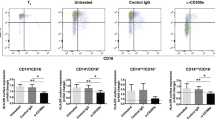
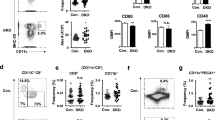
Abstract
Interleukin (IL)-10 is an important immunoregulatory cytokine that mediates its effects via a transmembrane receptor complex consisting of two different chains, IL-10R1 and IL-10R2. While IL-10R2 is ubiquitously expressed and does not bind IL-10 primarily, the expression of IL-10R1 determines cellular responsiveness. However, the current knowledge about the expression and regulation of IL-10R1 is still limited. Here we analyzed the expression of IL-10R1 on monocytic cells and demonstrated that human blood monocytes carried about 720 IL-10-binding sites on their surface. Compared with lymphocytes and various tissue cells and tissues, blood monocytes expressed the highest IL-10R1 levels. The in vitro differentiation of these cells into macrophages provoked a further increase of IL-10R1 surface expression. In contrast, their differentiation into myeloid dendritic cells (mDCs) resulted in reduced surface IL-10R1 levels. The different IL-10R1 levels expressed by monocyte-derived antigen-presenting cell populations were reflected in their different responsiveness toward IL-10. Importantly, also in vivo developed immature macrophages and mDCs showed different IL-10 sensitivity. These data suggest that, compared with monocytes and macrophages, mDCs partially escape from IL-10's inhibitory mechanisms by downregulating IL-10R1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 digital issues and online access to articles
$119.00 per year
only $19.83 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Sabat R, Grutz G, Warszawska K, Kirsch S, Witte E, Wolk K et al. Biology of interleukin-10. Cytokine Growth Factor Rev 2010; 21: 331–344.
Slight SR, Monin L, Gopal R, Avery L, Davis M, Cleveland H et al. IL-10 restrains IL-17 to limit lung pathology characteristics following pulmonary infection with Francisella tularensis live vaccine strain. Am J Pathol 2013; 183: 1397–1404.
Duell BL, Carey AJ, Tan CK, Cui X, Webb RI, Totsika M et al. Innate transcriptional networks activated in bladder in response to uropathogenic Escherichia coli drive diverse biological pathways and rapid synthesis of IL-10 for defense against bacterial urinary tract infection. J Immunol 2012; 188: 781–792.
Kuhn R, Lohler J, Rennick D, Rajewsky K, Muller W . Interleukin-10-deficient mice develop chronic enterocolitis. Cell 1993; 75: 263–274.
Mao H, Yang W, Lee PP, Ho MH, Yang J, Zeng S et al. Exome sequencing identifies novel compound heterozygous mutations of IL-10 receptor 1 in neonatal-onset Crohn's disease. Genes Immun 2012; 13: 437–442.
Woiciechowsky C, Asadullah K, Nestler D, Eberhardt B, Platzer C, Schoning B et al. Sympathetic activation triggers systemic interleukin-10 release in immunodepression induced by brain injury. Nat Med 1998; 4: 808–813.
Wolk K, Hoflich C, Zuckermann-Becker H, Docke WD, Volk HD, Sabat R . Reduced monocyte CD86 expression in postinflammatory immunodeficiency. Crit Care Med 2007; 35: 458–467.
Bergenfelz C, Medrek C, Ekstrom E, Jirstrom K, Janols H, Wullt M et al. Wnt5a induces a tolerogenic phenotype of macrophages in sepsis and breast cancer patients. J Immunol 2012; 188: 5448–5458.
Lin SY, Hsieh SC, Lin YC, Lee CN, Tsai MH, Lai LC et al. A whole genome methylation analysis of systemic lupus erythematosus: hypomethylation of the IL10 and IL1R2 promoters is associated with disease activity. Genes Immun 2012; 13: 214–220.
Hedrich CM, Rauen T, Apostolidis SA, Grammatikos AP, Rodriguez Rodriguez N, Ioannidis C et al. Stat3 promotes IL-10 expression in lupus T cells through trans-activation and chromatin remodeling. Proc Natl Acad Sci USA 2014; 111: 13457–13462.
Jung M, Sabat R, Kratzschmar J, Seidel H, Wolk K, Schonbein C et al. Expression profiling of IL-10-regulated genes in human monocytes and peripheral blood mononuclear cells from psoriatic patients during IL-10 therapy. Eur J Immunol 2004; 34: 481–493.
Lingnau M, Hoflich C, Volk HD, Sabat R, Docke WD . Interleukin-10 enhances the CD14-dependent phagocytosis of bacteria and apoptotic cells by human monocytes. Hum Immunol 2007; 68: 730–738.
Randolph GJ, Beaulieu S, Lebecque S, Steinman RM, Muller WA . Differentiation of monocytes into dendritic cells in a model of transendothelial trafficking. Science 1998; 282: 480–483.
