Abstract
Purpose:
To undertake the first prospective cost-effectiveness study of whole-exome sequencing (WES) as an early, routine clinical test for infants with suspected monogenic disorders.
Methods:
Cost data for diagnosis-related investigations and assessments were collected for a prospective, sequential clinical cohort of infants (N = 40) who underwent singleton WES in parallel to usual diagnostic care. We determined costs per patient, costs per diagnosis, and incremental costs per additional diagnosis for three alternative strategies for integrating WES into the diagnostic trajectory. We performed a sensitivity analysis to examine the robustness of estimates and bootstrapping (500 replications) to examine their distributions.
Results:
Standard care achieved an average cost per diagnosis of AU$27,050 (US$21,099) compared with AU$5,047 (US$3,937) for singleton WES. If WES had been performed after exhaustive standard investigation, then there would have been an incremental cost per additional diagnosis of AU$8,112 (US$ 6,327). Using WES to replace some investigations decreases this incremental cost to AU$2,622 (US$2,045), whereas using it to replace most investigations results in a savings per additional diagnosis of AU$2,182 (US$1,702).
Conclusion:
Use of WES early in the diagnostic pathway more than triples the diagnostic rate for one-third the cost per diagnosis, providing strong support for reimbursement as a clinical test.
Genet Med advance online publication 26 January 2017
Similar content being viewed by others
Introduction
Next-generation sequencing (NGS) technologies enable rapid sequencing of genes simultaneously and are changing the paradigm of Mendelian disease diagnosis. However, numerous barriers to the translation of this technology into health care have been documented.1 Not least among these is the need for evidence of cost-effectiveness and optimal timing of patient testing to guide clinician decision-making and test reimbursement. Although it has been proposed that early and accurate diagnosis using whole-exome sequencing (WES) will optimize opportunities to influence clinical management and decrease costs,2 WES is commonly offered to patients who are difficult to diagnose clinically after exhaustive investigation. These patients commonly are analyzed as “trios,” with parents also sequenced. None of these situations reflects the most common potential use of WES: for single patients with features strongly suggestive of monogenic conditions and as an early test in the diagnostic pathway. With the increasing availability of WES in clinical practice, and in the setting of limited health-care resources, there is an urgent need to develop evidence regarding how best to integrate WES in the diagnostic pathway to maximize both clinical utility and cost-effectiveness.
The diagnostic utility of WES in clinical practice is increasingly clear, with diagnostic yields of 25–30%3,4,5,6 established by several large-scale studies of adult and pediatric patients referred to laboratories for testing. Cost-effectiveness could not be analyzed as part of these studies because data regarding prior genetic testing and other diagnostic investigations were not available.
Cost studies of genomic sequencing have, to date, been retrospective and conducted for “difficult to diagnose” patients who have not received a diagnosis through standard molecular investigations. Soden et al. (2014) performed a retrospective study of patients with a variety of phenotypes who received a diagnosis using parent–child (trio) genome sequencing (WES or whole-genome sequencing), generally after substantial prior investigation. As a surrogate for cost-effectiveness, they determined that the mean cost of prior negative diagnostic testing for these children was US$19,100 per family.7 Another retrospective study of 17 undiagnosed patients with intellectual disability who had been extensively tested over many years found that the mean cost of prior investigation was US$16,409.8 Each of these studies indicated that WES would have been less expensive than other investigations. However, because retrospective studies do not include those who received a diagnosis through usual diagnostic care, their applicability to clinical practice is very limited. In a clinical setting, it is not possible to know in advance which patients will receive a molecular diagnosis. Therefore, the true cost-effectiveness and optimal timing of genomic sequencing can only be determined prospectively, with a cohort of patients who have not previously undergone molecular investigations.
Although essential for its introduction into established and reimbursed clinical practice, to date there has been no prospective study of the cost-effectiveness of WES compared with standard diagnostic care, nor has there been an evaluation of the optimal timing of the use of WES. Furthermore, retrospective studies have conducted sequencing with trios, not singleton patients. We therefore investigated the cost-effectiveness of standard diagnostic care in comparison to singleton WES in a cohort of 40 infants with features suggestive of monogenic disorders. These infants underwent singleton WES as a first-line molecular test in parallel with standard diagnostic care. We have modeled the relative cost-effectiveness of integrating singleton WES at various points in the diagnostic trajectory and undertook bootstrapping (500 replications) to estimate the reliability of the results.
