Abstract
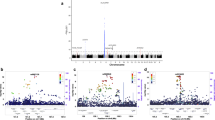
Multiple sclerosis (MS) is an autoimmune demyelinating disease characterized by complex genetics and multifaceted gene–environment interactions. Compared to whites, African Americans have a lower risk for developing MS, but African Americans with MS have a greater risk of disability. These differences between African Americans and whites may represent differences in genetic susceptibility and/or environmental factors. SNPs from 12 candidate genes have recently been identified and validated with MS risk in white populations. We performed a replication study using 918 cases and 656 unrelated controls to test whether these candidate genes are also associated with MS risk in African Americans. CD6, CLEC16a, EVI5, GPC5, and TYK2 contained SNPs that are associated with MS risk in the African American data set. EVI5 showed the strongest association outside the major histocompatibility complex (rs10735781, OR=1.233, 95% CI=1.06–1.43, P-value=0.006). In addition, RGS1 seems to affect age of onset whereas TNFRSF1A seems to be associated with disease progression. None of the tested variants showed results that were statistically inconsistent with the effects established in whites. The results are consistent with shared disease genetic mechanisms among individuals of European and African ancestry.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 digital issues and online access to articles
$119.00 per year
only $19.83 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Hauser SLaG DS . Multiple sclerosis and other demyelinating diseases. In: Kasper DL, Braunwald E, Fauci AD, Hauser SL, Longo DL, Jameson JL (eds). Harrsion's Principle of Internal Medicine, 16th edn. McGraw-Hill: New York, 2005. pp 2461–2471.
Orton SM, Herrera BM, Yee IM, Valdar W, Ramagopalan SV, Sadovnick AD et al. Sex ratio of multiple sclerosis in Canada: a longitudinal study. Lancet Neurol 2006; 5: 932–936.
Rosati G . The prevalence of multiple sclerosis in the world: an update. Neurol Sci 2001; 22: 117–139.
Wallin MT, Page WF, Kurtzke JF . Multiple sclerosis in US veterans of the Vietnam era and later military service: race, sex, and geography. Ann Neurol 2004; 55: 65–71.
Buchanan RJ, Martin RA, Wang S, Kim M . Racial analyses of longer-stay nursing home residents with multiple sclerosis. Ethn Dis 2006; 16: 159–165.
Buchanan RJ, Martin RA, Zuniga M, Wang S, Kim M . Nursing home residents with multiple sclerosis: comparisons of African American residents to white residents at admission. Mult Scler 2004; 10: 660–667.
Cree BA, Khan O, Bourdette D, Goodin DS, Cohen JA, Marrie RA et al. Clinical characteristics of African Americans vs Caucasian Americans with multiple sclerosis. Neurology 2004; 63: 2039–2045.
Marrie RA, Cutter G, Tyry T, Vollmer T, Campagnolo D . Does multiple sclerosis-associated disability differ between races? Neurology 2006; 66: 1235–1240.
Naismith RT, Trinkaus K, Cross AH . Phenotype and prognosis in African-Americans with multiple sclerosis: a retrospective chart review. Mult Scler 2006; 12: 775–781.
Weinstock-Guttman B, Jacobs LD, Brownscheidle CM, Baier M, Rea DF, Apatoff BR et al. Multiple sclerosis characteristics in African American patients in the New York State Multiple Sclerosis Consortium. Mult Scler 2003; 9: 293–298.
Oksenberg JR, Baranzini SE, Sawcer S, Hauser SL . The genetics of multiple sclerosis: SNPs to pathways to pathogenesis. Nat Rev 2008; 9: 516–526.
Wellcome Trust Case Control Consortium, Burton PR, Clayton DG, Cardon LR, Craddock N, Deloukas P, Duncanson A et al. Association scan of 14 500 nonsynonymous SNPs in four diseases identifies autoimmunity variants. Nat Genet 2007; 39: 1329–1337.
International Multiple Sclerosis Genetics Consortium, Hafler DA, Compston A, Sawcer S, Lander ES, Daly MJ, De Jager PL et al. Risk alleles for multiple sclerosis identified by a genomewide study. N Engl J Med 2007; 357: 851–862.
Baranzini SE, Wang J, Gibson RA, Galwey N, Naegelin Y, Barkhof F et al. Genome-wide association analysis of susceptibility and clinical phenotype in multiple sclerosis. Hum Mol Genet 2009; 18: 767–778.
