Abstract
Purpose
To investigate the recovery of photoreceptors following the treatment in Vogt–Koyanagi–Harada (VKH) disease.
Patients and methods
This was a retrospective study. We enrolled 28 patients with VKH (56 eyes). The clinical and optical coherence tomography (OCT) findings were recorded for 12 months after treatment. The patterns of photoreceptor recovery on OCT were defined: pattern F group=Foveal photoreceptor recovery visible first; pattern E group=Extrafoveal photoreceptor recovery visible first; and pattern S group=Simultaneous foveal and extrafoveal photoreceptor recovery.
Results
Photoreceptor recovery varied in different parts of the fundus among patients. Among the 56 eyes, the ellipsoid zone (EZ) recovery of 10 eyes and the interdigitation zone (IZ) recovery of 17 eyes belonged to pattern F group. In most eyes (46 eyes for EZ and 26 eyes for IZ), the recovery of these structures were pattern S. Only in 10 eyes, the recovery of IZ was pattern E. The different patterns of recovery correlated with how promptly the patients had been treated and with the anatomical and visual outcomes at 12 months. Patients in pattern F group were characterized by delayed treatment, delayed recovery of EZ or IZ, and a less favourable prognosis at 12 months relative to other patients, while those in pattern E group had the most prompt treatment and recovery as well as a more favourable outcome at 12 months.
Conclusions
In VKH patients with delayed treatment, foveal photoreceptors tended to recover more rapidly than photoreceptors in other regions.
Similar content being viewed by others
Introduction
Vogt–Koyanagi–Harada (VKH) disease is a granulomatous inflammatory disorder that affects the eyes, auditory system, meninges, and skin. It accounts for about 6.7–16% of all cases of uveitis in East Asia.1, 2 Spectral-domain optical coherence tomography (SD-OCT) studies have revealed morphological changes, including choroidal thickening, undulation of the retinal pigment epithelium (RPE), subretinal fluid accumulation, and blurring of the photoreceptor ellipsoid zone (EZ), in the eyes of patients with acute VKH disease.3, 4 The manifestations of late-stage VKH disease that are detected with OCT include RPE atrophy, loss of EZ, loss of the external limiting membrane, loss of the outer nuclear layer, and choroidal thinning.5, 6 However, little is known about the changes that occur between the acute and late stage. We previously reported that some late-stage VKH eyes had a fragmented EZ or interdigitation zone (IZ), but these structures were intact in the central fovea.7 Therefore, we performed this study to evaluate the recovery of photoreceptors in patients with acute VKH disease, and to explore whether the photoreceptors in different parts of the fundus recovered differently.
Methods
Study participants
We reviewed the medical records of patients diagnosed with VKH disease and who received standard corticosteroid treatment at the Department of Ophthalmology, Eye and ENT Hospital, Fudan University, between January 2013 and April 2015. VKH disease was diagnosed according to the revised diagnostic criteria proposed by the International Nomenclature Committee.3 The standard corticosteroid therapy defined as starting with steroid pulse (intravenous drip infusion of 1 g methylprednisolone/day for three consecutive days) followed by a 1 mg/kg daily dose of oral prednisolone. The dose of oral prednisolone was gradually tapered over more than 6 months. Beginning with low-dose of oral prednisolone is defined as non-standard treatment. If there was a relapse during the tapering of oral prednisolone, the dosage was increased or adjuvant cyclosporine was administered as needed at a dose of 3 mg/kg/day; and then the dose was tapered according to the severity of the ocular inflammation. The study conformed to the tenets of the Declaration of Helsinki, and was approved by the Institutional Review Board of the Eye and ENT Hospital of Fudan University. All of the subjects signed an informed consent form.
Data collection
Data were recorded at visits before treatment and at 1, 3, 6, and 12 months after treatment. Each visit involved a comprehensive ophthalmological examination, including the assessment of the best corrected visual acuity (BCVA), assessment of retinal sensitivity with a microperimeter (except at the first visit), measurement of intraocular pressure, slit-lamp biomicroscopy, fundoscopic examination with a noncontact lens, and SD-OCT.
Microperimetry
After pupil dilation, all patients were tested with microperimetry using an MP-1 microperimeter (software version SW1.4.1 SP1; Nidek Technologies, Padua, Italy). The settings included a Goldmann III-sized target and the 4−2 staircase strategy, with 45 stimulated locations within a diameter of 12°.7 Microperimetry was performed by an experienced ophthalmologist (ZS) who was masked to the patient’s medical history. The software automatically calculated the retinal sensitivity, which was defined as the mean of all 45 scores. The retina was then divided into two regions: the central foveal area (13 central spots covering 0–4°) and the peripheral area (the remaining 32 spots in an annulus of 5–12°) (Supplementary Figure S1).
