Abstract
Purpose
The purpose of this study was to introduce nurse-delivered intravitreal injections to increase medical retina treatment capacity.
Methods
Indemnity, clinical governance, training, planning, and implementation issues were addressed. The outcome measures were patient safety, patient experience, and clinic capacity.
Results
No serious vision-threatening complications were recorded in a consecutive series of 4000 nurse-delivered intravitreal injections. A Mann–Whitney test showed a significant increase in intravitreal injections (P=0.003) in the medical retina service after introduction of nurse-delivered intravitreal injections. The majority of patients accepted and were satisfied with a nurse-delivered intravitreal injection.
Discussion
Nurse-delivered intravitreal injections appear safe, acceptable to patients, and are an effective means to increase intravitreal injection capacity in medical retina clinics.
Similar content being viewed by others
Introduction
Anti-vascular endothelial growth factor (Anti-VEGF) treatment has revolutionised the management of neovascular age-related macular degeneration (AMD). The majority of patients attend for an intensive follow-up and injection regime at six weekly intervals in alignment with current NICE guidelines.1
Intravitreal ranibizumab is currently licensed only for administration by an ophthalmologist. Expansion of intravitreal injection services is necessary for conditions such as diabetic macular oedema and retinal vein occlusions.
Recent reforms to the UK medical training system have reduced the number of ophthalmologists in training. An ageing population, economic constraints, and the introduction of clinical commissioning have created an incentive to explore new methods of multidisciplinary working.
Traditional nursing work has changed considerably in recent years with the introduction of extended roles in many surgical specialties. There is a growing awareness of the positive impact of extended roles both for health professionals and for patients.
Purpose
The purpose of this study was to introduce nurse practitioner-delivered intravitreal injections to provide increased treatment capacity within the medical retina service.
Materials and methods
The following issues were addressed:
1) Indemnity
The Trust sought legal advice related to vicarious liability and confirmation of indemnity cover from the National Health Service Litigation Authority (NHSLA). A separate consent form and patient information leaflet were formulated for the nurse-delivered intravitreal injections. This was necessary in view of the current licensing of ranibizumab for administration by an ophthalmologist.
2) Clinical governance
A comprehensive policy and procedure document was produced including a stipulation that a continuous audit of each nurse practitioner’s complications would be recorded. It was agreed that consultant ophthalmologists would retain clinical responsibility for the patient. Nurse-delivered injections would be initially supervised, and then run independently but parallel to an ophthalmologist-delivered list so that a doctor is always available for advice. Approval from the Trust’s clinical audit, clinical governance, and management executive committees were obtained.
3) Training
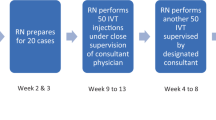
A rigorous training schedule was developed for nurse practitioners before undertaking this procedure (Figure 1). This included attendance at a full day intravitreal injection course including practical training on pig eyes in a wetlab.
Wetlab training is an established method of surgical training to allow staff to fully understand practical procedures using biological materials such as animal tissue. Each nurse training to perform intravitreal injections was provided with a disposable work station consisting of an instrument pack, gloves, apron, and a pig eye obtained from a UK supplier of fresh tissues. The procedure was then demonstrated to nurses and they performed the procedure themselves on the pig eye. This was valuable to practise identifying the injection site, the angle of needle insertion, and the speed of injection.
This was then followed by observation of the procedure on a patient and graded exposure under the supervision of a consultant trainer. At the end of the training period of 100 supervised procedures, nurses underwent competency assessment. Patients were fully informed and consented before a nurse performed the procedure. Regular teaching and professional development sessions were organised to support nurses undertaking this extended role.
4) Planning and implementation
Most jobs in the NHS are covered by the Agenda for Change payscales. This covers all staff except doctors, dentists, and most senior managers. The Agenda for Change job evaluation system determines a points score (http://www.nhscareers.nhs.uk/working-in-the-nhs/pay-and-benefits/agenda-for-change-pay-rates/), which is used to match jobs to one of the nine pay bands and determine levels of basic salary. A career in nursing may start at band 2 (clinical support worker) rising to band 8 (nurse consultant). A fully qualified nurse would start their career at band 5. After consultation with senior nurses and nurse managers, it was felt appropriate that senior nurses at band 7 perform this extended role. There then followed consultation with the Human Resources department on remuneration and banding for nurse intravitreal injection sessions. A review of timetables across different sites, and arrangements for additional cover to allow senior nurses to train, was organised. A business case was completed for additional funding. Planning incorporated provision for nurse injectors at the Trust’s site in central London and also in several satellite hospitals. A named consultant trainer was allocated for each nurse trainer. A pilot initiative was instigated, and after successful completion an expansion of this initiative was commenced. It was agreed that intravitreal bevacizumab would not be administered by nurses, as this is an unlicensed treatment for neovascular AMD in the UK.
