Abstract
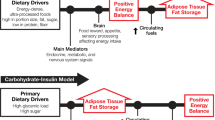
The carbohydrate–insulin model of obesity theorizes that diets high in carbohydrate are particularly fattening due to their propensity to elevate insulin secretion. Insulin directs the partitioning of energy toward storage as fat in adipose tissue and away from oxidation by metabolically active tissues and purportedly results in a perceived state of cellular internal starvation. In response, hunger and appetite increases and metabolism is suppressed, thereby promoting the positive energy balance associated with the development of obesity. Several logical consequences of this carbohydrate–insulin model of obesity were recently investigated in a pair of carefully controlled inpatient feeding studies whose results failed to support key model predictions. Therefore, important aspects of carbohydrate–insulin model have been experimentally falsified suggesting that the model is too simplistic. This review describes the current state of the carbohydrate–insulin model and the implications of its recent experimental tests.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Frayn KN, Karpe F, Fielding BA, Macdonald IA, Coppack SW . Integrative physiology of human adipose tissue. Int J Obes Relat Metab Disord 2003; 27: 875–888.
Ludwig DS, Friedman MI . Increasing adiposity: consequence or cause of overeating? JAMA 2014; 311: 2167–2168.
Lustig RH . Childhood obesity: behavioral aberration or biochemical drive? Reinterpreting the First Law of Thermodynamics. Nat Clin Pract Endocrinol Metab 2006; 2: 447–458.
Taubes G . The science of obesity: what do we really know about what makes us fat? An essay by Gary Taubes. BMJ 2013; 346: f1050.
Wells JC, Siervo M . Obesity and energy balance: is the tail wagging the dog? Eur J Clin Nutr 2011; 65: 1173–1189.
Astwood EB . The heritage of corpulence. Endocrinology 1962; 71: 337–341.
Pennington AW . Obesity. Med Times 1952; 80: 389–398.
Carver C . Insulin treatment and the problem of weight gain in type 2 diabetes. Diabetes Educ 2006; 32: 910–917.
Foster GD, Wyatt HR, Hill JO, McGuckin BG, Brill C, Mohammed BS et al. A randomized trial of a low-carbohydrate diet for obesity. N Engl J Med 2003; 348: 2082–2090.
Gardner CD, Kiazand A, Alhassan S, Kim S, Stafford RS, Balise RR et al. Comparison of the Atkins, Zone, Ornish, and LEARN diets for change in weight and related risk factors among overweight premenopausal women: the A TO Z Weight Loss Study: a randomized trial. Jama 2007; 297: 969–977.
Samaha FF, Iqbal N, Seshadri P, Chicano KL, Daily DA, McGrory J et al. A low-carbohydrate as compared with a low-fat diet in severe obesity. N Engl J Med 2003; 348: 2074–2081.
Atkins RC . Dr Atkins' Diet Revolution: The High Calorie Way to Stay Thin Forever. Bantam Books, 1973.
Ludwig DS . Always hungry? Conquer cravings, retrain your fat cells and lose weight permanetly. Grand Central Life & Style: New York, 2016.
Taubes G . Why We Get Fat And What To Do About It. Alfred A Knopf: New York, 2011.
Popper K . The Logic of Scientific Discovery. Hutchison & Co., 1959.
French AP . Special Relativity. W. W. Norton & Company, 1968.
Finegood DT . The importance of systems thinking to address obesity. Nestle Nutr Inst Workshop Ser 2012; 73: 123–137; discussion 139–141.
Das SK, Gilhooly CH, Golden JK, Pittas AG, Fuss PJ, Cheatham RA et al. Long-term effects of 2 energy-restricted diets differing in glycemic load on dietary adherence, body composition, and metabolism in CALERIE: a 1-y randomized controlled trial. Am J Clin Nutr 2007; 85: 1023–1030.
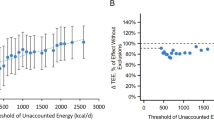
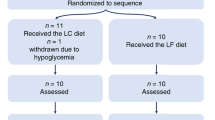
Hall KD, Bemis T, Brychta R, Chen KY, Courville A, Crayner EJ et al. Calorie for calorie, dietary fat restriction results in more body fat loss than carbohydrate restriction in people with obesity. Cell Metab 2015; 22: 427–436.