Spectral-domain optical coherence tomography
After the pupil was fully dilated, EDI-OCT images were captured with a Spectralis HRA+OCT version 1.5.12.0 (Heidelberg Engineering, Heidelberg, Germany) in the volume scan mode, which covered a 30° × 30° region centred on the fovea (1024 × 25; 25 lines). Only scans with good signal strength (signal strength ≥20) were used for the analysis. The OCT images were taken before treatment and at 1, 3, 6 and 12 months after treatment. The follow-up function on OCT was used to evaluate the same points. A horizontal scan line crossing the centre of the fovea was selected for further analysis. The OCT images were graded using a standardized protocol by an experienced ophthalmologist who was masked to the patient’s characteristics and the date of follow-up. To determine whether the photoreceptors within and outside the fovea recovered differently, EZ and IZ within and outside the fovea were graded separately. The patterns of photoreceptor recovery on OCT were defined like this: pattern F group=Foveal photoreceptor recovery visible first; pattern E group=Extrafoveal photoreceptor recovery visible first; and pattern S group=Simultaneous foveal and extrafoveal photoreceptor recovery. The fovea was defined as the central 1.5 mm region of the macular on each cross-section scan. OCT grading at the 1-, 3-, and 6-month visits recorded whether the photoreceptors were visible. Using images obtained at 12 months, the photoreceptors were graded as intact or not (Supplementary Figure S2).
Statistical analysis
The BCVA was converted to logarithms of the minimum angle of resolution (logMAR) for statistical analysis. The linear mixed model was performed to compare the differences in retinal sensitivity, BCVA and the recovery of out retinal structures. In all analyses, values of P<0.05 were considered statistically significant. All analyses were performed with SPSS for Windows version 17.0 (SPSS, Inc., Chicago, IL, USA).
Results
A total of 28 patients with VKH disease (11 males, 17 females; 56 eyes) attended all follow-up visits at 1, 3, 6, and 12 months, and were included in the study. At the first visit, the mean age of the patients was 34.14±12.49 years, the mean BCVA (logMAR) was 0.84±0.52 (Supplementary Table S1). Ten patients (20 eyes) had previously received non-standard corticosteroid therapy, and the mean time from the onset of symptoms to treatment (the standard treatment starting at our hospital) was 18.96±16.39 days.
After treatment, the BCVA and mean retinal sensitivity improved markedly (Supplementary Figure S3). At 12 months, the mean BCVA (logMAR) was 0.07±0.09 and the microperimetric sensitivity was 18.46±1.79 dB. Retinal detachment resolved quickly. At 1 month after treatment, 11 (19.64%) eyes had evidence of retinal detachment on OCT image, and in nine of them, the retinal detachment was localized to the central macular region (Supplementary Figure S4a). All the retinal detachments had resolved by 3 months. The EZ and IZ were gradually reappeared after treatment. At 1 month, 76.78 of EZ and 48.21% of IZ had reappeared on OCT images. The ratio increased to 94.64 for EZ and 71.42% for IZ by 3 months. At 12 months, all eyes (100%) showed visible EZ and 47(83.93%) eyes showed visible IZ. The IZ of nine eyes (16.07%) had not recovered at the end of 12 months.
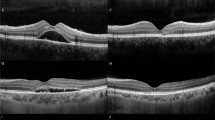
Although EZ and IZ became visible after treatment, there were different patterns in their recovery. Among the 56 eyes, the EZ recovery of 10 eyes (Figure 1a) and the IZ recovery of 17 eyes (Figure 2a) belonged to pattern F group. In most eyes (46 eyes for EZ and 26 eyes for IZ), the recovery of these structures were pattern S (Figures 1b and 2b). Only in 10 eyes, the recovery of IZ was pattern E (Figure 2c). And no case of EZ recovery was pattern E.
Different patterns of recovery in the interdigitation zone (IZ). Representative images showing (a) pattern F for IZ: Foveal IZ recovery visible first, (b) pattern S for IZ: Simultaneous foveal and extrafoveal IZ recovery, and (c) pattern E for IZ: Extrafoveal IZ recovery visible first. Lines indicate the macular region (1.5 mm).The arrow as IZ and asterisk as EZ.