Shouval DS, Biswas A, Goettel JA, McCann K, Conaway E, Redhu NS et al. Interleukin-10 receptor signaling in innate immune cells regulates mucosal immune tolerance and anti-inflammatory macrophage function. Immunity 2014; 40: 706–719.
Banchereau J, Steinman RM . Dendritic cells and the control of immunity. Nature 1998; 392: 245–252.
Ho AS, Liu Y, Khan TA, Hsu DH, Bazan JF, Moore KW . A receptor for interleukin 10 is related to interferon receptors. Proc Natl Acad Sci USA 1993; 90: 11267–11271.
Kotenko SV, Krause CD, Izotova LS, Pollack BP, Wu W, Pestka S . Identification and functional characterization of a second chain of the interleukin-10 receptor complex. EMBO J 1997; 16: 5894–5903.
Sabat R . IL-10 family of cytokines. Cytokine Growth Factor Rev 2010; 21: 315–324.
Finsterbusch M, Khare V, Campregher C, Evstatiev R, Gasche C . An intracytoplasmic IL-10 receptor variant permits rapid reduction in STAT3 activation. Genes Immun 2011; 12: 575–581.
Josephson K, Logsdon NJ, Walter MR . Crystal structure of the IL-10/IL-10R1 complex reveals a shared receptor binding site. Immunity 2001; 15: 35–46.
Reineke U, Schneider-Mergener J, Glaser RW, Stigler RD, Seifert M, Volk HD et al. Evidence for conformationally different states of interleukin-10: binding of a neutralizing antibody enhances accessibility of a hidden epitope. J Mol Recognit 1999; 12: 242–248.
Finbloom DS, Winestock KD . IL-10 induces the tyrosine phosphorylation of tyk2 and Jak1 and the differential assembly of STAT1 alpha and STAT3 complexes in human T cells and monocytes. J Immunol 1995; 155: 1079–1090.
Riley JK, Takeda K, Akira S, Schreiber RD . Interleukin-10 receptor signaling through the JAK-STAT pathway. Requirement for two distinct receptor-derived signals for anti-inflammatory action. J Biol Chem 1999; 274: 16513–16521.
Wolk K, Kunz S, Witte E, Friedrich M, Asadullah K, Sabat R . IL-22 increases the innate immunity of tissues. Immunity 2004; 21: 241–254.
Sabat R, Ouyang W, Wolk K . Therapeutic opportunities of the IL-22-IL-22R1 system. Nat Rev Drug Discov 2014; 13: 21–38.
Donnelly RP, Sheikh F, Dickensheets H, Savan R, Young HA, Walter MR . Interleukin-26: an IL-10-related cytokine produced by Th17 cells. Cytokine Growth Factor Rev 2010; 21: 393–401.
Witte K, Witte E, Sabat R, Wolk K . IL-28A, IL-28B, and IL-29: promising cytokines with type I interferon-like properties. Cytokine Growth Factor Rev 2010; 21: 237–251.
Wolk K, Witte E, Reineke U, Witte K, Friedrich M, Sterry W et al. Is there an interaction between interleukin-10 and interleukin-22? Genes Immun 2005; 6: 8–18.
Palucka KA, Taquet N, Sanchez-Chapuis F, Gluckman JC . Dendritic cells as the terminal stage of monocyte differentiation. J Immunol 1998; 160: 4587–4595.
Ziegler-Heitbrock HW, Fingerle G, Strobel M, Schraut W, Stelter F, Schutt C et al. The novel subset of CD14+/CD16+ blood monocytes exhibits features of tissue macrophages. Eur J Immunol 1993; 23: 2053–2058.
Palomares O, Martín-Fontecha M, Lauener R, Traidl-Hoffmann C, Cavkaytar O, Akdis M et al. Regulatory T cells and immune regulation of allergic diseases: roles of IL-10 and TGF-β. Genes Immun (e-pub ahead of print 24 July 2014; doi:10.1038/gene.2014.45).
Carson WE, Lindemann MJ, Baiocchi R, Linett M, Tan JC, Chou CC et al. The functional characterization of interleukin-10 receptor expression on human natural killer cells. Blood 1995; 85: 3577–3585.
Jurlander J, Lai CF, Tan J, Chou CC, Geisler CH, Schriber J et al. Characterization of interleukin-10 receptor expression on B-cell chronic lymphocytic leukemia cells. Blood 1997; 89: 4146–4152.
Liu Y, Wei SH, Ho AS, de Waal Malefyt R, Moore KW . Expression cloning and characterization of a human IL-10 receptor. J Immunol 1994; 152: 1821–1829.
Kunz S, Wolk K, Witte E, Witte K, Doecke WD, Volk HD et al. Interleukin (IL)-19, IL-20 and IL-24 are produced by and act on keratinocytes and are distinct from classical ILs. Exp Dermatol 2006; 15: 991–1004.
