cells. Gene Therapy 2008; 15: 695–699.
Tanaka Y, Morita CT, Nieves E, Brenner MB, Bloom BR . Natural and synthetic non-peptide antigens recognized by human γδ T cells. Nature 1995; 375: 155–158.
Nishikawa H, Qian F, Tsuji T, Ritter G, Old LJ, Gnjatic S et al. Influence of CD4+CD25+ regulatory T cells on low/high-avidity CD4+ T cells following peptide vaccination. J Immunol 2006; 176: 6340–6346.
Le Gal FA, Ayyoub M, Dutoit V, Widmer V, Jager E, Cerottini JC et al. Distinct structural TCR repertoires in naturally occurring versus vaccine-induced CD8+ T-cell responses to the tumor-specific antigen NY-ESO-1. J Immunother 2005; 28: 252–257.
Demotte N, Stroobant V, Courtoy PJ, Van Der Smissen P, Colau D, Luescher IF et al. Restoring the association of the T cell receptor with CD8 reverses anergy in human tumor-infiltrating lymphocytes. Immunity 2008; 28: 414–424.
Battistini L, Caccamo N, Borsellino G, Meraviglia S, Angelini DF, Dieli F et al. Homing and memory patterns of human γδ T cells in physiopathological situations. Microbes Infect 2005; 7: 510–517.
Moser B, Eberl M . T cells: novel initiators of adaptive immunity. Immunol Rev 2007; 215: 89–102.
Glatzel A, Wesch D, Schiemann F, Brandt E, Janssen O, Kabelitz D . Patterns of chemokine receptor expression on peripheral blood γδ T lymphocytes: strong expression of CCR5 is a selective feature of Vδ2Vγ9 γδT cells. J Immunol 2002; 168: 4920–4929.
Soria G, Yaal-Hahoshen N, Azenshtein E, Shina S, Leider-Trejo L, Ryvo L et al. Concomitant expression of the chemokines RANTES and MCP-1 in human breast cancer: a basis for tumor-promoting interactions. Cytokine 2008; 44: 191–200.
Sugasawa H, Ichikura T, Tsujimoto H, Kinoshita M, Morita D, Ono S et al. Prognostic significance of expression of CCL5/RANTES receptors in patients with gastric cancer. J Surg Oncol 2008; 97: 445–450.
Gong G, Shao L, Wang Y, Chen CY, Huang D, Yao S et al. Phosphoantigen-activated Vγ2Vδ2 T cells antagonize IL-2-induced CD4+CD25+Foxp3+ T regulatory cells. Blood 2009; 113: 837–845.
Ikeda H, Old LJ, Schreiber RD . The roles of IFNγ in protection against tumor development and cancer immunoediting. Cytokine Growth Factor Rev 2002; 13: 95–109.
Nishikawa H, Kato T, Tawara I, Ikeda H, Kuribayashi K, Allen PM et al. IFN-γ controls the generation/activation of CD4+ CD25+ regulatory T cells in antitumor immune response. J Immunol 2005; 175: 4433–4440.
Nagata Y, Furugen R, Hiasa A, Ikeda H, Ohta N, Furukawa K et al. Peptides derived from a wild-type murine proto-oncogene c-erbB-2/HER2/neu can induce CTL and tumor suppression in syngeneic hosts. J Immunol 1997; 159: 1336–1343.
Ikuta Y, Okugawa T, Furugen R, Nagata Y, Takahashi Y, Wang L et al. A HER2/NEU-derived peptide, a Kd-restricted murine tumor rejection antigen, induces HER2-specific HLA-A2402-restricted CD8+ cytotoxic T lymphocytes. Int J Cancer 2000; 87: 553–558.
Kim S, Lee K, Kim MD, Kang S, Joo CW, Kim JM et al. Factors affecting the performance of different long terminal repeats in the retroviral vector. Biochem Biophys Res Commun 2006; 343: 1017–1022.
Nishikawa H, Sato E, Briones G, Chen LM, Matsuo M, Nagata Y et al. In vivo antigen delivery by a Salmonella typhimurium type III secretion system for therapeutic cancer vaccines. J Clin Invest 2006; 116: 1946–1954.
Nishikawa H, Jager E, Ritter G, Old LJ, Gnjatic S . CD4+CD25+ regulatory T cells control the induction of antigen-specific CD4+ helper T cell responses in cancer patients. Blood 2005; 106: 1008–1011.
Nishikawa H, Tsuji T, Jager E, Briones G, Ritter G, Old LJ et al. Induction of regulatory T cell-resistant helper CD4+ T cells by bacterial vector. Blood 2008; 111: 1404–1412.
Acknowledgements
We thank C Hyuga, S Hori and Y Shirakura for their excellent technical support. This study was supported by a Grant-in-Aid for Scientific Research on Priority Areas from the Ministry of Education, Culture, Sports, Science and Technology of Japan. AH, HN, JM, IK and HS designed the research and analyzed the data. AH, HN, JM, YT and HS wrote the paper. YT and NM contributed vital new reagents. AH, MH, SK, SO, HC and SSY performed the research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hiasa, A., Nishikawa, H., Hirayama, M. et al. Rapid αβ TCR-mediated responses in γδ T cells transduced with cancer-specific TCR genes. Gene Ther 16, 620–628 (2009). https://doi.org/10.1038/gt.2009.6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2009.6
Keywords
This article is cited by
-
The promise of γδ T cells and the γδ T cell receptor for cancer immunotherapy
Cellular & Molecular Immunology (2015)
-
Potential of human γδ T cells for immunotherapy of osteosarcoma
Molecular Biology Reports (2013)
-
A Promising Vector for TCR Gene Therapy: Differential Effect of siRNA, 2A Peptide, and Disulfide Bond on the Introduced TCR Expression
Molecular Therapy - Nucleic Acids (2012)
-
Single-chain VαVβ T-cell receptors function without mispairing with endogenous TCR chains
Gene Therapy (2012)
-
Emerging concepts in biomarker discovery; The US-Japan workshop on immunological molecular markers in oncology
Journal of Translational Medicine (2009)