Abstract
MicroRNAs (miRNAs), a class of small non-coding RNAs, mediate gene expression by either cleaving target mRNAs or inhibiting their translation. They have key roles in the tumorigenesis of several cancers, including non-small cell lung cancer (NSCLC). The aim of this study was to investigate the clinical significance of miR-638 in the evaluation of NSCLC patient prognosis in response to chemotherapy. First, we detected miR-638 expression levels in vitro in the culture supernatants of the NSCLC cell line SPC-A1 treated with cisplatin, as well as the apoptosis rates of SPC-A1. Second, serum miR-638 expression levels were detected in vivo by using nude mice xenograft models bearing SPC-A1 with and without cisplatin treatment. In the clinic, the serum miR-638 levels of 200 cases of NSCLC patients before and after chemotherapy were determined by quantitative real-time PCR, and the associations of clinicopathological features with miR-638 expression patterns after chemotherapy were analyzed. Our data helped in demonstrating that cisplatin induced apoptosis of the SPC-A1 cells in a dose- and time-dependent manner accompanied by increased miR-638 expression levels in the culture supernatants. In vivo data further revealed that cisplatin induced miR-638 upregulation in the serum derived from mice xenograft models, and in NSCLC patient sera, miR-638 expression patterns after chemotherapy significantly correlated with lymph node metastasis. Moreover, survival analyses revealed that patients who had increased miR-638 levels after chemotherapy showed significantly longer survival time than those who had decreased miR-638 levels. Our findings suggest that serum miR-638 levels are associated with the survival of NSCLC patients and may be considered a potential independent predictor for NSCLC prognosis.
Similar content being viewed by others
Introduction
Lung cancer is characterized with a high incidence and poor prognosis, and it is the leading cause of cancer-related death worldwide, with a dismal 5-year survival rate of less than 15%.1, 2, 3 The most common lung cancer variant is non-small cell lung cancer (NSCLC), which accounts for ~85% of lung cancer cases. Chemotherapy is the most frequent treatment for NSCLC and helps to improve the life quality and prolong the survival of patients.4 However, further work is required to monitor NSCLC patient responses to chemotherapy and thereby improving the patient outcomes.
Mature, biologically active microRNAs (miRNAs) are endogenous short non-coding RNAs and are known to have essential roles in the regulation of numerous biological processes, such as development, differentiation, proliferation and apoptosis.5, 6, 7 They achieve this by interfering with the translation or stability of genes by binding the 3′ UTR of target mRNAs at the posttranscriptional level.8 Over the past few decades, the number of identified miRNAs has continued to grow, and our knowledge of miRNA biology has increased accordingly.9 On the basis of their role in cancer pathology and the types of gene that they target, miRNAs can be divided into oncogenes and tumor suppressor genes. miR-638 has been reported to be downregulated in several types of cancer, such as gastric cancer,10 leukemia11 and basal cell carcinoma,12 and may therefore function as a tumor suppressor gene.
Previously, we successfully prepared a monoclonal antibody named NJ001 that is specific to NSCLC.13 In this earlier study, the lung cancer cell line SPC-A1 was treated with NJ001 in vitro for different periods of time and expression microarray analyses were then performed with the extracted RNA. We found that miR-638 exhibited the most significant time-dependent differential expression in response to NJ001 (GEO accession number: GSE51947). Furthermore, recent studies have revealed that miR-638 is aberrantly expressed in several cancers,10, 14, 15 suggesting that it may be involved in cancer development and progression. Thus, we next focused on exploring the potential clinical application of miR-638. Specifically, the aim of this study was to explore the usefulness of serum miR-638 detection for predicting the NSCLC outcome.