De Jager PL, Jia X, Wang J, de Bakker PI, Ottoboni L, Aggarwal NT et al. Meta-analysis of genome scans and replication identify CD6, IRF8 and TNFRSF1A as new multiple sclerosis susceptibility loci. Nat Genet 2009; 41: 776–782.
Caillier SJ, Briggs F, Cree BA, Baranzini SE, Fernandez-Vina M, Ramsay PP et al. Uncoupling the roles of HLA-DRB1 and HLA-DRB5 genes in multiple sclerosis. J Immunol 2008; 181: 5473–5480.
Oksenberg JR, Barcellos LF, Cree BA, Baranzini SE, Bugawan TL, Khan O et al. Mapping multiple sclerosis susceptibility to the HLA-DR locus in African Americans. Am J Hum Genet 2004; 74: 160–167.
Cree BA, Reich DE, Khan O, De Jager PL, Nakashima I, Takahashi T et al. Modification of multiple sclerosis phenotypes by African ancestry at HLA. Arch Neurol 2009; 66: 226–233.
Ban M, Goris A, Lorentzen AR, Baker A, Mihalova T, Ingram G et al. Replication analysis identifies TYK2 as a multiple sclerosis susceptibility factor. Eur J Hum Genet 2009; 17: 1309–1313.
Hoppenbrouwers IA, Aulchenko YS, Ebers GC, Ramagopalan SV, Oostra BA, van Duijn CM et al. EVI5 is a risk gene for multiple sclerosis. Genes Immun 2008; 9: 334–337.
Australia and New Zealand Multiple Sclerosis Genetics Consortium, Bahlo M, Booth DR, Broadley SA, Brown MA, Foote SJ, Griffiths LR et al. Genome-wide association study identifies new multiple sclerosis susceptibility loci on chromosomes 12 and 20. Nat Genet 2009; 41: 824–828.
Liao X, Buchberg AM, Jenkins NA, Copeland NG . Evi-5, a common site of retroviral integration in AKXD T-cell lymphomas, maps near Gfi-1 on mouse chromosome 5. J Virol 1995; 69: 7132–7137.
Faitar SL, Sossey-Alaoui K, Ranalli TA, Cowell JK . EVI5 protein associates with the INCENP-aurora B kinase-survivin chromosomal passenger complex and is involved in the completion of cytokinesis. Exp Cell Res 2006; 312: 2325–2335.
Dabbeekeh JT, Faitar SL, Dufresne CP, Cowell JK . The EVI5 TBC domain provides the GTPase-activating protein motif for RAB11. Oncogene 2007; 26: 2804–2808.
Westlake CJ, Junutula JR, Simon GC, Pilli M, Prekeris R, Scheller RH et al. Identification of Rab11 as a small GTPase binding protein for the Evi5 oncogene. Proc Natl Acad Sci USA 2007; 104: 1236–1241.
Ullrich O, Reinsch S, Urbe S, Zerial M, Parton RG . Rab11 regulates recycling through the pericentriolar recycling endosome. J Cell Biol 1996; 135: 913–924.
Green EG, Ramm E, Riley NM, Spiro DJ, Goldenring JR, Wessling-Resnick M . Rab11 is associated with transferrin-containing recycling compartments in K562 cells. Biochem Biophys Res Commun 1997; 239: 612–616.
Wang X, Kumar R, Navarre J, Casanova JE, Goldenring JR . Regulation of vesicle trafficking in madin-darby canine kidney cells by Rab11a and Rab25. J Biol Chem 2000; 275: 29138–29146.
Fan GH, Lapierre LA, Goldenring JR, Sai J, Richmond A . Rab11-family interacting protein 2 and myosin Vb are required for CXCR2 recycling and receptor-mediated chemotaxis. Mol Biol Cell 2004; 15: 2456–2469.
Fielding AB, Schonteich E, Matheson J, Wilson G, Yu X, Hickson GR et al. Rab11-FIP3 and FIP4 interact with Arf6 and the exocyst to control membrane traffic in cytokinesis. EMBO J 2005; 24: 3389–3399.