Materials and Methods
Study design
This was a prospective study of patients attending a tertiary-level children’s hospital within the publically funded health-care system in Australia. The study was conducted to determine whether testing of individual patients (singletons) using WES as a first-line test is more cost-effective than standard investigations and to identify the optimal timing of WES in the diagnostic pathway. To ascertain this, we performed singleton WES in parallel with standard investigations (including commercial single-gene or multigene panel sequencing when clinically indicated). Health economic analysis was undertaken from the funded hospital system perspective encompassing costs per patient, costs per diagnosis, and incremental costs per additional diagnosis for three alternative strategies for integrating WES into the diagnostic trajectory.
Study participants
Cost data for the first 40 infants (0–2 years of age) recruited into a previously published study assessing the diagnostic and clinical utility of singleton WES9 were available and were used to perform the cost-effectiveness analysis. Recruitment is fully described elsewhere. In brief, infants were ascertained during clinical care and eligible if they presented with multiple congenital abnormalities and dysmorphic features or other features strongly suggestive of monogenic disorders such as neurometabolic conditions and skeletal dysplasias. Infants were not considered for the study if they had specific clinical presentations indicative of conditions that are not genetically heterogeneous (e.g., Apert syndrome or achondroplasia), except when testing for the suspected disorder was not commercially available, and “orphan phenotypes” unlikely to be caused by currently known genes. Infants who had undergone previous sequencing tests were not eligible for inclusion. Infants whose families consented to the study underwent WES in parallel with standard investigations, including commercial single-gene or multigene panel sequencing when clinically indicated and available.
The study was part of the Melbourne Genomics Health Alliance demonstration project and received Human Research Ethics Committee approval (13/MH/326). Informed written consent was obtained from the parents of the participants.
Diagnostic trajectory
We calculated the duration of the diagnostic trajectory from age of onset of symptoms until a diagnosis was established or an uninformative WES report was issued. Procedures for exome sequencing, variant detection and filtering, and assessment of variant pathogenicity have been previously described.9 Analysis of WES data was limited to genes known to cause Mendelian conditions, and only variants relevant to the participant’s phenotype were assessed with regard to pathogenicity.
Costs
We collected cost data using a bottom-up approach10,11,12 by identifying all of the resources that were used to provide a service and by assigning a value to each of those resources. We summed these values and linked to a unit of activity to derive a total unit cost.
A clinical geneticist extracted all investigations, procedures, and assessments that occurred for diagnostic purposes from the medical records. We obtained the costs of investigations and patient encounters from the hospital, state government, and testing laboratories. General patient care costs were not included. We converted costs in overseas currencies into Australian dollars (AU$) based on the exchange rate of AU$1=US$0.78 on 30 January 2015 (source: http://www.xe.com), which is approximately the average time when accounts were paid. The costs of individual investigations are presented in Table 2.
Economic analysis
We calculated the costs of the standard diagnostic and the singleton WES pathways and the diagnostic yield for each pathway. We present counterfactual diagnostic pathway models of care incorporating the use of WES into standard diagnostic care as the “last resort” diagnostic approach (model 1) and at earlier points in the diagnostic trajectory (models 2 and 3) to investigate which model was most cost-effective ( Figure 1 ).13,14,15 We defined the base case as the standard diagnostic pathway comprising the investigations and outpatient assessments that occurred primarily for diagnostic purposes. We defined the cost of the WES diagnostic pathway as the clinical service cost of singleton WES provided by the local clinical laboratory services (Victorian Clinical Genetic Services Pathology). The service delivery model included two consultations with a clinical geneticist and a genetic counselor, one at initial assessment and test initiation and the other at result disclosure. The cost of singleton WES includes analysis restricted to genes associated with Mendelian disease and reporting of variants related to phenotype only. It utilized a custom-built pipeline, Cpipe, which was designed using publically available resources.16
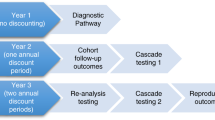
Diagnostic trajectory and resulting diagnostic yield and costs per patient for standard care and for integrating singleton whole-exome sequencing (WES) using three models. In model 1, standard investigations are exhausted first, including all planned tests, resulting in an additional six diagnoses (a total of 13), and WES is performed as a last resort for patients who remain undiagnosed. In model 2, WES replaces some investigations, particularly gene sequencing tests, complex biochemical tests, and invasive tests. In model 3, WES replaces most investigations. Dx, diagnosis.