Materials and methods
Cell culture and in vitro experiments
The SPC-A1 cell line was purchased from the type culture collection of the Chinese Academy of Sciences, Shanghai, China, and was proven to be uncontaminated by bacterium or mycoplasma by the European Collection of Cell Cultures using mass spectrometry. The cells were grown in RPMI1640 medium supplemented with 10% fetal bovine serum, 1% penicillin and 1% streptomycin at 37 °C and 5% CO2. For in vitro experiments, SPC-A1 cells in the exponential growth phase were seeded in six-well plates (2 × 106 per well) and treated with different concentrations (0.5, 2.5 and 5 μg ml−1) of cisplatin or complete RPMI1640 medium alone (as the control group) and incubated at 37 °C and 5% CO2 for 24, 48 or 72 h. Cultured supernatants were then harvested for miR-638 detection. Each time point was set in triplicates, and the whole experiment was repeated three times.
Detection of apoptosis
The apoptosis rates of the SPC-A1 cells were determined by flow cytometry. Approximately 1 × 105 cells were collected for apoptosis rate detection by flow cytometry. Cells were re-suspended and stained with Annexin V-FITC and propidium iodide (Annexin V-FITC Apoptosis Detection Kit, BD Biosciences, Franklin, NJ, USA) according to the manufacturer’s instruction. FACS analysis was then carried out on a FACScan cytometer (BD Biosciences).
In vivo tumor establishment, cisplatin treatment and serum isolation
Experimental nude mice were purchased from Slack Experimental Animal Co. in Shanghai with Certification No.: SCXK (Shanghai, China) 2009-0002 and SPF level status. The mice were housed in an SPF animal facility in the Experimental Animal Center of Jiangsu Province. In vivo experiments were carried out using nude mice aged 6 weeks. The xenograft models were established by subcutaneous injection of 2 × 106 SPC-A1 cells, and non-injected mice (n=5) served as a control group. After 2 weeks, when tumor tubercles were significantly observed, the mice were divided into three groups. The first group was injected with cisplatin at a dose of 0.5 μg per 100 μl via the caudal vein (n=6); the second group received an equal volume of normal saline (NS) (n=5) and served as a negative control group; and the remaining xenograft models were not treated with either cisplatin or NS (n=5). The treatments were performed three times every other day. Blood was collected the day after the last administration and centrifuged at a speed of 16 000 g for 10 min at room temperature to isolate serum after coagulation.
Patients and serum specimens
Clinical samples were collected from a cohort of 200 cases of NSCLC patients during their hospitalization from July 2009 to March 2013 at the First Affiliated Hospital of Nanjing Medical University, China. Blood was collected before and after chemotherapy with patients’ informed consent. Serum was then separated from whole blood by centrifugation at 2500 g for 10 min, followed by 16 000 g for 10 min to completely remove cell debris, and then stored at −70 °C until needed for use.
RNA extraction and quantitative RT–PCR
Total RNA was extracted using the miRNeasy mini kit (QIAgen, Valencia, CA, USA) according to the manufacturer’s protocol, with cel-miR-39 (59-ucaccggguguaaaucagcuug-39; final concentration: 10−5 pmol ml−1) (Applied Biosystems, Foster City, CA, USA) added as an internal control. RNA concentration and quality were validated by using a UV spectrophotometer at 260 and 280 nm. Quantitative PCR with reverse transcription (RT–PCR) was used to detect miR-638 expression levels. For reverse transcription, 9.16 μl RNA was used in each reaction and mixed with 5.84 μl reverse transcription reagents (Applied Biosystems), including stem-loop reverse transcription primers, according to the manufacturer’s instructions. The reverse transcription reactions were performed at 16 °C for 30 min, 42 °C for 30 min, 85 °C for 5 min and then maintained at 4 °C. Following this, real-time PCR was conducted at 95 °C for 10 min, followed by 95 °C for 15 s with 45 cycles and 60 °C for 1 min in an ABI 7500 Real-Time PCR system. miR-638 expression levels are presented in terms of fold change normalized to cel-miR-39 expression using the formula 2−ΔΔCt in which ΔΔCt=(Ct miR-638−Ct miR-39)treated−(Ct miR-638−Ct miR-39)untreated.