Wilson GM, Fielding AB, Simon GC, Yu X, Andrews PD, Hames RS et al. The FIP3-Rab11 protein complex regulates recycling endosome targeting to the cleavage furrow during late cytokinesis. Mol Biol Cell 2005; 16: 849–860.
Saita S, Shirane M, Natume T, Iemura S, Nakayama KI . Promotion of neurite extension by protrudin requires its interaction with vesicle-associated membrane protein-associated protein. J Biol Chem 2009; 284: 13766–13777.
Gorska MM, Liang Q, Karim Z, Alam R . Uncoordinated 119 protein controls trafficking of Lck via the Rab11 endosome and is critical for immunological synapse formation. J Immunol 2009; 183: 1675–1684.
Molina TJ, Kishihara K, Siderovski DP, van Ewijk W, Narendran A, Timms E et al. Profound block in thymocyte development in mice lacking p56lck. Nature 1992; 357: 161–164.
Straus DB, Weiss A . Genetic evidence for the involvement of the lck tyrosine kinase in signal transduction through the T cell antigen receptor. Cell 1992; 70: 585–593.
Hassan NJ, Simmonds SJ, Clarkson NG, Hanrahan S, Puklavec MJ, Bomb M et al. CD6 regulates T-cell responses through activation-dependent recruitment of the positive regulator SLP-76. Mol Cell Biol 2006; 26: 6727–6738.
International Multiple Sclerosis Genetics Consortium (IMSGC). The expanding genetic overlap between multiple sclerosis and type I diabetes. Genes Immun 2009; 10: 11–14.
Zoledziewska M, Costa G, Pitzalis M, Cocco E, Melis C, Moi L et al. Variation within the CLEC16A gene shows consistent disease association with both multiple sclerosis and type 1 diabetes in Sardinia. Genes Immun 2009; 10: 15–17.
Awata T, Kawasaki E, Tanaka S, Ikegami H, Maruyama T, Shimada A et al. Association of type 1 diabetes with two Loci on 12q13 and 16p13 and the influence coexisting thyroid autoimmunity in Japanese. J Clin Endocrinol Metab 2009; 94: 231–235.
Marquez A, Varade J, Robledo G, Martinez A, Mendoza JL, Taxonera C et al. Specific association of a CLEC16A/KIAA0350 polymorphism with NOD2/CARD15(-) Crohn's disease patients. Eur J Hum Genet 2009; 17: 1304–1308.
Robinson MJ, Sancho D, Slack EC, LeibundGut-Landmann S, Reis e Sousa C . Myeloid C-type lectins in innate immunity. Nat Immunol 2006; 7: 1258–1265.
Veugelers M, Vermeesch J, Reekmans G, Steinfeld R, Marynen P, David G . Characterization of glypican-5 and chromosomal localization of human GPC5, a new member of the glypican gene family. Genomics 1997; 40: 24–30.
Lee JS, Chien CB . When sugars guide axons: insights from heparan sulphate proteoglycan mutants. Nat Rev 2004; 5: 923–935.
Van Vactor D, Wall DP, Johnson KG . Heparan sulfate proteoglycans and the emergence of neuronal connectivity. Curr Opin Neurobiol 2006; 16: 40–51.
van Horssen J, Bo L, Dijkstra CD, de Vries HE . Extensive extracellular matrix depositions in active multiple sclerosis lesions. Neurobiol Dis 2006; 24: 484–491.
Chernousov MA, Rothblum K, Stahl RC, Evans A, Prentiss L, Carey DJ . Glypican-1 and alpha4(V) collagen are required for Schwann cell myelination. J Neurosci 2006; 26: 508–517.
Worapamorn W, Haase HR, Li H, Bartold PM . Growth factors and cytokines modulate gene expression of cell-surface proteoglycans in human periodontal ligament cells. J Cell Physiol 2001; 186: 448–456.
Han SB, Moratz C, Huang NN, Kelsall B, Cho H, Shi CS et al. Rgs1 and Gnai2 regulate the entrance of B lymphocytes into lymph nodes and B cell motility within lymph node follicles. Immunity 2005; 22: 343–354.
Hunt KA, Zhernakova A, Turner G, Heap GA, Franke L, Bruinenberg M et al. Newly identified genetic risk variants for celiac disease related to the immune response. Nat Genet 2008; 40: 395–402.
