Abstract
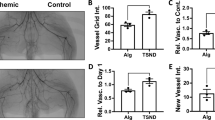
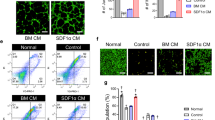
Vascular endothelial growth factor (VEGF) gene transfer-mediated angiogenesis has been proposed for peripheral artery disease. However, protocols using single administration have shown little benefit. Given that the transient nature of VEGF gene expression provokes instability of neovasculature, we hypothesized that repeated administration would provide efficient tissue protection. We thus compared single vs repeated transfection in a rabbit model of hindlimb ischemia by injecting a plasmid encoding human VEGF165 (pVEGF165) at 7 (GI, n=10) or 7 and 21 (GII, n=10) days after surgery. Placebo animals (GIII, n=10) received empty plasmid. Fifty days after surgery, single and repeated administration similarly increased saphenous peak flow velocity and quantity of angiographically visible collaterals. However, microvasculature increased only with repeated transfection: capillary density was 49.4±15.4 capillaries per 100 myocytes in GI, 84.6±14.7 in GII (P<0.01 vs GI and GIII) and 49.3±13.6 in GIII, and arteriolar density was 1.9±0.6 arterioles per mm2 in GI, 3.0±0.9 in GII (P<0.01 vs GI and GIII) and 1.5±0.6 in GIII. Muscle lesions were reduced only within repeated transfection. With single administration, gene expression peaked at 7 days and declined rapidly, but with repeated administration, it remained positive at 50 days. At 90 days of repeated transfection (additional animals), gene expression decreased significantly, but neovessel densities did not. Thus, repeated, but not single, VEGF gene transfection resulted in increased microvasculature, which, in turn, afforded effective protection against ischemic muscle damage.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Rothwell P, Coull A, Silver L, Fairhead J, Giles M, Lovelock C et al. Population-based study of event-rate, incidence, case fatality, and mortality for all acute vascular events in all arterial territories (Oxford Vascular Study). Lancet 2005; 366: 1773–1783.
Mahmud E, Cavendish J, Salami A . Current treatment of peripheral arterial disease: role of percutaneous interventional therapies. J Am Coll Cardiol 2007; 50: 473–490.
Melillo G, Serino F, Cirielli C, Capogrossi M . Gene therapy in peripheral artery disease. Curr Drug Targets Cardiovasc Haematol Disord 2004; 4: 295–300.
Isner J, Pieczek A, Schainfeld R, Blair R, Haley L, Asahara T et al. Clinical evidence of angiogenesis after arterial transfer of phVEGF165 in patient with ischaemic limb. Lancet 1996; 348: 370–374.
Baumgartner I, Pieczek A, Manor O, Blair R, Kearney M, Walsh K et al. Constitutive expression of VEGF165 after intramuscular gene transfer promotes collateral vessel development in patients with critical limb ischemia. Circulation 1998; 97: 1114–1123.
Isner J, Baumgartner I, Rauh G, Schainfeld R, Blair R, Manor O et al. Treatment of thromboangiitis obliterans (Buerger's disease) by intramuscular gene transfer of vascular endothelial growth factor: preliminary clinical results. J Vasc Surg 1998; 28: 964–975.
Simovic D, Isner JM, Ropper AH, Pieczek A, Weinberg DH . Improvement in chronic ischemic neuropathy after intramuscular phVEGF165 gene transfer in patients with critical limb ischemia. Arch Neurol 2001; 58: 761–768.
Kim H, Jang S, Park J, Byun J, Kim D, Do Y et al. Vascular endothelial growth factor-induced angiogenic gene therapy in patients with peripheral artery disease. Exp Mol Med 2004; 36: 336–344.
Morishita R, Aoki M, Hashiya N, Makino H, Yamasaki K, Azuma J et al. Safety evaluation of clinical gene therapy using hepatocyte growth factor to treat peripheral arterial disease. Hypertension 2004; 44: 203–209.
Mäkinen K, Manninen H, Hedman M, Matsi P, Mussalo H, Alhava E et al. Increased vascularity detected by digital subtraction angiography after VEGF gene transfer to human lower limb artery: a randomized, placebo-controlled, double-blinded phase II study. Mol Ther 2002; 6: 127–133.