Statistical analysis
Statistical analyses were performed using Graphpad Prism 5.0 (San Diego, CA, USA) and SPSS 16.0 software (Chicago, IL, USA). A survival analysis was performed with the Kaplan–Meier method, and the log-rank test was used to compare survival times between groups. A P-value <0.05 was considered statistically significant.
Results
Cisplatin induces apoptosis of SPC-A1 cells
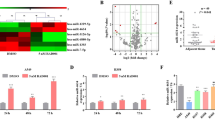
To evaluate the effect of cisplatin on SPC-A1 apoptosis, SPC-A1 cells were treated with different doses of cisplatin for different periods of time. The apoptosis rates for 24, 48 and 72 h were then determined after treatment with/without cisplatin using flow cytometry. The apoptosis rates of the SPC-A1 cells cultured for 24, 48 and 72 h with cisplatin at a concentration of either 2.5 or 5 μg ml−1 were significantly greater than those of paired untreated control groups (Figures 1a and b), and these apoptosis rates increased more markedly with prolonged exposure to higher concentrations of cisplatin. These data suggest that cisplatin induces apoptosis of SPC-A1 cells in a dose- and time-dependent manner.
Cisplatin treatment induces miR-638 expression in a time- and dose-dependent manner in SPC-A1 cultured supernatants
Quantitative RT–PCR (qRT–PCR) was used to detect the expression levels of miR-638 in the culture supernatants of SPC-A1 cells treated with cisplatin. The results showed that the miR-638 expression levels in the SPC-A1-cultured supernatants were more than four-fold higher after being treated with cisplatin for 72 h at a concentration of 2.5 μg ml−1 when compared with expression levels in the untreated control group (Figure 2a). Furthermore, treatment of SPC-A1 cells with either 2.5 or 5.0 μg ml−1 cisplatin both resulted in a significant increase in miR-638 expression levels in the supernatants compared with that of the control group. Moreover, it was observed that the miR-638 levels in supernatants also increased in a time-dependent manner when 5.0 μg ml−1 cisplatin was used for treatment for different periods of time (24–72 h) (Figure 2b).
Relative expression levels of miR-638 in SPC-A1 cultured supernatants treated with cisplatin of 2.5 μg ml−1 (a) and 5 μg ml−1 (b). SPC-A1 cells were treated with different concentrations (0.5, 2.5 and 5 μg ml−1) of cisplatin for 24, 48 and 72 h, and miR-638 expression levels in SPC-A1 cultured supernatants were determined by qRT–PCR. Data represent the mean fold-change in miR-638 expression in three independent groups relative to the untreated control group (*P<0.05).
Cisplatin treatment in vivo up-regulates miR-638 expression levels
To address whether cisplatin treatment affects miR-638 expression in vivo, we analyzed miR-638 expression levels in sera of nude mice SPC-A1 xenograft models with and without cisplatin treatment by using qRT–PCR. A marked increase in miR-638 expression was observed in the sera of the SPC-A1 lung carcinoma group with cisplatin administration compared with the lung carcinoma group without any treatment, the lung carcinoma group with NS treatment or the blank control group. However, no significant difference in serum miR-638 expression was observed in either of the two lung carcinoma groups without any treatment, the lung carcinoma group with NS treatment or the blank control group. An increase in serum miR-638 expression levels was observed as early as 24 h post-treatment with cisplatin. These data suggest that cisplatin administration in SPC-A1 xenograft models induces upregulation of miR-638 expression as early as 24 h post-treatment (Figure 3).
MiR-638 expression levels in serum of nude mice bearing SPC-A1 xenografts. Mice bearing SPC-A1 xenografts were treated with 5 μg ml−1 of cisplatin (200 μl, n=6), NS (200 μl, n=5) or received no treatment (n=5). The next day after the last drug administration, serum was collected and miR-638 expression levels were determined. The graph represents the mean serum miR-638 expression levels relative to the control group without SPC-A1 cells (*P<0.05).
