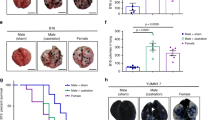
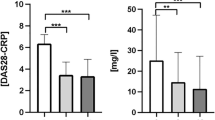
Abstract
Behcet’s disease (BD) exhibits more severe disease course and higher mortality among male patients. However, underlying mechanisms of gender differences in clinical manifestations and disease severity are unclear. The aim of this study was to determine whether testosterone (T) has any role on BD pathogenesis. We studied peripheral blood mononuclear cells (PBMC) and neutrophils of BD patients and controls. Functional assay of neutrophils, cytokine measurements of culture supernatants and gene expressions on both cells were analyzed before and after T incubation. Neutrophils were significantly activated after incubation with T in only BD patients. Incubation with T caused significantly elevated interleukin (IL)-12 and IL-2 in BD. Gene expression of IL-10 was significantly downregulated after incubation with T in BD, especially in male patients. The same difference was observed in IL-10 levels in culture supernatant after T. Baseline TLR4 expression was significantly higher in BD patients compared to healthy donors (HC). Toll-like receptor (TLR) 4 expression on PBMC was significantly elevated in female BD patients. ERAP1 expressions of all patients and controls were decreased under the T effect but it differed significantly between BD vs HC. Baseline IL23R expression was higher in BD males compared with females but the difference disappeared after T. When BD patients were analyzed separately, baseline C-C motif chemokine receptor1 (CCR1), STAT4, TLR4 and KLRC4 expressions were lower in males. Despite immunosuppressive behavior in healthy subjects, T causes neutrophil hyperactivation and TH1 type immune alterations in BD patients. Our results suggest that T may have a role in BD pathogenesis by altering the expression level of IL-10, TLR4, ERAP1, CCR1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 digital issues and online access to articles
$119.00 per year
only $19.83 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Gul A . Behcet’s disease: an update on the pathogenesis. Clin Exp Rheumatol 2001; 19 (5 suppl 24): S6–S12.
Kural-Seyahi E, Fresko I, Seyahi N, Ozyazgan Y, Mat C, Hamuryudan V et al. The long-term mortality and morbidity of Behcet syndrome: a 2-decade outcome survey of 387 patients followed at a dedicated center. Medicine 2003; 82: 60–76.
Alpsoy E, Donmez L, Onder M, Gunasti S, Usta A, Karincaoglu Y et al. Clinical features and natural course of Behcet’s disease in 661 cases: a multi-center study. Br J Dermatol 2007; 157: 901–906.
Yazici H, Tuzun Y, Pazarli H, Yurdakul S, Ozyazgan Y, Ozdoğan H et al. Influence of age of onset and patient’s sex on the prevalence and severity of manifestations of Behcet’s syndrome. Ann Rheum Dis 1984; 43: 783–789.
Maldini C, Lavalley MP, Cheminant M, de Menthon M, Mahr A . Relationships of HLA-B51 or B5 genotype with Behcet’s disease clinical characteristics: systematic review and meta-analyses of observational studies. Rheumatology 2012; 51: 887–900.
Fei Y, Webb R, Cobb BL, Direskeneli H, Saruhan-Direskeneli G, Sawalha AH . Identification of novel genetic susceptibility loci for Behcet’s disease using a genome-wide association study. Arthritis Res Ther 2009; 11: R66.
Remmers EF, Cosan F, Kirino Y, Ombrello MJ, Abaci N, Satorius C et al. Genome-wide association study identifies variants in the MHC class I, IL10, and IL23R-IL12RB2 regions associated with Behcet’s disease. Nat Genet 2010; 42: 698–702.
Mizuki N, Meguro A, Ota M, Ohno S, Shiota T, Kawagoe T et al. Genome-wide association studies identify IL23R-IL12RB2 and IL10 as Behcet’s disease susceptibility loci. Nat Genet 2010; 42: 703–706.
Hou S, Yang Z, Du L, Jiang Z, Shu Q, Chen Y et al. Identification of a susceptibility locus in STAT4 for Behcet‘s disease in Han Chinese in a genome-wide association study. Arthritis Rheum 2012; 64: 4104–4113.
Kirino Y, Bertsias G, Ishigatsubo Y, Mizuki N, Tugal-Tutkun I, Seyahi E et al. Genome-wide association analysis identifies new susceptibility loci for Behcet’s disease and epistasis between HLA-B 51 and ERAP1. Nat Genet 2013; 45: 202–207.
Kirino Y, Zhou Q, Ishigatsubo Y, Mizuki N, Tugal-Tutkun I, Seyahi E et al. Targeted resequencing implicates the familial Mediterranean fever gene MEFV and the toll-like receptor 4 geneTLR4 in Behcet disease. Proc Natl Acad Sci USA 2013; 110: 8134–8139.
Karasneh J, Gul A, Ollier WE, Silman AJ, Worthington J . Whole-genome screening for susceptibility genes in multicase families with Behcet’s disease. Arthritis Rheum 2005; 52: 1836–1842.
Alpsoy E, Elpek GO, Yilmaz F, Ciftcioglu MA, Akman A, Uzun S et al. Androgen receptor levels of oral and genital ulcers and skin pathergy test in patients with Behçet's disease. Dermatology 2005; 210: 31–35.
Yavuz S, Ozilhan G, Elbir Y, Tolunay A, Eksioglu-Demiralp E, Direskeneli H et al. Activation of neutrophils by testosterone in Behçet's disease. Clin Exp Rheumatol 2007; 25 (4 Suppl 45): S46–S51.