Kusumanto Y, van Weel V, Mulder N, Smit A, van den Dungen J, Hooymans J et al. Treatment with intramuscular vascular endothelial growth factor gene compared with placebo for patients with diabetes mellitus and critical limb ischemia: a double-blind randomized trial. Hum Gene Ther 2006; 17: 683–691.
Nikol S, Baumgartner I, Van Belle E, Diehm C, Visoná A, Capogrossi MC et al. Therapeutic angiogenesis with intramuscular NV1FGF improves amputation-free survival in patients with critical limb ischemia. Mol Ther 2008; 16: 972–978.
Rajagopalan S, Mohler III E, Lederman R, Mendelsohn F, Saucedo J, Goldman C et al. Regional angiogenesis with vascular endothelial growth factor in peripheral arterial disease: a phase II randomized, double-blind, controlled study of adenoviral delivery of vascular endothelial growth factor 121 in patients with disabling intermittent claudication. Circulation 2003; 108: 1933–1938.
Crottogini A, Cabeza Meckert P, Vera Janavel G, Lascano E, Negroni J, Del Valle H et al. Arteriogenesis induced by intramyocardial vascular endothelial growth factor 165 gene transfer in chronically ischemic pigs. Hum Gene Ther 2003; 14: 1307–1318.
Laguens R, Cabeza Meckert P, Vera Janavel G, De Lorenzi A, Lascano E, Negroni J et al. Cardiomyocyte hyperplasia after plasmid-mediated vascular endothelial growth factor gene transfer in pigs with chronic myocardial ischemia. J Gene Med 2004; 6: 222–227.
Vera Janavel G, Crottogini A, Cabeza Meckert P, Cuniberti L, Mele A, Papouchado M et al. Plasmid-mediated VEGF gene transfer induces cardiomyogenesis and reduces myocardial infarct size in sheep. Gene Therapy 2006; 13: 1133–1142.
Gounis M, Spiga M, Graham R, Wilson A, Haliko S, Lieber B et al. Angiogenesis is confined to the transient period of VEGF expression that follows adenoviral gene delivery to ischemic muscle. Gene Therapy 2005; 12: 762–771.
Tang K, Breen E, Gerber H, Ferrara N, Wagner P . Capillary regression in vascular endothelial growth factor-deficient skeletal muscle. Physiol Genomics 2004; 18: 63–69.
Germani A, Di Carlo A, Mangoni A, Straino S, Giacinti C, Turrini P et al. Vascular endothelial growth factor modulates skeletal myoblast function. Am J Pathol 2003; 163: 1417–1428.
Dor Y, Djonov V, Abramovitch R, Itin A, Fishman GI, Carmeliet P et al. Conditional switching of VEGF provides new insights into adult neovascularization and pro-angiogenic therapy. EMBO J 2002; 21: 1939–1947.
Leong-Poi H, Kuliszewski MA, Lekas M, Sibbald M, Teichert-Kuliszewska K, Klibanov AL et al. Therapeutic arteriogenesis by ultrasound-mediated VEGF165 plasmid gene delivery to chronically ischemic skeletal muscle. Circ Res 2007; 101: 295–303.
Muhs A, Lenter M, Seidler R, Zweigerdt R, Kirchengast M, Weser R et al. Nonviral monocyte chemoattractant protein-1 gene transfer improves arteriogenesis after femoral artery occlusion. Gene Therapy 2004; 11: 1685–1693.
Kondoh K, Koyama H, Miyata T, Takato T, Hamada H, Shigematsu H . Conduction performance of collateral vessels induced by vascular endothelial growth factor or basic fibroblast growth factor. Cardiovasc Res 2004; 61: 132–142.
Babiak A, Schumm AM, Wangler C, Loukas M, Wu J, Dombrowski S et al. Coordinated activation of VEGFR-1 and VEGFR-2 is a potent arteriogenic stimulus leading to enhancement of regional perfusion. Cardiovasc Res 2004; 61: 789–795.