Associations between clinicopathological features and serum miR-638 expression patterns
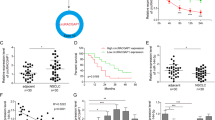
To further explore the relationship between serum miR-638 expression and the clinicopathological features of NSCLC, the correlation between serum miR-638 expression patterns after chemotherapy and clinicopathological characteristics was evaluated. The data presented in Table 1 show that the upregulation of miR-638 in serum was significantly correlated with a lower rate of lymph node metastasis (P=0.017); however, there were no correlations between serum miR-638 expression and age, gender, smoking history or tumor-lymph node-metastasis (TNM) stage (all P>0.05). These results suggest that elevated miR-638 expression may be inversely correlated with NSCLC progression.
Correlation between miR-638 levels and NSCLC patients’ survival
As demonstrated, increased miR-638 expression levels were observed in SPC-A1 culture supernatants in a time-dependant manner after cisplatin treatment. To assess the prognostic value of miR-638 in NSCLC patients receiving chemotherapy, we determined its expression in serum before and after chemotherapy derived from 200 cases of NSCLC patients in which 189 cases displayed miR-638 expression changes. We found that serum miR-638 expression levels increased in 112 cases (~60%) of the NSCLC patients after chemotherapy. After a 1-year follow-up, we observed that the cumulative survival rate of patients with increased miR-638 expression was 59.2% compared with 47.5% of those with decreased miR-638 expression. The survival data for the 183 cases of NSCLC with complete records were analyzed by Kaplan–Meier survival curve and compared with a log-rank test. This analysis revealed a significant association between serum miR-638 levels and the survival of NSCLC patients. Indeed, survival for patients with increased miR-638 expression after chemotherapy was significantly greater than that for patients with a decreased miR-638 expression (P=0.0061, Figures 4 and 5). Moreover, Cox proportional hazards regression analysis performed at the univariate level indicated a significant correlation between overall survival and serum miR-638 expression patterns (HR=2.142; 95% CI, 1.130–4.060; P=0.02) and regional lymph node metastasis (HR=6.035; 95% CI, 2.041–17.845; P=0.001) (Table 2). Clinicopathological features that were significantly correlated with overall survival at the univariate level were then analyzed by multivariate analysis, and the results demonstrated that miR-638 (HR=2.258; 95% CI, 1.031–3.456; P=0.04) and regional lymph node metastasis (HR=3.196; 95% CI, 1.938–15.869; P=0.001) remained significant as independent prognostic factors for overall survival rates (Table 2).
Expression levels of miR-638 in NSCLC patients before and after chemotherapy. Fold changes of serum miR-638 levels in serum of each individual paired NSCLC patient sample before and after chemotherapy. Relative data are represented as log2 (fold change) (after/before), in which a ‘Y’ value >1 was defined as an increase in miR-638 levels, whereas a value <–1 was defined as a decrease in levels.
Kaplan–Meier survival curve for overall survival of NSCLC patients (n=183). Kaplan–Meier analysis was performed to evaluate the overall survival of NSCLC patients according to miR-638 expression patterns after one cycle of first-line chemotherapy, in which 109 patients had higher miR-638 expression levels, and 74 patients had lower miR-638 expression levels.
Dynamic changes in miR-638 levels during chemotherapy
To monitor the dynamics of serum miR-638 during chemotherapy, serum samples from NSCLC patients who received several cycles of chemotherapy were collected after different cycles of chemotherapy (Figure 6). For each individual, serum samples were collected on the last day of each administration of medication, and the relative fold change of 1 represented the serum miR-638 expression level before chemotherapy. Figure 6 shows the dynamic changes in the serum miR-638 expression levels of four representative patients after chemotherapy. Patient A (Figure 6a) received six cycles of chemotherapy, and CT scans showed that the tumor shrank significantly, with decreased levels of tumor markers that were inversely associated with serum miR-638 expression. Indeed, the serum miR-638 level after the last administration of medication was 2.5-fold greater than that before chemotherapy, and the patient was alive at 15 months of follow-up. Patient B (Figure 6b) displayed a stable condition after three cycles of chemotherapy, as determined by CT scans, along with decreased levels of tumor markers, such as CEA, NSE and CYFRA21-1, in the reference range and gradually increased serum miR-638 expression levels. At the time of our most recent follow-up, 27 months after treatment, patient B was still alive. By contrast, patient C (Figure 6c) received four cycles of chemotherapy, during which CT scans determined that the patient’s condition had deteriorated because of tumor metastasis. Throughout treatment, the patient’s serum miR-638 levels were lower after chemotherapy, and the patient lived for only 9 months after cessation of treatment. Likewise, patient D (Figure 6d) received three cycles of chemotherapy and displayed decreased serum miR-638 levels together with tumor progression and metastasis, as determined by CT scans. Additionally, serum levels of CEA, CYFRA21-1 were observed to be higher than before chemotherapy. In particular, serum miR-638 levels decreased significantly after the second and third cycle of chemotherapy in patient D. The relative fold change in serum miR-638 level after the last cycle compared to the pre-treatment level was 0.06, trending to 0. The patient died 5 months after the final diagnosis.