WES as a last resort (counterfactual model 1). Standard diagnostic care options are exhausted first, including completing all investigations that were planned by clinicians at the time of enrollment, but were not performed within the timeframe of the study. WES is performed as a last resort in patients who remain undiagnosed through standard care.
WES replaces some tests (counterfactual model 2). In this model, singleton WES replaces some investigations. Patients undergo basic biochemical, imaging, neurophysiological studies, and subspecialist assessments as in the standard pathway because these can guide empiric management while a definitive diagnosis is awaited. WES replaces all genetic testing except single-nucleotide polymorphism microarray analysis due to the current limited ability of WES to detect structural variation. In this model, WES also replaces complex biochemistry testing, which is performed in specialized laboratories, and testing that is burdensome for patients, such as invasive tissue biopsies. Costs of sample shipments and of operating room/anesthetic time are eliminated as a result. There are two consultations with a clinical geneticist and genetic counselor per patient, one for initial assessment and WES initiation and the other for return of results.
WES replaces most tests (counterfactual model 3). WES is used as a first-line investigation that replaces all other diagnostic investigations and assessments, including biochemical investigations and imaging studies. Single-nucleotide polymorphism microarray is still performed to exclude copy-number variants. There are two appointments with a clinical geneticist and genetic counselor for initial assessment and test initiation and for return of results.
Statistical analysis
We examined characteristic variables of patients such as age, sex, parental consanguinity, principal phenotypic feature, primary indication for WES, and duration of diagnostic trajectory. We estimated the average cost per patient, cost per diagnosis, and incremental cost per additional diagnosis for standard diagnostic care and for the three counterfactual WES integration pathways. To allow extrapolation of the findings in our cohort of 40 to a larger population and to estimate the reliability of the estimates, we created 500 replicated data sets using bootstrap methods. To do this, we repeatedly drew a random sample of 40 records (i.e., total number of records) with replacement from the study participant records (repeated 500 times to create 500 replicated data sets). Then, to estimate the distribution of each outcome, we calculated the outcomes (such as the average cost per patient, cost per diagnosis, and incremental cost per additional diagnosis) for each replicated data set, thus generating 500 estimates of each outcome. Based on these distributions, we estimated 95% confidence intervals (CIs) for each outcome using the percentile method.17 We performed bootstrap simulations in SAS version 9.4. All other statistical analyses were performed using Microsoft Excel. We performed a sensitivity analysis to determine the impact of the price of WES by using the highest cost of singleton WES as a clinical service in Australian laboratories (AU$3,200; A. Sinclair, personal communication).
Results
Diagnostic yield and costs
Characteristics and indications for testing of the 40 infants included in this study are summarized in Table 1 . Features of the underlying disorder were present at birth in 36 participants (90%). The mean duration of the diagnostic trajectory was 395 days (range, 167–831 days). Table 2 shows the cost of each diagnostic test. The costs for each patient are provided in the supplementary material and ranged between AU$1,054.59 and 20,743.69 (Supplementary Table S1 online).