Hall KD, Chen KY, Guo J, Lam YY, Leibel RL, Mayer LE et al. Energy expenditure and body composition changes after an isocaloric ketogenic diet in overweight and obese men. Am J Clin Nutr 2016; 104: 324–333.
Feinman RD, Fine EJ . Thermodynamics and metabolic advantage of weight loss diets. Metab Syndr Relat Disord 2003; 1: 209–219.
Astrup A, Buemann B, Christensen NJ, Toubro S . Failure to increase lipid oxidation in response to increasing dietary fat content in formerly obese women. Am J Physiol 1994; 266 (4 Pt 1), E592–E599.
Dirlewanger M, di Vetta V, Guenat E, Battilana P, Seematter G, Schneiter P et al. Effects of short-term carbohydrate or fat overfeeding on energy expenditure and plasma leptin concentrations in healthy female subjects. Int J Obes Relat Metab Disord 2000; 24: 1413–1418.
Horton TJ, Drougas H, Brachey A, Reed GW, Peters JC, Hill JO . Fat and carbohydrate overfeeding in humans: different effects on energy storage. Am J Clin Nutr 1995; 62: 19–29.
Shepard TY, Weil KM, Sharp TA, Grunwald GK, Bell ML, Hill JO et al. Occasional physical inactivity combined with a high-fat diet may be important in the development and maintenance of obesity in human subjects. Am J Clin Nutr 2001; 73: 703–708.
Davy KP, Horton T, Davy BM, Bessessen D, Hill JO . Regulation of macronutrient balance in healthy young and older men. Int J Obes Relat Metab Disord 2001; 25: 1497–1502.
Eckel RH, Hernandez TL, Bell ML, Weil KM, Shepard TY, Grunwald GK et al. Carbohydrate balance predicts weight and fat gain in adults. Am J Clin Nutr 2006; 83: 803–808.
Hill JO, Peters JC, Reed GW, Schlundt DG, Sharp T, Greene HL . Nutrient balance in humans: effects of diet composition. Am J Clin Nutr 1991; 54: 10–17.
Leibel RL, Hirsch J, Appel BE, Checani GC . Energy intake required to maintain body weight is not affected by wide variation in diet composition. Am J Clin Nutr 1992; 55: 350–355.
Rumpler WV, Seale JL, Miles CW, Bodwell CE . Energy-intake restriction and diet-composition effects on energy expenditure in men. Am J Clin Nutr 1991; 53: 430–436.
Schrauwen P, van Marken Lichtenbelt WD, Saris WH, Westerterp KR . Changes in fat oxidation in response to a high-fat diet. Am J Clin Nutr 1997; 66: 276–282.
Smith SR, de Jonge L, Zachwieja JJ, Roy H, Nguyen T, Rood JC et al. Fat and carbohydrate balances during adaptation to a high-fat. Am J Clin Nutr 2000; 71: 450–457.
Thearle MS, Pannacciulli N, Bonfiglio S, Pacak K, Krakoff J . Extent and determinants of thermogenic responses to 24 hours of fasting, energy balance, and five different overfeeding diets in humans. J Clin Endocrinol Metab 2013; 98: 2791–2799.
Treuth MS, Sunehag AL, Trautwein LM, Bier DM, Haymond MW, Butte NF . Metabolic adaptation to high-fat and high-carbohydrate diets in children and adolescents. Am J Clin Nutr 2003; 77: 479–489.
Yerboeket-van de Venne WP, Westerterp KR . Effects of dietary fat and carbohydrate exchange on human energy metabolism. Appetite 1996; 26: 287–300.
Bogardus C, LaGrange BM, Horton ES, Sims EA . Comparison of carbohydrate-containing and carbohydrate-restricted hypocaloric diets in the treatment of obesity. Endurance and metabolic fuel homeostasis during strenuous exercise. J Clin Invest 1981; 68: 399–404.
Golay A, Allaz AF, Morel Y, de Tonnac N, Tankova S, Reaven G . Similar weight loss with low- or high-carbohydrate diets. Am J Clin Nutr 1996; 63: 174–178.