The age and sex of patients in pattern F group were similar to those in pattern S or pattern E group. However, the eyes in pattern F group were associated with worse initial vision, delayed treatment, delayed recovery of EZ or IZ, and worse anatomical and visual outcomes at 12 months (Tables 1 and 2). Eyes in pattern S group had better initial vision, received prompt treatment, showed faster recovery of EZ or IZ, and had better anatomical and visual outcomes at 12 months (Tables 1 and 2). The 10 eyes in pattern E group were treated most rapidly, with a mean of 9.8 days after symptom onset (Table 2). The differences in clinical variables among the groups of eyes were still significant when patients who had previously received non-standard treatment were excluded from the analysis (Table 3). Compared with patients in pattern S group, patients in pattern F group were characterized by delayed treatment, delayed recovery of IZ, and worse retinal sensitivity at 12 months after treatment (Table 3).
Discussion
Many studies had proved that the reappearance of EZ or IZ could be considered as the site of photoreceptor recovery in many retinal diseases.8, 9, 10 In this study, we describe the recovery of photoreceptors, determined by the reappearance of EZ or IZ on SD-OCT, after standard corticosteroid treatment in patients with acute VKH disease. The patterns of photoreceptor recovery varied among patients and in different parts of the posterior pole. In most eyes, these structures reappeared simultaneously within and outside the fovea, while in some eyes, the EZ and IZ reappeared firstly within the fovea and in some firstly outside the fovea. These different patterns of recovery were associated with how promptly the patients had been treated and with the visual or anatomical prognosis at 12 months. As well as delayed treatment, preoperative pathology such as the presence of choroidal undulation and subretinal fluid were linked to a delay in recovery of the outer retina. Compared with the eyes with choroidal undulations before treatment, the ones without choroidal undulations showed faster recovery of EZ/IZ, and had better retinal function at 12 months (Supplementary Table S2). And also the ones without subretinal fluid before treatment had better initial vision, showed faster recovery of IZ, and had better anatomical at 12 months than eyes with subretinal fluid (Supplementary Table S3).
In our result, in 9 of the 11 eyes with retinal detachment at 1 month, the detachment was localized only at the central macular region, and in the other two beside the localized retinal detachment at the central macula, there was only slight subretinal fluid around the disc (Supplementary Figure S4). Patients with acute VKH disease commonly display macular retinal detachment. However, it is unclear whether this retinal detachment originates from the macular region. Our data at least indicated that retinal detachment in the central macular region was the last to resolve. The 10 eyes in which the photoreceptors in IZ recovered first outside the fovea had the shortest time from the onset of symptoms to treatment (mean 9.8 days), and a quick recovery (the mean time from treatment to the finding of IZ 1±1 month) (Supplementary Figure S5). The reason might be as follows. In these patients, because of prompt treatment, the retinal detachment might exist for a shorter period of time, and as the retinal detachment outside the fovea resolved first, the detachment outside the fovea lasted for even shorter period of time. So in these eyes the photoreceptors outside the fovea might have been mildly impaired, thus allowing fast recovery.
In most eyes, EZ or IZ (46/56 for EZ and 26/56 for IZ) were found simultaneously at and outside the fovea, and in some eyes they (10 for EZ and 17 for IZ) were found firstly at the fovea, despite the fact that the retinal detachment at the fovea resolved last. This might result from the high density of cones in the fovea,11 which made it easier to detect their reappearance or recovery. Also the foveal RPE is taller and contains more melanosomes than the RPE in other regions.11
What is more interesting, analysis found that eyes with photoreceptors found first at the fovea seemed to have a significantly delayed treatment, and this difference was still significant when we excluded patients who had previously received non-standard treatment (Table 3). And in nine eyes with localized foveal detachment at 1 month, the EZ in four eyes and IZ in five eyes recovered first within the fovea (Supplementary Figure S4b,c). Also in our former study of late stage VKH eyes, we found that in eyes with EZ or IZ defects outside the fovea, these structures could remain intact at the fovea, but in eyes with EZ or IZ defects at the fovea there were always defects in these structures outside the fovea.7 These might suggest that at the fovea, beside the high density of the photoreceptor and the taller RPE, the photoreceptors there might be either resistant to VKH-related inflammation or might recover much better than photoreceptors outside the fovea. One more thing, formerly Curcio reported that the photoreceptor density of human was highly variable between individuals,12 such variation might also exist in the structure or density of RPE as well. These foveal structures might also be relevant to the pattern of recovery of outer retinal structure.
Patients whose EZ or IZ outside the fovea recovered last were characterized by delayed treatment and worse anatomic and visual outcomes at 12 months. This association between delayed treatment and worse outcomes strengthens the importance of prompt standardized treatment for VKH. And at the end of 12-month follow-up, the BCVA and retinal sensitivity were comparable to healthy subjects (BCVA logMAR 0.07±0.09 and retinal sensitivity 18.46±1.79 dB).13 So with standard treatment, VKH patients could achieve a rather good visual and structural outcome.