Through standard investigations, we obtained a molecular diagnosis for seven participants (17.5%) in this cohort. An average of AU$4,733.81 (95% CI: $3,693.33–5,894.96) was spent on diagnostic investigations and assessments per patient ( Table 3 ), and the cost per successful diagnosis was AU$27,050.36 (95% CI: $15,365.51–68,529.77). Seventeen of 40 participants (42.5%) underwent a total of 22 genetic tests in addition to single-nucleotide polymorphism microarray as part of standard clinical care during the study period. Another 18 tests were considered at enrollment but not performed by clinicians within the timeframe of the study due to financial or other constraints. These would have yielded another six diagnoses if performed. There was no perceived alteration in the pattern of clinician ordering of standard investigations during the timeframe of the study.
Singleton WES resulted in a molecular diagnosis of 26 genetic conditions in 25 of 40 infants, indicating a diagnostic rate of 62.5%. All molecular diagnoses made by standard testing were also made by singleton WES. The average cost per patient of clinical WES for this cohort was AU$3154.79 (AU$2,000 for clinical WES and AU$1,154.79 for consultations with a clinical geneticist and genetic counselor), and cost per successful diagnosis was AU$5,047.66.
Incremental cost-effectiveness throughout the diagnostic trajectory
Integrating WES after exhaustive standard investigation (counterfactual model 1) resulted in an incremental cost per additional diagnosis of AU$8,112 (95% CI: $5,850.95–11,966.87) in this patient cohort ( Table 3 ), although the cost per successful diagnosis (AU$13,414.61; 95% CI: $10,164.61–18,351.32) was lower compared with standard care (AU$27,050.36; 95% CI: $15,365.51–68,529.77).
For model 2, in which WES replaced some investigations, the incremental cost per additional diagnosis was AU$2,622.16 (95% CI: $847.09–4,459.16) compared with standard care. The cost per diagnosis was AU$9,462.06 (95% CI: $7,497.48 to AU$12,618.76), which was $17,588.30 less than standard diagnostic care.
Integrating WES as a first-line test replacing most investigations (counterfactual model 3) was cost-saving compared with standard diagnostic care. The incremental cost saving per additional diagnosis was AU$2,182.27 (95% CI: $129.92–5,855.02). The cost per diagnosis was AU$6,002.86 (95% CI: $4,841.02–7,898.51), which was AU$21,047.50 less than standard diagnostic care, and the average cost per patient was AU$3751.79, $982 less than standard diagnostic care.
The order of investigation proposed for each model, diagnostic yield, and costs per diagnosis are summarized in Figure 1 . Results of bootstrapped simulations are presented as scatter plots on a cost-effectiveness plane ( Figure 2 ), where each point represents the result of each simulation. The cost-effectiveness plane demonstrates that WES as a first-line test replacing most investigations (counterfactual model 3) is dominant (i.e., less cost with a higher number of diagnoses) compared with standard diagnostic care in 97% of 500 simulations, thus offering better value for money than the other two WES integration models.
Cost-effectiveness for whole-exome sequencing (WES). Model 1, WES as a last resort; model 2, replacing some investigations; and model 3, WES as first-line test. Each point in the scatter plot represents the result of each of 500 bootstrapped simulations. Points below the x axis indicate that the use of WES is less costly while providing a higher number of diagnoses compared with standard diagnostic care for those simulations. This means that for each of these simulations WES is “dominant,” that is, costs less while providing greater benefit than standard diagnostic care, and is thus considered cost-effective. Points above the x axis indicate that the use of WES is more expensive but provides a higher number of diagnoses compared with standard diagnostic care for those simulations. WES may, in this situation, still be regarded as cost-effective if the incremental cost for one additional diagnosis is less than the cost we are willing to pay for each additional diagnosis.
Sensitivity analysis
When we consider the highest cost per WES of AU$3200, WES still performs well compared with standard care ( Table 3 ), particularly when integrated as a first-line test replacing most other investigations (counterfactual model 3). The average cost per diagnosis is then AU$7,922.86 (95% CI: $6,041.02–9,098.51), approximately one-third of the cost of standard diagnostic care, and an incremental cost per additional diagnosis is AU$484.39 (95% CI: −$2,548.09 to 2,872.49).