Expression levels of miR-638 at different time points following chemotherapy. (a–d) Serum miR-638 expression levels in four patients who received several cycles of chemotherapy were monitored by qRT–PCR. A relative log2 (fold change) of 1 was considered the initial miR-638 expression level before chemotherapy.
Discussion
miRNAs have been shown to function in the regulation of cell proliferation, apoptosis and cell transformation at a posttranscriptional level.16, 17, 18 In lung cancer, studies have shown that miRNAs play an important role in the development of chemosensitivity or chemoresistance.19, 20 Indeed, miRNAs have been found to have a crucial role in the regulation of tumor biological behavior, and their expression patterns have been linked with tumor development. In the field of oncology, miRNA detection has been confirmed to be applicable in early diagnosis, prognosis and therapeutic evaluation.21, 22, 23, 24
Accumulating studies have revealed the dysregulation of several miRNAs in NSCLC, suggesting that they play important roles in tumorigenesis and the progression of NSCLC. For instance, the downregulation of miR-497 in NSCLC promotes cell proliferation and angiogenesis.25 miR-149 is also significantly downregulated in NSCLC cells and is inversely correlated with invasion through the inhibition of epithelial mesenchymal transition by targeting FOXM1.26 Similarly, miR-181b is markedly downregulated in NSCLC and significantly correlates with tumor size, p-TNM/TNM stage, and the status of lymph node metastasis.27 Moreover, miR-449a has been shown to inhibit both cell migration and invasion by directly targeting the c-Met gene in NSCLC, downregulation of which has been associated with poor prognosis.28 Here, we aimed to investigate the clinical significance of miR-638 in NSCLC.
A previous expression microarray analysis illustrated that SPC-A1 cells treated with NJ001 exhibited the most significantly increased expression of miR-638 in a time-dependent manner. To further elucidate the potential role and clinical value of miR-638 in NSCLC, we treated SPC-A1 cells with different concentrations of cisplatin for different periods of time in vitro and measured the apoptosis rates and the expression levels of miR-638 in culture supernatants. The results showed that cisplatin induced SPC-A1 apoptosis in a dose- and time-dependent manner. Importantly, this positively correlated with a time-dependent increase in miR-638 expression in the culture supernatant when the concentration of cisplatin was 2.5 or 5.0 μg ml−1. Moreover, miR-638 expression gradually increased following prolonged exposure times and increased apoptosis rates, indicating a possible positive role for miR-638 in apoptosis. Furthermore, in vivo tests demonstrated that cisplatin-induced upregulation of miR-638 in the sera of nude mice xenograft models of NSCLC could be detected 24 h after administration, suggesting that miR-638 expression is increased at an early stage in response to cisplatin. We also measured the serum miR-638 levels of 189 NSCLC patients by qRT–PCR analysis and found that the 1-year cumulative survival rate of patients with increased serum miR-638 levels was 61.4% compared with 49.2% in patients who had decreased serum miR-638 levels after one standard chemotherapy cycle. Survival analyses further indicated that NSCLC patients with higher serum miR-638 expression after chemotherapy had a longer mean survival time than those with lower serum miR-638 expression. Serum expression patterns of miR-638 after chemotherapy were also associated with lymph node metastasis, suggesting that expression patterns may be inversely related to cell invasion in NSCLC. In addition, monitoring the dynamic changes in miR-638 expression before and after chemotherapy in the sera of representative patients demonstrated that those who had positive responses to chemotherapy showed increased serum miR-638 levels, whereas those who displayed stable or progressive disease exhibited decreased serum miR-638 levels.