Outcome measures
1) Safety
Each nurse practitioner recorded details of procedures performed, as well as complications.
2) Patient experience
The numbers of patients who declined nurse-delivered intravitreal injections and the reasons for this were recorded. A modified externally validated patient questionnaire2 was formulated (Figure 2). In this questionnaire, seven elements of patient satisfaction were examined: general satisfaction, technical quality, interpersonal manner, staff attitude, information/communication, time spent with the doctor, and staff competence.
3) Number of injections before and after initiation of the nurse injector programme
A retrospective audit of the number of intravitreal injections performed before and after the introduction of three nurse practitioners at the Trust’s main site was performed.
Results
1) Safety
Complications for consecutive nurse-delivered intravitreal injections (n=4000) were recorded over a 24-month period (Table 1). No cases of endophthalmitis, retinal detachment, lens damage, loss of central artery perfusion, uveitis, or vitreous haemorrhage occurred.
2) Patient experience
During the pilot phase of a consecutive series of the first 100 nurse-delivered intravitreal injections, thirteen patients declined the injections. Of these, 10 declined in the first month and 3 declined subsequently.
Some of the reasons given for declining nurse-delivered injections included anxiety related to lack of experience of the nurse and preference for a doctor.
Patient experience questionnaires (n=50) were completed and the results were analysed.
Out of 50 patients, 31 (62%) patients reported complete satisfaction in all elements of care with a maximal score of 5/5 for all questions answered. A further 19 (38%) patients were satisfied with all elements of care with a score of 4/5 for all questions answered. Table 2 shows the mean score in each of the seven elements of patient care.
3) Numbers of injections before and after initiation of the nurse injector programme
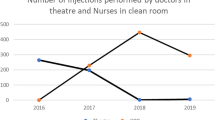
Our results show that introduction of three trained nurse intravitreal injectors increased the mean numbers of intravitreal injections by 25% compared with a similar 5-month period the previous year (Figure 3). A Mann–Whitney test showed a significant (P=0.003) increase in intravitreal injection numbers. This result refers to nurse-delivered intravitreal injection numbers for neovascular AMD only as the initiation phase of the nurse injector programme occurred before NICE approval for ranibizumab for diabetic macular oedema and retinal vein occlusions.
Discussion
AMD is the commonest cause of visual loss and blind registration in patients over the age of 50 years in the developed world.3, 4 In the United Kingdom, it has been estimated that there are about 26 000 new cases of neovascular AMD in the UK each year. 4 The increasingly ageing population have led to a projected rise in cases of sight loss due to neovascular AMD from ∼145 000 to 190 000 by the year 2020.5
The advent of intravitreal ranibizumab therapy has changed the goal of treatment for neovascular age-related macular degeneration from stabilising visual loss to visual improvement. The majority of patients require long-term follow-up and intravitreal ranibizumab retreatment within a 6-week interval for clinical effectiveness. A high-quality service for these patients is challenging to provide with current economic constraints on spending. NICE approval of ranibizumab for diabetic macular oedema has further increased the demand for intravitreal injection therapy. Figures from 2010 showed that the United Kingdom has the lowest ratio of ophthalmologists per head of the population in the European Union (5.2 per 100 000).6 The challenges of providing increased capacity coupled with reduced numbers of medically trained staff have been acknowledged by the Royal College of Ophthalmologists.7, 8 The Royal College of Ophthalmologists position is that, where circumstances and facilities allow, intravitreal injections should be administered by a specialist doctor trained in the procedure.7 However, the College is also aware of the enormous pressure that eye departments are under to deliver this treatment to thousands of new and follow-up patients. The College therefore considers that it is reasonable for non-medical health-care practitioners to administer anti-VEGF agents provided that certain stipulations are met. These include provision of appropriate indemnity arrangements, training, and supervision from ophthalmic specialist doctors particularly with regard to managing any complications.7
We have successfully introduced nurse-delivered intravitreal injections as part of a consultant-led multidisciplinary team. The rationale for this was to increase the capacity to cope with the rising demand for intravitreal injections, reduce reliance on medical staff, and provide continuity of care for patients.