Discussion
Currently, genomic sequencing is predominantly used in clinical research studies or in those patients who have been extensively investigated but remain undiagnosed. Our study provides the basis for the transition of genomic sequencing from this use toward its application as a first-tier test in a clinical setting. It is the first study to prospectively compare the cost-effectiveness of a genomic sequencing test relative with standard diagnostic care. Using singleton WES as a first-line sequencing test in our cohort in parallel with standard diagnostic care enabled us to demonstrate that introducing WES early in the diagnostic trajectory provides the greatest opportunity to improve the cost-effectiveness of the diagnostic process.
The traditional pathway for rare disease diagnosis is burdensome for patients, families, and health-care systems. The process is typically protracted and involves repeated assessments. Some patients, particularly those with neurodevelopmental phenotypes, undergo complex biochemical, imaging, and neurophysiological investigations, which serve to narrow the diagnostic possibilities. Some tests are invasive and require pediatric patients to be anesthetized. If the patient’s phenotype suggests a single gene or group of genes, then specific genetic testing is undertaken, using a tiered approach, whereby the results of one test are awaited before another is initiated.
To date, addressing the backlog of extensively investigated, undiagnosed patients has been a priority for clinicians. Many of the patients in the previous studies reporting the diagnostic and clinical utility of WES appear to be in this category.2,3,4,7,18,19 This may reflect deliberate selection procedures, clinician referral patterns, or, in the case of FORGE Canada,2 research primarily aimed at new gene discovery. It is common to use WES as a last resort, with referrals for WES often made after multiple previous uninformative sequencing tests, including gene panels. Previously, in the absence of evidence, and given the cost and the additional interpretation and counseling burdens of WES, it has appeared attractive to reserve it for patients when a diagnosis could not be reached through standard investigations. This reduced the overall number of patients undergoing WES, thereby avoiding the use of a complex and expensive test when a simpler and cheaper test might achieve the same result. However, the average cost of a single gene or panel test either performed or planned in our cohort was AU$2,186.56—greater than the cost of WES—and the majority of traditional gene tests did not yield a diagnosis.
Our data clearly demonstrate that applying WES at the end of the diagnostic trajectory is the least cost-effective option, resulting in more than twice the average expenditure per patient and an incremental cost per additional diagnosis of AU$8,111.82 compared with standard diagnostic care. All the costs of standard diagnostic care are incurred, but because only a small proportion of patients are diagnosed, the majority of patients would still undergo WES. Because the standard diagnostic pathway is time-consuming and tiered, a definitive diagnosis by WES would be delayed for a number of patients, with delays in the benefits in clinical management, cascade testing of relatives, and enabling informed reproductive choices for parents.9
The alternative option is to integrate WES early in the diagnostic trajectory. In our infant cohort, singleton WES is a highly effective diagnostic tool, with a large proportion of infants receiving accurate molecular diagnosis.9 When WES replaces most other investigations (counterfactual model 3), there is a cost savings per additional diagnosis. Although this approach may be appropriate for patients presenting in an ambulatory setting with dysmorphic features and developmental delay, it would not be suitable for acutely unwell patients with monogenic disorders, in whom biochemical, imaging, and neurophysiological studies could provide information to guide management while a definitive diagnosis is awaited. In this latter scenario—when WES replaces some investigations (counterfactual model 2)—there is still a considerably lower incremental cost per additional diagnosis than applying it as a last resort test. Thus, integrating WES early in the diagnostic pathway provides the greatest opportunity to reduce costs and improve the cost-effectiveness of Mendelian disease diagnosis.
Our estimates of the cost-effectiveness of WES are conservative due to the young age of the patients and their relatively short diagnostic trajectory. The impact of uninformative WES on patient investigation and care is not yet known and merits further research. Costs may be relatively contained if clinicians focus on investigation of relevant conditions that were not adequately excluded by WES for technical reasons. Alternatively, clinicians may continue to rely on a broad range of standard investigations for these patients. Achieving the greatest cost-effectiveness in acutely unwell patients may be reliant on the ability to perform genomic testing rapidly. Our own experience with singleton WES shows that it is possible to achieve a result in less than 3 weeks (unpublished data), and one proof-of-principle study has reported a 26-h time to result using whole-genome sequencing.20
This study was conducted in Australia within a socialized medicine system. Budgetary considerations place constraints on the investigations performed as part of standard care. Therefore, these data are likely to be a conservative estimate of cost-effectiveness in health-care settings with fewer constraints. We also note that unit costs vary across health-care settings and systems, and diagnostic yields vary depending on indications for testing. To assist clinicians in gaining insight into the potential cost-effectiveness in their own setting and develop cases for funding, costs for investigations in other health-care systems can be entered into the accompanying spreadsheet (Supplementary Material 2 online) to calculate the cost per diagnosis for different cohorts.