The exact reason for the comparatively rapid and good recovery of photoreceptors in the fovea in patients with a delayed treatment is unclear. Therefore, further studies are required to understand the reason for this. Such studies might reveal new ways to improve the prognosis of VKH and other retinal diseases, especially macular diseases.
In conclusion, OCT findings over 12 months after treatment revealed that the recovery of photoreceptor differed among the patients, and different patterns were associated with how prompt the treatment was. And in patients for whom treatment was delayed, the photoreceptors at the fovea seemed to recover more rapidly and better than those in the peripheral regions of eyes.

References
Yang P, Ren Y, Li B, Fang W, Meng Q, Kijlstra A . Clinical characteristics of Vogt–Koyanagi–Harada syndrome in Chinese patients. Ophthalmology 2007; 114 (3): 606–614.
Goto H, Mochizuki M, Yamaki K, Kotake S, Usui M, Ohno S . Epidemiological survey of intraocular inflammation in Japan. Jpn J Ophthalmol 2007; 51 (1): 41–44.
Read RW, Holland GN, Rao NA, Tabbara KF, Ohno S, Arellanes-Garcia L et al. Revised diagnostic criteria for Vogt–Koyanagi–Harada disease: report of an international committee on nomenclature. Am J Ophthalmol 2001; 131 (5): 647–652.
Tsuboi K, Nakai K, Iwahashi C, Gomi F, Ikuno Y, Nishida K . Analysis of choroidal folds in acute Vogt–Koyanagi–Harada disease using high-penetration optical coherence tomography. Graefes Arch Clin Exp Ophthalmol 2015; 253 (6): 959–964.
Lee EK, Lee SY, Yu HG . A clinical grading system based on ultra-wide field retinal imaging for sunset glow fundus in Vogt–Koyanagi–Harada disease. Graefes Arch Clin Exp Ophthalmol 2015; 253: 359–368.
Takahashi H, Takase H, Ishizuka A, Miyanaga M, Kawaguchi T, Ohno-Matsui K et al. Choroidal thickness in convalescent vogt–koyanagi–harada disease. Retina 2014; 34 (4): 775–780.
Zhou M, Jiang C, Gu R, Sun Z, Huynh N, Chang Q . Correlation between retinal changes and visual function in late-stage Vogt–Koyanagi–Harada disease: an optical coherence tomography study. J Ophthalmol 2015; 2015: 916485.
Itoh Y, Inoue M, Rii T, Hiraoka T, Hirakata A . Significant correlation between visual acuity and recovery of foveal cone microstructures after macular hole surgery. Am J Ophthalmol 2012; 153 (1): 111–119.
Hirota K, Itoh Y, Rii T, Inoue M, Hirakata A . Correlation between foveal interdigitation zone band defect and visual acuity after surgery for macular pseudohole. Retina 2015; 35 (5): 908–914.
Shimozono M, Oishi A, Hata M, Matsuki T, Ito S, Ishida K et al. The significance of cone outer segment tips as a prognostic factor in epiretinal membrane surgery. Am J Ophthalmol 2012; 153 (4): 698–704, 701–704.
Spaide RF, Curcio CA . Anatomical correlates to the bands seen in the outer retina by optical coherence tomography: literature review and model. Retina 2011; 31 (8): 1609–1619.
Curcio CA, Sloan KR, Kalina RE, Hendrickson AE . Human photoreceptor topography. J Comp Neurol 1990; 22 (4): 497–523.
Springer C, Volcker H E, Rohrschneider K . Static fundus perimetry in normals. Microperimeter 1 versus SLO. Ophthalmologe 2006; 3 (3): 214–220.
Acknowledgements
This study was supported in part by research grants from the National Natural Science Foundation of China (81570854), the National Key Basic Research Program of China (2013CB967503), the National Major Scientific Equipment Program (grant no. 2012YQ12008003), and the National Key Basic Research Program of China (grant no. 2013CB967503).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on Eye website
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Zhou, M., Gu, R., Sun, Z. et al. Differences in photoreceptor recovery among patients and between different parts of the posterior pole in Vogt–Koyanagi–Harada disease. Eye 32, 572–578 (2018). https://doi.org/10.1038/eye.2017.250
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/eye.2017.250
This article is cited by
-
Fingerprint sign in Vogt-Koyanagi-Harada disease: a case series
International Journal of Retina and Vitreous (2022)