Shared care is an integrated model of health-care provision characterised by collaborative working across professional boundaries. This acknowledges the contribution of different professional groups towards delivering safe, high-quality care through agreed patient pathways. Shared care is instrumental to remove professional barriers, increasing job satisfaction among allied health-care professionals, and providing a cost-effective health-care system.9 Hospital-based shared care involving optometrists and ophthalmic nurses working alongside ophthalmologists to manage patients with ophthalmic disease have been well established.10, 11, 12
The concept of extended roles has been supported by documents such as the NHS Plan,13 which promotes the use of allied health professionals to perform some of the roles traditionally performed by doctors. NHS reforms, such as the European working time directive and modernising medical careers, have led nurses to develop their roles. The Nursing and Midwifery Council supports the idea of extended roles provided that nurses are competently trained and accountable for their own practice.14
Clinical management of neovascular age-related macular degeneration places an increasing burden on the hospital eye service. Despite ongoing efforts to streamline services and patient pathways, considerable problems related to the provision of treatment for neovascular AMD continue. In 2008, NICE-approved ranibizumab and the implications of long-term management are becoming significant. Few of these patients will ever be discharged from follow-up with regular monitoring required to detect disease reactivation. Demand for intravitreal therapy is likely to increase considerably following NICE approval for conditions such as diabetic macular oedema and retinal vein occlusions. NICE approval for these indications was granted in 2013.15, 16
Implementing a shared care model provides a long-term solution to managing eye clinic capacity and increased demand. Medical retina services in many parts of the United Kingdom are under considerable pressure and are operating at maximal capacity. Medical retinal diseases comprise a spectrum of chronic conditions that may be amenable for appropriately trained and supervised non-medical practitioners to manage once stable. Clinical management of these conditions should be driven by protocol, local, and national guidelines based on evidence-based practice.
Our results have shown that trained nurse practitioners can safely deliver intravitreal injections to increase the capacity in medical retina intravitreal injection clinics. Our preliminary results of a series of 4000 nurse-delivered injections associated without serious vision-threatening complication is indicative that this procedure can be safely administered by a nurse. No cases of post-intravitreal anti-VEGF endophthalmitis occurred in our series. A prospective study17 from the British Ophthalmological Survelliance Unit estimated that the incidence rate of post-intravitreal anti-VEGF endophthalmitis in the United Kingdom was 0.025%.
Other units in the United Kingdom have recently introduced nurse-delivered intravitreal injections, and the results published at present indicate that this is safe.18
Our results demonstrate that nurse-delivered intravitreal injections provided a high level of patient satisfaction and were accepted by the majority of patients.
This initiative met several challenges before accomplishing successful implementation. The major challenge to overcome was a legal issue relating to licensing restriction of ranibizumab. Ranibizumab is currently licensed for administration by a trained ophthalmologist only. However, there are treatments in ophthalmology, such as intravitreal administration of triamcinolone, that are not licensed and are in widespread use and are generally accepted. The issue of licensing, therefore, is distinct from practice that would be regarded as not standard. At the time of commencement of the nurse intravitreal programme at Moorfields Eye Hospital, there was no generally accepted body of evidence to support the safety of nurse-administered intravitreal injections. Therefore, should a patient suffer loss of vision as a result of a nurse-administered intravitreal injection this raised potential for a claim of clinical negligence. Legal consultation regarding vicarious liability cover for nurse-administered intravitreal injections was obtained. Vicarious liability in a workplace context refers to situations where employers may be held liable for the acts or omissions of employees provided that it can be shown that this took place in the course of their employment. Written confirmation of vicarious liability cover for nurse practitioner-delivered intravitreal injections by the NHS Litigation Authority (NHSLA) was obtained.
Consultant-led training and individual professional development of the nurse practitioner is critical to driving this change forward. Clarification of indemnity cover, detailed planning, available funding, and a supportive framework to maintain motivation are essential factors for successful implementation.

References
NICE technology appraisal guidance 155; issued August 2008, modified May 2012.