Romanos J, Barisani D, Trynka G, Zhernakova A, Bardella MT, Wijmenga C . Six new coeliac disease loci replicated in an Italian population confirm association with coeliac disease. J Med Genet 2009; 46: 60–63.
Smyth DJ, Plagnol V, Walker NM, Cooper JD, Downes K, Yang JH et al. Shared and distinct genetic variants in type 1 diabetes and celiac disease. N Engl J Med 2008; 359: 2767–2777.
Probert L, Eugster HP, Akassoglou K, Bauer J, Frei K, Lassmann H et al. TNFR1 signalling is critical for the development of demyelination and the limitation of T-cell responses during immune-mediated CNS disease. Brain 2000; 123 (Pt 10): 2005–2019.
Kassiotis G, Kollias G . Uncoupling the proinflammatory from the immunosuppressive properties of tumor necrosis factor (TNF) at the p55 TNF receptor level: implications for pathogenesis and therapy of autoimmune demyelination. J Exp Med 2001; 193: 427–434.
TNF neutralization in MS: results of a randomized, placebo-controlled multicenter study. The Lenercept multiple sclerosis study group and the university of British Columbia MS/MRI analysis group. Neurology 1999; 53: 457–465.
Fromont A, De Seze J, Fleury MC, Maillefert JF, Moreau T . Inflammatory demyelinating events following treatment with anti-tumor necrosis factor. Cytokine 2009; 45: 55–57.
Mohan N, Edwards ET, Cupps TR, Oliverio PJ, Sandberg G, Crayton H et al. Demyelination occurring during anti-tumor necrosis factor alpha therapy for inflammatory arthritis. Arthritis Rheum 2001; 44: 2862–2869.
Sicotte NL, Voskuhl RR . Onset of multiple sclerosis associated with anti-TNF therapy. Neurology 2001; 57: 1885–1888.
Patterson N, Hattangadi N, Lane B, Lohmueller KE, Hafler DA, Oksenberg JR et al. Methods for high-density admixture mapping of disease genes. Am J Hum Genet 2004; 74: 979–1000.
McDonald WI, Compston A, Edan G, Goodkin D, Hartung HP, Lublin FD et al. Recommended diagnostic criteria for multiple sclerosis: guidelines from the International Panel on the diagnosis of multiple sclerosis. Ann Neurol 2001; 50: 121–127.
Wingerchuk DM, Lennon VA, Pittock SJ, Lucchinetti CF, Weinshenker BG . Revised diagnostic criteria for neuromyelitis optica. Neurology 2006; 66: 1485–1489.
Sole X, Guino E, Valls J, Iniesta R, Moreno V . SNPStats: a web tool for the analysis of association studies. Bioinformatics 2006; 22: 1928–1929.
Blom G . Statistical Estimates and Transformed Beta-Variables. Wiley: New York, 1958.
Acknowledgements
We thank the MS patients and controls that participated in this study. We thank L Piccio for her effort in the recruitment of study participants and sample management. We thank RR Lincoln, R Guerrero, H Mousavi, and A Santaniello for sample and database management. We thank R Gomez for recruitment of participants. We also thank J McElroy for insightful discussions. This work was funded by grants from the National Institute of Health (R01 NS046297) and the National Multiple Sclerosis Society (RG3060C8). PLD is a Harry Weaver Neuroscience Scholar Award of the National MS Society.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Genes and Immunity website
Supplementary information
Rights and permissions
About this article
Cite this article
Johnson, B., Wang, J., Taylor, E. et al. Multiple sclerosis susceptibility alleles in African Americans. Genes Immun 11, 343–350 (2010). https://doi.org/10.1038/gene.2009.81
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gene.2009.81
Keywords
This article is cited by
-
Towards a global view of multiple sclerosis genetics
Nature Reviews Neurology (2022)
-
Prediction of combination therapies based on topological modeling of the immune signaling network in multiple sclerosis
Genome Medicine (2021)
-
Association of CD58 polymorphism and multiple sclerosis in Malaysia: a pilot study
Autoimmunity Highlights (2019)
-
Analysis of Associations of Polymorphisms of Genes Encoding Cytokine Receptors with the Clinical Features of Multiple Sclerosis
Neuroscience and Behavioral Physiology (2018)
-
RGS10 deficiency ameliorates the severity of disease in experimental autoimmune encephalomyelitis
Journal of Neuroinflammation (2016)