Abstract
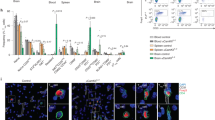
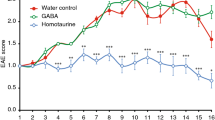
Central nervous system (CNS) delivery of anti-inflammatory cytokines, such as interleukin 4 (IL4), holds promise as treatment for multiple sclerosis (MS). We have previously shown that short-term herpes simplex virus type 1-mediated IL4 gene therapy is able to inhibit experimental autoimmune encephalomyelitis (EAE), an animal model of MS, in mice and non-human primates. Here, we show that a single administration of an IL4-expressing helper-dependent adenoviral vector (HD-Ad) into the cerebrospinal fluid (CSF) circulation of immunocompetent mice allows persistent transduction of neuroepithelial cells and long-term (up to 5 months) CNS transgene expression without toxicity. Mice affected by chronic and relapsing EAE display clinical and neurophysiological recovery from the disease once injected with the IL4-expressing HD-Ad vector. The therapeutic effect is due to the ability of IL4 to increase, in inflamed CNS areas, chemokines (CCL1, CCL17 and CCL22) capable of recruiting regulatory T cells (CD4+CD69−CD25+Foxp3+) with suppressant functions. CSF delivery of HD-Ad vectors expressing anti-inflammatory molecules might represent a valuable therapeutic option for CNS inflammatory disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Pardridge WM . Drug and gene delivery to the brain: the vascular route. Neuron 2002; 36: 555–558.
Pardridge WM . Drug and gene targeting to the brain with molecular Trojan horses. Nat Rev Drug Discov 2002; 1: 131–139.
Consiglio A, Quattrini A, Martino S, Bensadoun JC, Dolcetta D, Trojani A et al. In vivo gene therapy of metachromatic leukodystrophy by lentiviral vectors: correction of neuropathology and protection against learning impairments in affected mice. Nat Med 2001; 7: 310–316.
Davidson BL, Breakefield XO . Viral vectors for gene delivery to the nervous system. Nat Rev Neurosci 2003; 4: 353–364.
Regardsoe EL, McMenamin MM, Charlton HM, Wood MJ . Local adenoviral expression of Fas ligand upregulates pro-inflammatory immune responses in the CNS. Gene Ther 2004; 11: 1462–1474.
Tada T, Nguyen JB, Hitoshi Y, Watson NP, Dunn JF, Ohara S et al. Diffuse encephaloventriculitis and substantial leukoencephalopathy after intraventricular administration of recombinant adenovirus. Neurol Res 2005; 27: 378–386.
Thomas CE, Schiedner G, Kochanek S, Castro MG, Lowenstein PR . Peripheral infection with adenovirus causes unexpected long-term brain inflammation in animals injected intracranially with first-generation, but not with high-capacity, adenovirus vectors: toward realistic long-term neurological gene therapy for chronic diseases. Proc Natl Acad Sci USA 2000; 97: 7482–7487.
Chou AK, Yang LC, Wu PC, Wong WT, Liu GS, Chen JT et al. Intrathecal gene delivery of glial cell line-derived neurotrophic factor ameliorated paraplegia in rats after spinal ischemia. Brain Res Mol Brain Res 2005; 133: 198–207.
Driesse MJ, Esandi MC, Kros JM, Avezaat CJ, Vecht C, Zurcher C et al. Intra-CSF administered recombinant adenovirus causes an immune response-mediated toxicity. Gene Ther 2000; 7: 1401–1409.
Martino G, Furlan R, Comi G, Adorini L . The ependymal route to the CNS: an emerging gene-therapy approach for MS. Trends Immunol 2001; 22: 483–490.
Takada J, Ooboshi H, Ago T, Kitazono T, Yao H, Kadomatsu K et al. Postischemic gene transfer of midkine, a neurotrophic factor, protects against focal brain ischemia. Gene Ther 2005; 12: 487–493.
Poliani PL, Brok H, Furlan R, Ruffini F, Bergami A, Desina G et al. Delivery to the central nervous system of a nonreplicative herpes simplex type 1 vector engineered with the interleukin 4 gene protects rhesus monkeys from hyperacute autoimmune encephalomyelitis. Hum Gene Ther 2001; 12: 905–920.
Pluchino S, Zanotti L, Rossi B, Brambilla E, Ottoboni L, Salani G et al. Neurosphere-derived multipotent precursors promote neuroprotection by an immunomodulatory mechanism. Nature 2005; 436: 266–271.
Amadio S, Pluchino S, Brini E, Morana P, Guerriero R, Boneschi FM et al. Motor evoked potentials in a mouse model of chronic multiple sclerosis. Muscle Nerve 2006; 33: 265–273.
Sakaguchi S . Naturally arising Foxp3-expressing CD25+CD4+ regulatory T cells in immunological tolerance to self and non-self. Nat Immunol 2005; 6: 345–352.
Giarratana N, Penna G, Amuchastegui S, Mariani R, Daniel KC, Adorini L . A vitamin D analog down-regulates proinflammatory chemokine production by pancreatic islets inhibiting T cell recruitment and type 1 diabetes development. J Immunol 2004; 173: 2280–2287.