Tokunaga N, Nagaya N, Shirai M, Tanaka E, Ishibashi-Ueda H, Harada-Shiba M et al. Adrenomedullin gene transfer induces therapeutic angiogenesis in a rabbit model of chronic hind limb ischemia: benefits of a novel nonviral vector, gelatin. Circulation 2004; 109: 526–531.
Patel T, Kimura H, Weiss C, Semenza G, Hofmann L . Constitutively active HIF-1alpha improves perfusion and arterial remodeling in an endovascular model of limb ischemia. Cardiovasc Res 2005; 68: 144–154.
Yamada N, Li W, Ihaya A, Kimura T, Morioka K, Uesaka T et al. Platelet-derived endothelial cell growth factor gene therapy for limb ischemia. J Vasc Surg 2006; 44: 1322–1328.
Gowdak L, Poliakova L, Wang X, Kovesdi I, Fishbein K, Zacheo A et al. Adenovirus-mediated VEGF(121) gene transfer stimulates angiogenesis in normoperfused skeletal muscle and preserves tissue perfusion after induction of ischemia. Circulation 2000; 102: 565–571.
Pelisek J, Fuchs A, Engelmann M, Shimizu M, Golda A, Mekkaoui C et al. Vascular endothelial growth factor response in porcine coronary and peripheral arteries using nonsurgical occlusion model, local delivery, and liposome-mediated gene transfer. Endothelium 2003; 10: 247–255.
Shyu K, Chang H, Wang B, Kuan P . Intramuscular vascular endothelial growth factor gene therapy in patients with chronic critical leg ischemia. Am J Med 2003; 114: 85–92.
Ylä-Herttuala S, Rissanen TT, Vajanto I, Hartikainen J . Vascular endothelial growth factors: biology and current status of clinical applications in cardiovascular medicine. J Am Coll Cardiol 2007; 49: 1015–1026.
Rissanen TT, Korpisalo P, Markkanen JE, Liimatainen T, Ordén MR, Kholová I et al. Blood flow remodels growing vasculature during vascular endothelial growth factor gene therapy and determines between capillary arterialization and sprouting angiogenesis. Circulation 2005; 112: 3937–3946.
Takeshita S, Zheng L, Brogi E, Kearney M, Pu L, Bunting S et al. Therapeutic angiogenesis. A single intraarterial bolus of vascular endothelial growth factor augments revascularization in a rabbit ischemic hind limb model. J Clin Invest 1994; 93: 662–670.
Laguens R, Cabeza Meckert P, Vera Janavel G, Del Valle H, Lascano E, Negroni J et al. Entrance in mitosis of adult cardiomyocytes in ischemic pig hearts after plasmid-mediated rhVEGF165 gene transfer. Gene Therapy 2002; 9: 1676–1681.
Acknowledgements
We are grateful to medical student Anna Hnatiuk and technicians, Julio Martínez, Fabián Gauna and Luis Palacios, for surgical and morphometric analysis assistance. We also thank veterinarians, María Inés Besansón, Pedro Iguain and Marta Tealdo, for anesthetic management, and animal house assistants, Juan Ocampo, Osvaldo Sosa and Juan Carlos Mansilla, for animal care. This work was supported by Grant BID 1728/OC-AR PID 268 from the Agencia Nacional de Promoción Científica y Tecnológica (ANPCyT) of Argentina. AC, RL and LC are established investigators of Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), Argentina. AB, MC and CM are scientists of Bio Sidus, the biotechnological company developing the plasmid used in this report.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Olea, F., Janavel, G., Cuniberti, L. et al. Repeated, but not single, VEGF gene transfer affords protection against ischemic muscle lesions in rabbits with hindlimb ischemia. Gene Ther 16, 716–723 (2009). https://doi.org/10.1038/gt.2009.30
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2009.30
Keywords
This article is cited by
-
High-dose intramyocardial HMGB1 induces long-term cardioprotection in sheep with myocardial infarction
Drug Delivery and Translational Research (2019)
-
Diabetes-associated macrovascular complications: cell-based therapy a new tool?
Endocrine (2013)
-
Long-term effects of hepatocyte growth factor gene therapy in rat myocardial infarct model
Gene Therapy (2012)
-
Osteogenic role of endosomal chloride channels in MC3T3-E1 cells
Molecular and Cellular Biochemistry (2010)