Jose P, Avdiushko MG, Akira S, Kaplan AM, Cohen DA . Inhibition of interleukin-10 signaling in lung dendritic cells by toll-like receptor 4 ligands. Exp Lung Res 2009; 35: 1–28.
Monteleone I, Platt AM, Jaensson E, Agace WW, Mowat AM . IL-10-dependent partial refractoriness to Toll-like receptor stimulation modulates gut mucosal dendritic cell function. Eur J Immunol 2008; 38: 1533–1547.
Cella M, Engering A, Pinet V, Pieters J, Lanzavecchia A . Inflammatory stimuli induce accumulation of MHC class II complexes on dendritic cells. Nature 1997; 388: 782–787.
Wakkach A, Fournier N, Brun V, Breittmayer JP, Cottrez F, Groux H . Characterization of dendritic cells that induce tolerance and T regulatory 1 cell differentiation in vivo. Immunity 2003; 18: 605–617.
Min S, Li L, Zhang M, Zhang Y, Liang X, Xie Y et al. TGF-beta-associated miR-27a inhibits dendritic cell-mediated differentiation of Th1 and Th17 cells by TAB3, p38 MAPK, MAP2K4 and MAP2K7. Genes Immun 2012; 13: 621–631.
Dhodapkar MV, Steinman RM, Sapp M, Desai H, Fossella C, Krasovsky J et al. Rapid generation of broad T-cell immunity in humans after a single injection of mature dendritic cells. J Clin Invest 1999; 104: 173–180.
Hsu FJ, Benike C, Fagnoni F, Liles TM, Czerwinski D, Taidi B et al. Vaccination of patients with B-cell lymphoma using autologous antigen-pulsed dendritic cells. Nat Med 1996; 2: 52–58.
Anguille S, Smits EL, Lion E, van Tendeloo VF, Berneman ZN . Clinical use of dendritic cells for cancer therapy. Lancet Oncol 2014; 15: e257–e267.
Edele F, Dudda JC, Bachtanian E, Jakob T, Pircher H, Martin SF . Efficiency of dendritic cell vaccination against B16 melanoma depends on the immunization route. PLoS ONE 2014; 9: e105266.
Wolk K, Docke WD, von Baehr V, Volk HD, Sabat R . Impaired antigen presentation by human monocytes during endotoxin tolerance. Blood 2000; 96: 218–223.
Wolk K, Warszawska K, Hoeflich C, Witte E, Schneider-Burrus S, Witte K et al. Deficiency of IL-22 contributes to a chronic inflammatory disease: pathogenetic mechanisms in acne inversa. J Immunol 2011; 186: 1228–1239.
Wolk K, Haugen HS, Xu W, Witte E, Waggie K, Anderson M et al. IL-22 and IL-20 are key mediators of the epidermal alterations in psoriasis while IL-17 and IFN-gamma are not. J Mol Med (Berl) 2009; 87: 523–536.
Witte E, Kokolakis G, Witte K, Philipp S, Doecke WD, Babel N et al. IL-19 is a component of the pathogenetic IL-23/IL-17 cascade in psoriasis. J Invest Dermatol 2014; 134: 2757–2767.
Wolk K, Witte E, Wallace E, Docke WD, Kunz S, Asadullah K et al. IL-22 regulates the expression of genes responsible for antimicrobial defense, cellular differentiation, and mobility in keratinocytes: a potential role in psoriasis. Eur J Immunol 2006; 36: 1309–1323.
Witte K, Gruetz G, Volk HD, Looman AC, Asadullah K, Sterry W et al. Despite IFN-lambda receptor expression, blood immune cells, but not keratinocytes or melanocytes, have an impaired response to type III interferons: implications for therapeutic applications of these cytokines. Genes Immun 2009; 10: 702–714.
Wolk K, Witte K, Witte E, Raftery M, Kokolakis G, Philipp S et al. IL-29 is produced by T(H)17 cells and mediates the cutaneous antiviral competence in psoriasis. Sci Transl Med 2013; 5: 204ra129.
Acknowledgements
We thank Antje Haeußler-Quade, Christa Liebenthal and Ricardo Wieseke for excellent technical help. Special thanks is due to Dr Nigel EA Crompton for his continuous courtesy to read the manuscript and to correct the English syntax.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
von Lanzenauer, S., Wolk, K., Höflich, C. et al. Interleukin-10 receptor-1 expression in monocyte-derived antigen-presenting cell populations: dendritic cells partially escape from IL-10's inhibitory mechanisms. Genes Immun 16, 8–14 (2015). https://doi.org/10.1038/gene.2014.69
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gene.2014.69
This article is cited by
-
Tissue-based IL-10 signalling in helminth infection limits IFNγ expression and promotes the intestinal Th2 response
Mucosal Immunology (2022)
-
Lactobacillus delivery of bioactive interleukin-22
Microbial Cell Factories (2017)