Marshall GN, Hays RD The patient satisfaction questionnaire short form (PSQ-18) 1994: RAND.
Augood CA, Vingerling JR, de Jong PT, Chakravarthy U, Seland J, Soubrane G et al. Prevalence of age-related maculopathy in older Europeans: the European Eye Study (EUREYE). Arch Ophthalmol 2006; 124: 529–535.
Bunce C, Wormald R . Leading causes of certification for blindness and partial sight in England & Wales. BMC Public Health 2006; 6: 58.
Minassian DC, Reidy A, Lightstone A, Desai P . Modelling the prevalence of age-related macular degeneration (2010-2020) in the UK: expected impact of anti-vascular endothelial growth factor (VEGF) therapy. Br J Ophthalmol 2011; 95: 1433–1436.
Resnikoff S, Felch W, Gauthier T-M, Spivey B . The number of ophthalmologists in practice and training worldwide: a growing gap despite more than 200,000 practitioners. Br J Ophthalmol 2012; 96: 783–787.
College statement on intraocular injections by non medical health care professionals, April 2013. www.rcophth.ac.uk. (accessed 26 January 2014).
Royal College of Ophthalmology statement. Maximising capacity in AMD services, 1/12/09 www.rcophth.ac.uk. (accessed 29 January 2014).
Burns-Cox CJ . Shared care, past and future. Ophthalmic Physiol Opt 1995; 15 (5): 379–381.
Department of Health. Shared care guidelines, Issued 2004.
Royal College of Ophthalmologists. Cataract surgery guidelines, Issued 2004.
Riad RF, Dart JK, Cooling RJ . Primary care and ophthalmology in the United Kingdom. Br J Ophthalmol 2003; 87 (4): 493–499.
Department for Health. The NHS Plan: a plan for investment, a plan for reform, Issued 2000.
Nursing and Midwifery Council (NMC) 2008 Code of Professional conduct: Standards for conduct, performance and ethics. NCM, London.
The National Institute for Health and Clinical Excellence. Ranibizumab for treating diabetic macular oedema. NICE technology appraisal guidance 274, Issued 2013.
The National Institute for Health and Clinical Excellence. Ranibizumab for treating visual impairment caused by macular oedema secondary to retinal vein occlusion. NICE technology appraisal guidance 283, Issued 2013.
Lyall DAM, Tey A, Foot B, Roxburgh STD, Virdi M, Robertson C et al. Post-intravitreal anti-VEGF endophthalmitis in the United Kingdom: incidence, features, risk factors, and outcomes. Eye 2012; 26: 1517–1526.
Varma D, Lunt D, Johnson P, Stanley S . A novel approach to expanding the role of nurses to deliver intravitreal injections for patients with age related macular degeneration. Int J Ophthalm Pract 2013; 4 (2): 68–74.
Acknowledgements
Thanks to Bhavin Maru, Gavin Kissondeeal, Mary Masih, Helen Carpenter, Helen Gibbons, Anita Aubrey, Linda Langton, Mary Hardy, Ann Arnold, Merel Reinink, Peter Addison, Aires Lobo, Zoe Ockrim, Praveen Patel, Sobha Sivaprasad, Adnan Tufail, Melanie Hingorani, Frances Stobbs, and John Pelly.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Preliminary data presented at Royal College of Ophthalmologists Annual Congress, Liverpool, UK, May 2013
Rights and permissions
About this article
Cite this article
DaCosta, J., Hamilton, R., Nago, J. et al. Implementation of a nurse-delivered intravitreal injection service. Eye 28, 734–740 (2014). https://doi.org/10.1038/eye.2014.69
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/eye.2014.69
This article is cited by
-
Cost consequences of task-shifting intravitreal injections from physicians to nurses in a tertiary hospital in Norway
BMC Health Services Research (2023)
-
Evaluation of standard-of-care intravitreal aflibercept treatment practices in patients with diabetic macular oedema in the UK: DRAKO study outcomes
Eye (2023)
-
Situational analysis of diabetic retinopathy treatment Services in Ghana
BMC Health Services Research (2021)
-
Task shifting of intravitreal injections from physicians to nurses: a qualitative study
BMC Health Services Research (2021)
-
Safety of nurse-led intravitreal injection of dexamethasone (Ozurdex) implant service. Audit of first 1000 cases
Eye (2021)