The value of miRNAs as prognostic tools has previously been demonstrated.29, 30, 31, 32 Indeed, as non-invasive biomarkers of disease and therapy response, miRNAs could serve as prognostic indicators in some solid tumors. For example, serum miR-221 is closely related to ovarian cancer prognosis, and its elevated expression indicates a poor prognosis.23 Additionally, colorectal cancer patients with higher serum expression of miR-92a have a poorer prognosis.22
It has been reported that miR-638 is down-regulated in several types of cancer, including lung cancer. miR-638 is dramatically down-regulated in gastric cancer tissues, and it has been shown to inhibit the expression of cyclin D1 by inhibiting specificity protein 2 (Sp2) expression, thus suppressing gastric cancer cell proliferation.33 Downregulation of miR-638 expression has been confirmed in NSCLC tissues and is reported to be involved in promoting the transformation of normal cells into tumor cells.14 Moreover, downregulated miR-638 has been shown to induce cell invasion and proliferation by regulating SRY-box 2, which is related to epithelial-to-mesenchymal transition in the development of NSCLC, and the effect is reversed by high expression of miR-638 or silencing of SRY-box 2.34 A similar functional role has also been found in colorectal carcinoma cells.35 However, over-expression of miR-638 in vascular smooth muscle cells has been shown to inhibit cell proliferation and migration by targeting the NOR1 receptor,36 which might partially explain the correlation between the increased expression of miR-638 and favorable prognosis. In our previous study, over-expression of miR-638 in vitro induced apoptosis of lung cancer cells by regulating P21-activated protein kinase. However, a clinically significant link between levels of miR-638 in serum and NSCLC prognosis has not been previously reported.
In conclusion, we have revealed that serum miR-638 expression levels varied in NSCLC patients after chemotherapy, which was associated with NSCLC prognosis. More specifically, upregulation of serum miR-638 was correlated with a favorable prognosis for NSCLC patients. This is also the first study to indicate that miR-638 expression patterns correlate with lymph node metastasis and clinical outcome. Serum miR-638 is a useful independent factor for NSCLC prognosis and could be used as a potential prognostic biomarker for overall survival of NSCLC patients. However, further studies are required to explore the underlying molecular mechanisms of miR-638 in lung cancer tumorigenesis and progression and its possible use as a therapeutic target for NSCLC.
Accession codes
References
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T et al. Cancer statistics, 2008. CA Cancer J Clin 2008; 58: 71–96.
Jemal A, Siegel R, Xu J, Ward E . Cancer statistics, 2010. CA Cancer J Clin 2010; 60: 277–300.
Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM . Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 2010; 127: 2893–2917.
Hotta K, Kiura K, Fujiwara Y, Takigawa N, Hisamoto A, Ichihara E et al. Role of survival post-progression in phase III trials of systemic chemotherapy in advanced non-small-cell lung cancer: a systematic review. PLoS ONE 2011; 6: e26646.
Ambros V . The functions of animal microRNAs. Nature 2004; 431: 350–355.
Bartel DP . MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 2004; 116: 281–297.
Broderick JA, Zamore PD . MicroRNA therapeutics. Gene Ther 2011; 18: 1104–1110.
Behm-Ansmant I, Rehwinkel J, Izaurralde E . MicroRNAs silence gene expression by repressing protein expression and/or by promoting mRNA decay. Cold Spring Harb Symp Quant Biol 2006; 71: 523–530.