Miyashita Y, Koide N, Ohtsuka M, Ozaki H, Itoh Y, Oyama T et al. Beneficial effect of low carbohydrate in low calorie diets on visceral fat reduction in type 2 diabetic patients with obesity. Diabetes Res Clin Pract 2004; 65: 235–241.
Yang MU, Van Itallie TB . Composition of weight lost during short-term weight reduction. Metabolic responses of obese subjects to starvation and low-calorie ketogenic and nonketogenic diets. J Clin Invest 1976; 58: 722–730.
Ebbeling CB, Swain JF, Feldman HA, Wong WW, Hachey DL, Garcia-Lago E et al. Effects of dietary composition on energy expenditure during weight-loss maintenance. Jama 2012; 307: 2627–2634.
Leidy HJ, Clifton PM, Astrup A, Wycherley TP, Westerterp-Plantenga MS, Luscombe-Marsh ND et al. The role of protein in weight loss and maintenance. Am J Clin Nutr 2015; 101: 1320S–1329S.
Wycherley TP, Moran LJ, Clifton PM, Noakes M, Brinkworth GD . Effects of energy-restricted high-protein, low-fat compared with standard-protein, low-fat diets: a meta-analysis of randomized controlled trials. Am J Clin Nutr 2012; 96: 1281–1298.
Hall KD . Comment on PMID: 22735432 effects of dietary composition on energy expenditure during weight-loss maintenance. PubMed Commons 2016. Available at: http://www.ncbi.nlm.nih.gov/pubmed/22735432#cm22735432_22716096.
Kahlhofer J, Lagerpusch M, Enderle J, Eggeling B, Braun W, Pape D et al. Carbohydrate intake and glycemic index affect substrate oxidation during a controlled weight cycle in healthy men. Eur J Clin Nutr 2014; 68: 1060–1066.
Bortz WM, Paul P, Haff AC, Holmes WL . Glycerol turnover and oxidation in man. J Clin Invest 1972; 51: 1537–1546.
Balasse EO, Fery F . Ketone body production and disposal: effects of fasting, diabetes, and exercise. Diabetes Metab Rev 1989; 5: 247–270.
Phinney SD . Ketogenic diets and physical performance. Nutr Metab (Lond) 2004; 1: 2.
Gibson AA, Seimon RV, Lee CM, Ayre J, Franklin J, Markovic TP et al. Do ketogenic diets really suppress appetite? A systematic review and meta-analysis. Obes Rev 2015; 16: 64–76.
Paoli A, Bosco G, Camporesi EM, Mangar D . Ketosis, ketogenic diet and food intake control: a complex relationship. Front Psychol 2015; 6: 27.
Westman EC, Feinman RD, Mavropoulos JC, Vernon MC, Volek JS, Wortman JA et al. Low-carbohydrate nutrition and metabolism. Am J Clin Nutr 2007; 86: 276–284.
Acknowledgements
This research was supported by the Intramural Research Program of the NIH, National Institute of Diabetes and Digestive and Kidney Diseases.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
I have received funding from the Nutrition Science Initiative to investigate the effects of ketogenic diets on human energy expenditure. I also have a patent pending on a method of personalized dynamic feedback control of body weight (US Patent Application No 13/754058; assigned to the National Institutes of Health).
Rights and permissions
About this article
Cite this article
Hall, K. A review of the carbohydrate–insulin model of obesity. Eur J Clin Nutr 71, 323–326 (2017). https://doi.org/10.1038/ejcn.2016.260
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2016.260
This article is cited by
-
Increased dietary intake of ultraprocessed foods and mitochondrial metabolism alterations in pediatric obesity
Scientific Reports (2023)
-
Multi-omics microsampling for the profiling of lifestyle-associated changes in health
Nature Biomedical Engineering (2023)
-
The effects of low-fat, high-carbohydrate diets vs. low-carbohydrate, high-fat diets on weight, blood pressure, serum liquids and blood glucose: a systematic review and meta-analysis
European Journal of Clinical Nutrition (2022)
-
Competing paradigms of obesity pathogenesis: energy balance versus carbohydrate-insulin models
European Journal of Clinical Nutrition (2022)
-
Significantly different roles of economic affluence in sex-specific obesity prevalence rates: understanding more modifications within female body weight management
Scientific Reports (2022)