Cost-effective genomic medicine depends on clinicians using the right test for the right patient at the right time. This study supports the use of singleton WES early in the diagnostic trajectory for patients with features strongly suggestive of monogenic conditions. Alternative diagnostic pathways may be more cost-effective for patients with other phenotypes. The greater cost of trio WES, for example, may be offset if this testing results in a higher diagnostic yield for patients who have apparently unique phenotypes or nonsyndromic intellectual disability. Further research is needed to compare the cost-effectiveness of trios with that of singleton WES. For our cohort of infants, however, the diagnostic yield is comparable to those found in trio studies of similar cohorts.21,22 As discussed elsewhere, the diagnostic yield of singleton WES in our cohort is likely to reflect the timing of the test, that patients had not undergone previous sequencing tests, and the phenotypic characteristics of the patients.9
In conclusion, this prospective study of a clinical cohort provides essential evidence for the translation of WES into clinical practice. We have demonstrated that singleton WES is cost-effective when provided to patients as an early investigation rather than reserved as a last resort. These data support arguments for test reimbursement—a significant barrier to the application of genomic sequencing tests in the clinic—and are the basis of a health economic case to the Australian Government for reimbursement of WES for infants suspected to have monogenic disorders. Furthermore, this study provides an innovative model for determining the cost-effectiveness for different clinical indications in the future.
Disclosure
The authors declare no conflict of interest.
References
Manolio TA, Abramowicz M, Al-Mulla F, et al. Global implementation of genomic medicine: We are not alone. Sci Transl Med 2015;7:290ps13.
Sawyer SL, Hartley T, Dyment DA, et al.; FORGE Canada Consortium; Care4Rare Canada Consortium. Utility of whole-exome sequencing for those near the end of the diagnostic odyssey: time to address gaps in care. Clin Genet 2016;89:275–284.
Lee H, Deignan JL, Dorrani N, et al. Clinical exome sequencing for genetic identification of rare Mendelian disorders. JAMA 2014;312:1880–1887.
Yang Y, Muzny DM, Reid JG, et al. Clinical whole-exome sequencing for the diagnosis of mendelian disorders. N Engl J Med 2013;369:1502–1511.
Yang Y, Muzny DM, Xia F, et al. Molecular findings among patients referred for clinical whole-exome sequencing. JAMA 2014;312:1870–1879.
Farwell KD, Shahmirzadi L, El-Khechen D, et al. Enhanced utility of family-centered diagnostic exome sequencing with inheritance model-based analysis: results from 500 unselected families with undiagnosed genetic conditions. Genet Med 2015;17:578–586.
Soden SE, Saunders CJ, Willig LK, et al. Effectiveness of exome and genome sequencing guided by acuity of illness for diagnosis of neurodevelopmental disorders. Sci Transl Med 2014;6:265ra168.
Monroe GR, Frederix GW, Savelberg SM, et al. Effectiveness of whole-exome sequencing and costs of the traditional diagnostic trajectory in children with intellectual disability. Genet Med 2016;18:949–956.
Stark Z, Tan TY, Chong B, et al. A prospective evaluation of whole-exome sequencing as a first-tier molecular test in infants with suspected monogenic disorders. Genet Med 2016;18:1090–1096.
Chapko MK, Liu CF, Perkins M, Li YF, Fortney JC, Maciejewski ML. Equivalence of two healthcare costing methods: bottom-up and top-down. Health Econ 2009;18:1188–1201.
Mercier G, Naro G. Costing hospital surgery services: the method matters. PLoS One 2014;9:e97290.