Ruby JG, Jan C, Player C, Axtell MJ, Lee W, Nusbaum C et al. Large-scale sequencing reveals 21U-RNAs and additional microRNAs and endogenous siRNAs in C. elegans. Cell 2006; 127: 1193–1207.
Yao Y, Suo AL, Li ZF, Liu LY, Tian T, Ni L et al. MicroRNA profiling of human gastric cancer. Mol Med Rep 2009; 2: 963–970.
Zhu DX, Zhu W, Fang C, Fan L, Zou ZJ, Wang YH et al. miR-181a/b significantly enhances drug sensitivity in chronic lymphocytic leukemia cells via targeting multiple anti-apoptosis genes. Carcinogenesis 2012; 33: 1294–1301.
Sand M, Skrygan M, Sand D, Georgas D, Hahn SA, Gambichler T et al. Expression of microRNAs in basal cell carcinoma. Br J Dermatol 2012; 167: 847–855.
Pan S, Wang F, Huang P, Xu T, Zhang L, Xu J et al. The study on newly developed McAb NJ001 specific to non-small cell lung cancer and its biological characteristics. PLoS ONE 2012; 7: e33009.
Li D, Wang Q, Liu C, Duan H, Zeng X, Zhang B et al. Aberrant expression of miR-638 contributes to benzo(a)pyrene-induced human cell transformation. Toxicol Sci 2012; 125: 382–391.
Xu L, Liang YN, Luo XQ, Liu XD, Guo HX . Association of miRNAs expression profiles with prognosis and relapse in childhood acute lymphoblastic leukemia. Zhonghua Xue Ye Xue Za Zhi 2011; 32: 178–181.
Brennecke J, Hipfner DR, Stark A, Russell RB, Cohen SM . Bantam encodes a developmentally regulated microRNA that controls cell proliferation and regulates the proapoptotic gene hid in Drosophila. Cell 2003; 113: 25–36.
Poy MN, Eliasson L, Krutzfeldt J, Kuwajima S, Ma X, Macdonald PE et al. A pancreatic islet-specific microRNA regulates insulin secretion. Nature 2004; 432: 226–230.
Thai TH, Calado DP, Casola S, Ansel KM, Xiao C, Xue Y et al. Regulation of the germinal center response by microRNA-155. Science 2007; 316: 604–608.
Acunzo M, Visone R, Romano G, Veronese A, Lovat F, Palmieri D et al. miR-130a targets MET and induces TRAIL-sensitivity in NSCLC by downregulating miR-221 and 222. Oncogene 2012; 31: 634–642.
Garofalo M, Romano G, Di Leva G, Nuovo G, Jeon YJ, Ngankeu A et al. EGFR and MET receptor tyrosine kinase-altered microRNA expression induces tumorigenesis and gefitinib resistance in lung cancers. Nat Med 2012; 18: 74–82.
Xu YZ, Xi QH, Ge WL, Zhang XQ . Identification of Serum MicroRNA-21 as a biomarker for early detection and prognosis in human epithelial ovarian cancer. Asian Pac J Cancer Prev 2013; 14: 1057–1060.
Liu GH, Zhou ZG, Chen R, Wang MJ, Zhou B, Li Y et al. Serum miR-21 and miR-92a as biomarkers in the diagnosis and prognosis of colorectal cancer. Tumour Biol 2013; 34: 2175–2181.
Hong F, Li Y, Xu Y, Zhu L . Prognostic significance of serum microRNA-221 expression in human epithelial ovarian cancer. J Int Med Res 2013; 41: 64–71.
Gimenes-Teixeira HL, Lucena-Araujo AR, Dos Santos GA, Zanette DL, Scheucher PS, Oliveira LC et al. Increased expression of miR-221 is associated with shorter overall survival in T-cell acute lymphoid leukemia. Exp Hematol Oncol 2013; 2: 10.
Zhao WY, Wang Y, An ZJ, Shi CG, Zhu GA, Wang B et al. Downregulation of miR-497 promotes tumor growth and angiogenesis by targeting HDGF in non-small cell lung cancer. Biochem Biophys Res Commun 2013; 435: 466–471.