Glabinski AR, Bielecki B, Ransohoff RM . Chemokine upregulation follows cytokine expression in chronic relapsing experimental autoimmune encephalomyelitis. Scand J Immunol 2003; 58: 81–88.
Nakajima T, Inagaki N, Tanaka H, Tanaka A, Yoshikawa M, Tamari M et al. Marked increase in CC chemokine gene expression in both human and mouse mast cell transcriptomes following Fcepsilon receptor I cross-linking: an interspecies comparison. Blood 2002; 100: 3861–3868.
Lu LF, Lind EF, Gondek DC, Bennett KA, Gleeson MW, Pino-Lagos K et al. Mast cells are essential intermediaries in regulatory T-cell tolerance. Nature 2006; 442: 997–1002.
Furlan R, Poliani PL, Marconi PC, Bergami A, Ruffini F, Adorini L et al. Central nervous system gene therapy with interleukin-4 inhibits progression of ongoing relapsing-remitting autoimmune encephalomyelitis in Biozzi AB/H mice. Gene Ther 2001; 8: 13–19.
Pace L, Rizzo S, Palombi C, Brombacher F, Doria G . Cutting edge: IL-4-induced protection of CD4+CD25− Th cells from CD4+CD25+ regulatory T cell-mediated suppression. J Immunol 2006; 176: 3900–3904.
Bluestone JA, Tang Q . Therapeutic vaccination using CD4+CD25+ antigen-specific regulatory T cells. Proc Natl Acad Sci USA 2004; 101 (Suppl 2): 14622–14626.
Giarratana N, Penna G, Gregori S, Daniel KC, Adorini L . Exploiting the potential of regulatory T cells in the control of type 1 diabetes. In: Taams L, Akbar AN, Wauben MHM (eds). Regulatory T Cells in Inflammation. Birkhäuser: Basel, Switzerland, 2005, pp 95–110.
Iellem A, Mariani M, Lang R, Recalde H, Panina-Bordignon P, Sinigaglia F et al. Unique chemotactic response profile and specific expression of chemokine receptors CCR4 and CCR8 by CD4(+)CD25(+) regulatory T cells. J Exp Med 2001; 194: 847–853.
Sather BD, Treuting P, Perdue N, Miazgowicz M, Fontenot JD, Rudensky AY et al. Altering the distribution of Foxp3+ regulatory T cells results in tissue-specific inflammatory disease. J Exp Med 2007; 204: 1335–1347.
Columba-Cabezas S, Serafini B, Ambrosini E, Sanchez M, Penna G, Adorini L et al. Induction of macrophage-derived chemokine/CCL22 expression in experimental autoimmune encephalomyelitis and cultured microglia: implications for disease regulation. J Neuroimmunol 2002; 130: 10–21.
Furlan R, Kurne A, Bergami A, Brambilla E, Maucci R, Gasparini L et al. A nitric oxide releasing derivative of flurbiprofen inhibits experimental autoimmune encephalomyelitis. J Neuroimmunol 2004; 150: 10–19.
Kohm AP, Carpentier PA, Anger HA, Miller SD . Cutting edge: CD4+CD25+ regulatory T cells suppress antigen-specific autoreactive immune responses and central nervous system inflammation during active experimental autoimmune encephalomyelitis. J Immunol 2002; 169: 4712–4716.
Chen HH, Mack LM, Kelly R, Ontell M, Kochanek S, Clemens PR . Persistence in muscle of an adenoviral vector that lacks all viral genes. Proc Natl Acad Sci USA 1997; 94: 1645–1650.
Recchia A, Parks RJ, Lamartina S, Toniatti C, Pieroni L, Palombo F et al. Site-specific integration mediated by a hybrid adenovirus/adeno-associated virus vector. Proc Natl Acad Sci USA 1999; 96: 2615–2620.
Acknowledgements
This work was supported by grants from the Italian Multiple Sclerosis Foundation (FISM), the Myelin Project, the Cariplo Foundation and the Italian Ministry of Education and Scientific Research (MIUR-FIRB). We acknowledge the technical help of V Lampasona, C Porcheri and L Muzio.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on Gene Therapy website (http://www.nature.com/gt)
Rights and permissions
About this article
Cite this article
Butti, E., Bergami, A., Recchia, A. et al. IL4 gene delivery to the CNS recruits regulatory T cells and induces clinical recovery in mouse models of multiple sclerosis. Gene Ther 15, 504–515 (2008). https://doi.org/10.1038/gt.2008.10
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2008.10
Keywords
This article is cited by
-
Intranasal administration of interleukin-4 ameliorates depression-like behavior and biochemical alterations in mouse submitted to the chronic unpredictable mild stress: modulation of neuroinflammation and oxidative stress
Psychopharmacology (2023)
-
A review of possible therapies for multiple sclerosis
Molecular and Cellular Biochemistry (2021)
-
Alternatively activated macrophages; a double-edged sword in allergic asthma
Journal of Translational Medicine (2020)
-
Prolonged residence of an albumin–IL-4 fusion protein in secondary lymphoid organs ameliorates experimental autoimmune encephalomyelitis
Nature Biomedical Engineering (2020)
-
Intracisternal delivery of PEG-coated gold nanoparticles results in high brain penetrance and long-lasting stability
Journal of Nanobiotechnology (2019)