Vallejo-Torres L, Morris S, Kinge JM, Poirier V, Verne J. Measuring current and future cost of skin cancer in England. J Public Health (Oxf) 2014;36:140–148.
Gertler PJ, Martinez S, Premand P, Rawlings LB, Vermeersch CM. Impact evaluation in practice. World Bank Publications; 2011. https://siteresources.worldbank.org/EXTHDOFFICE/Resources/5485726-1295455628620/Impact_Evaluation_in_Practice.pdf.
Khandker SR, Koolwal GB, Samad HA. Handbook on Impact Evaluation: Quantitative Methods and Practices. World Bank Publications, 2010. https://openknowledge.worldbank.org/handle/10986/2693.
World Health Organization. WHO Guide to Identifying the Economic Consequences of Disease and Injury. 2009.
Sadedin SP, Dashnow H, James PA, et al.; Melbourne Genomics Health Alliance. Cpipe: a shared variant detection pipeline designed for diagnostic settings. Genome Med 2015;7:68.
Briggs AH, Wonderling DE, Mooney CZ. Pulling cost-effectiveness analysis up by its bootstraps: a non-parametric approach to confidence interval estimation. Health Econ 1997;6:327–340.
Wright CF, Fitzgerald TW, Jones WD, et al.; DDD study. Genetic diagnosis of developmental disorders in the DDD study: a scalable analysis of genome-wide research data. Lancet 2015;385:1305–1314.
Yang W, Lee PP, Thong MK, et al. Compound heterozygous mutations in TTC7A cause familial multiple intestinal atresias and severe combined immunodeficiency. Clin Genet 2015;88:542–549.
Saunders CJ, Miller NA, Soden SE, et al. Rapid whole-genome sequencing for genetic disease diagnosis in neonatal intensive care units. Sci Transl Med 2012;4:154ra135.
Willig LK, Petrikin JE, Smith LD, et al. Whole-genome sequencing for identification of Mendelian disorders in critically ill infants: a retrospective analysis of diagnostic and clinical findings. Lancet Respir Med 2015;3:377–387.
Daoud H, Luco SM, Li R, et al. Next-generation sequencing for diagnosis of rare diseases in the neonatal intensive care unit. CMAJ 2016;188:E254–E260.
Acknowledgements
We thank all collaborators in the Melbourne Genomics Health Alliance demonstration project and Leeanne Cavanough for administrative support in preparation of the manuscript. The study was funded by the founding organizations of the Melbourne Genomics Health Alliance (Royal Melbourne Hospital, Royal Children’s Hospital, University of Melbourne, Walter and Eliza Hall Institute, Murdoch Childrens Research Institute, Australian Genome Research Facility, and CSIRO) and the State Government of Victoria (Department of Health and Human Services). The involvement of AGRF was supported by sponsorship from Bioplatforms Australia and the NCRIS program.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Table S1
(DOCX 31 kb)
Rights and permissions
About this article
Cite this article
Stark, Z., Schofield, D., Alam, K. et al. Prospective comparison of the cost-effectiveness of clinical whole-exome sequencing with that of usual care overwhelmingly supports early use and reimbursement. Genet Med 19, 867–874 (2017). https://doi.org/10.1038/gim.2016.221
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gim.2016.221
Keywords
This article is cited by
-
Clinically significant changes in genes and variants associated with epilepsy over time: implications for re-analysis
Scientific Reports (2024)
-
Role of next generation sequencing in diagnosis and management of critically ill children with suspected monogenic disorder
European Journal of Human Genetics (2024)
-
Exploring the Cost Effectiveness of a Whole-Genome Sequencing-Based Biomarker for Treatment Selection in Patients with Advanced Lung Cancer Ineligible for Targeted Therapy
PharmacoEconomics (2024)
-
Genome sequencing as a generic diagnostic strategy for rare disease
Genome Medicine (2024)
-
Decoding complex inherited phenotypes in rare disorders: the DECIPHERD initiative for rare undiagnosed diseases in Chile
European Journal of Human Genetics (2024)