Ke Y, Zhao W, Xiong J, Cao R . miR-149 inhibits non-small-cell lung cancer cells EMT by targeting FOXM1. Biochem Res Int 2013; 2013: 506731.
Yang J, Liu H, Wang H, Sun Y . Down-regulation of microRNA-181b is a potential prognostic marker of non-small cell lung cancer. Pathol Res Pract 2013; 209: 490–494.
Luo W, Huang B, Li Z, Li H, Sun L, Zhang Q et al. MicroRNA-449a is downregulated in non-small cell lung cancer and inhibits migration and invasion by targeting c-Met. PLoS ONE 2013; 8: e64759.
Yamashita S, Yamamoto H, Mimori K, Nishida N, Takahashi H, Haraguchi N et al. MicroRNA-372 is associated with poor prognosis in colorectal cancer. Oncology 2012; 82: 205–212.
Fabbri M . miRNAs as molecular biomarkers of cancer. Expert Rev Mol Diagn 2010; 10: 435–444.
Cortez MA, Welsh JW, Calin GA . Circulating microRNAs as noninvasive biomarkers in breast cancer. Recent Results Cancer Res 2012; 195: 151–161.
Madhavan D, Cuk K, Burwinkel B, Yang R . Cancer diagnosis and prognosis decoded by blood-based circulating microRNA signatures. Front Genet 2013; 4: 116.
Zhao LY, Yao Y, Han J, Yang J, Wang XF et al. miR-638 suppresses cell proliferation in gastric cancer by targeting Sp2. Dig Dis Sci 2014; 59: 1743–1753.
Xia Y, Wu Y, Liu B, Wang P, Chen Y . Downregulation of miR-638 promotes invasion and proliferation by regulating SOX2 and induces EMT in NSCLC. FEBS Lett 2014; 588: 2238–2245.
Ma K, Pan X, Fan P, He Y, Gu J, Wang W et al. Loss of miR-638 in vitro promotes cell invasion and a mesenchymal-like transition by influencing SOX2 expression in colorectal carcinoma cells. Mol Cancer 2014; 13: 118.
Li P, Liu Y, Yi B, Wang G, You X, Zhao X et al. MicroRNA-638 is highly expressed in human vascular smooth muscle cells and inhibits PDGF-BB-induced cell proliferation and migration through targeting orphan nuclear receptor NOR1. Cardiovasc Res 2013; 99: 185–193.
Acknowledgements
We are grateful for the technical support from National Key Clinical Department of Laboratory Medicine of Jiangsu Province Hospital. This study was supported by the National Natural Science Foundation of China (Numbers 81371894, 81272324, 81201359, 81101322 and 81000754), the Key Laboratory for Laboratory Medicine of Jiangsu Province of China (Number XK201114), A Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions and the Doctoral Supervisor Foundation of the Ministry of Education (20113234110012).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Wang, F., Lou, Jf., Cao, Y. et al. miR-638 is a new biomarker for outcome prediction of non-small cell lung cancer patients receiving chemotherapy. Exp Mol Med 47, e162 (2015). https://doi.org/10.1038/emm.2015.17
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/emm.2015.17
This article is cited by
-
MicroRNA: trends in clinical trials of cancer diagnosis and therapy strategies
Experimental & Molecular Medicine (2023)
-
MicroRNA-638 inhibits the progression of breast cancer through targeting HOXA9 and suppressing Wnt/β-cadherin pathway
World Journal of Surgical Oncology (2021)
-
A mathematical model as a tool to identify microRNAs with highest impact on transcriptome changes
BMC Genomics (2019)
-
Epigenetic predictive biomarkers for response or outcome to platinum-based chemotherapy in non-small cell lung cancer, current state-of-art
The Pharmacogenomics Journal (2019)
-
MeCP2, a target of miR-638, facilitates gastric cancer cell proliferation through activation of the MEK1/2–ERK1/2 signaling pathway by upregulating GIT1
Oncogenesis (2017)