Gul A . Behcet’s disease as an autoinflammatory disorder. Curr Drug Targets Inflamm Allergy 2005; 4: 81–83.
González F . Inflammation in Polycystic Ovary Syndrome: underpinning of insulin resistance and ovarian dysfunction. Steroids 2012; 77: 300–305.
Hu W, Qiao J, Yang Y, Wang L, Li R . Elevated C-reactive protein and monocyte chemoattractant protein-1 in patients with polycystic ovary syndrome. Eur J Obstet Gynecol Reprod Biol 2011; 157: 53–56.
Repaci A, Gambineri A, Pasquali R . The role of low-grade inflammation in the polycystic ovary syndrome. Mol Cell Endocrinol 2011; 335: 30–41.
Gul A . Genetics of Behcet’s disease:lessons learned from genomwide association studies. Curr Opin Rheumatol 2014; 26: 56–63.
Wallace GR, Kondeatis E, Vaughan RW, Verity DH, Chen Y, Fortune F et al. IL-10 genotype analysis in patients with Behcet’s Disease. Hum Immunol 2007; 68: 122–127.
Onal S, Kazokoglu H, Koç A, Bavbek T, Direskeneli H, Yavuz S . Low dose and dose escalating therapy of interferon alfa-2a in the treatment of refractory and sight-threatening Behçet's uveitis. Clin Exp Rheumatol 2009; 27 (2 Suppl 53): S113–S114.
Kirino Y, Zhou Q, Ishigatsubo Y, Mizuki N, Tugal-Tutkun I, Seyahi E et al. Targeted resequencing implicates the familial Mediterranean fever gene MEFV and the toll-like receptor 4 gene TLR4 in Behçet disease. Proc Natl Acad Sci USA 2013; 110: 8134–8139.
Denkers EY, Del Rio L, Bennouna S . Neutrophil production of IL-12 and other cytokines during microbial infection. Chem Immunol Allergy 2003; 83: 95–114.
Hou S, Xiao X, LiF, Jiang Z, Kijlstra A et al. Two stage association study in Chinese Han identifies two independent associations in CCR1/CCR3 locus as candidate for Behcet’s disease susceptibility. Hum Genet 2012; 131: 1841–1850.
Ananthakrishnan P, Deitch EA . Gut origin sepsis and MODS: the role of sex hormones in modulating intestinal and distant organ injury: a review. XX vs XY 2003; 1: 108–117.
Sheth SU, Palange D, Xu DZ, Wei D, Feketeova E, Lu Q et al. Testosterone depletion or blockade in male rats protects against trauma hemorrhagic shock-induced distant organ injury by limiting gut injury and subsequent production of biologically active mesenteric lymph. J Trauma 2011; 71: 1652–1658.
Zolin SJ, Vodovotz Y, Forsythe RM, Rosengart MR, Namas R, Brown JB et al. The early evolving sex hormone environment is associated with significant outcome and inflammatory response differences after injury. J Trauma Acute Care Surg 2015; 78: 451–457.
Gonzales RJ, Duckles SP, Krause DN . Dihydrotestosterone stimulates cerebrovascular inflammation through NFkappaB, modulating contractile function. J Cereb Blood Flow Metab 2009; 29: 244–253.
Krause DN, Duckles SP, Gonzales RJ . Local oestrogenic/androgenic balance in the cerebral vasculature. Acta Physiol (Oxf) 2011; 203: 181–186.
Giltay EJ, Fonk JC, von Blomberg BM, Drexhage HA, Schalkwijk C, Gooren LJ . In vivo effects of sex steroids on lymphocyte responsiveness and immunoglobulin levels in humans. J Clin Endocrinol Metab 2000; 85: 1648–1657.
Bartel Y, Bauer B, Steinle A . Modulation of NK cell function by genetically coupled C-type lectin-like receptor/ligand pairs encoded in the human natural killer gene complex. Front Immunol 2013; 4: 362.
International Study Group for Behcet's Disease. Criteria for diagnosis of Behcet's disease. Lancet 1990; 335: 1078–1080.
Lehman R, Lehmann J, Sack U, Tamok A, Rothe G (eds). Flow cytometric analysis of intracellular stained cytokines, phosphoproteins and microbial antigens. Cellular Diagnostics, Basics Methods and Clinical Applications of Flow Cytometry. Karger: Basel, Switzerland, 2009: 459–475.
Acknowledgements
We are thankful to DrHasan Yazici and Haner Direskeneli for their valuable discussions about study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on Genes and Immunity website
Supplementary information
Rights and permissions
About this article
Cite this article
Yavuz, S., Akdeniz, T., Hancer, V. et al. Dual effects of testosterone in Behcet's disease: implications for a role in disease pathogenesis. Genes Immun 17, 335–341 (2016). https://doi.org/10.1038/gene.2016.28
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gene.2016.28
This article is cited by
-
Vascular Behçet syndrome: from pathogenesis to treatment
Nature Reviews Rheumatology (2023)
-
Nonendocrine mechanisms of sex bias in rheumatic diseases
Nature Reviews Rheumatology (2019)
-
Behçet’s syndrome: focus on pathogenetic background, clinical phenotypes and specific treatments
Internal and Emergency Medicine (2019)
-
Triggering agents and microbiome as environmental factors on Behçet’s syndrome
Internal and Emergency Medicine (2019)