Abstract
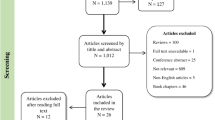
In developing countries, deficiencies of micronutrients are thought to have a major impact on child development; however, a consensus on the specific relationship between dietary zinc intake and cognitive function remains elusive. The aim of this systematic review was to examine the relationship between zinc intake, status and indices of cognitive function in children and adults. A systematic literature search was conducted using EMBASE, MEDLINE and Cochrane Library databases from inception to March 2014. Included studies were those that supplied zinc as supplements or measured dietary zinc intake. A meta-analysis of the extracted data was performed where sufficient data were available. Of all of the potentially relevant papers, 18 studies met the inclusion criteria, 12 of which were randomised controlled trials (RCTs; 11 in children and 1 in adults) and 6 were observational studies (2 in children and 4 in adults). Nine of the 18 studies reported a positive association between zinc intake or status with one or more measure of cognitive function. Meta-analysis of data from the adult’s studies was not possible because of limited number of studies. A meta-analysis of data from the six RCTs conducted in children revealed that there was no significant overall effect of zinc intake on any indices of cognitive function: intelligence, standard mean difference of <0.001 (95% confidence interval (CI) –0.12, 0.13) P=0.95; executive function, standard mean difference of 0.08 (95% CI, –0.06, 022) P=0.26; and motor skills standard mean difference of 0.11 (95% CI –0.17, 0.39) P=0.43. Heterogeneity in the study designs was a major limitation, hence only a small number (n=6) of studies could be included in the meta-analyses. Meta-analysis failed to show a significant effect of zinc supplementation on cognitive functioning in children though, taken as a whole, there were some small indicators of improvement on aspects of executive function and motor development following supplementation but high-quality RCTs are necessary to investigate this further.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Huskisson E, Maggini S, Ruf M . The influence of micronutrients on cognitive function and performance. J Int Med Res 2007; 35: 1–19.
Dauncey MJ . New insights into nutrition and cognitive neuroscience. Proc Nutr Soc 2009; 68: 408–415.
Georgieff MK . Nutrition and the developing brain: nutrient priorities and measurement. Am J Clin Nutr 2007; 85: 614S–620S.
Sensi SL, Paoletti P, Koh JY, Aizenman E, Bush AI, Hershfinkel M . The neurophysiology and pathology of brain zinc. J Neurosci 2011; 31: 16076–16085.
Black MM . Zinc deficiency and child development. Am J Clin Nutr 1998; 68: 464–469.
Levenson CW . Regulation of the NMDA receptor: implications for neuropsychological development. Nutr Rev 2006; 64: 428–432.
Takeda A . Movement of zinc and its functional significance in the brain. Brain Res Rev 2000; 34: 137–148.
Flinn JM, Hunter D, Linkous DH, Lanzirotti A, Smith LN, Brightwell J et al. Enhanced zinc consumption causes memory deficits and increased brain levels of zinc. Physiol Behav 2005; 83: 793–803.
Bitanihirwe BKY, Cunningham MG . Zinc: the brain's dark horse. Synapse 2009; 63: 1029–1049.
Terhune MW, Sandstead HH . Decreased RNA polymerase activity in mammalian zinc deficiency. Science 1972; 177: 68–69.
Black MM . The evidence linking zinc deficiency with children's cognitive and motor functioning. J Nutr 2003; 133: 1473S–1476S.
Black MM . Micronutrient deficiencies and cognitive functioning. J Nutr 2003; 133: 3927S–3931S.
Hurley LS, Swenert'on H . Congenital malformations resulting from zinc deficiency in rats. Exp Biol Med 1966; 123: 692–696.
Dvergsten CL, Fosmire GJ, Ollerich DA, Sandstead HH . Alterations in the postnatal development of the cerebellar cortex due to zinc deficiency. I. Impaired acquisition of granule cells. Brain Res 1983; 271: 217–226.
Takeda A . Zinc homeostasis and functions of zinc in the brain. Biometals 2001; 14: 343–351.
Sandstead HH, Frederickson CJ, Penland JG . History of zinc as related to brain function. J Nutr 2000; 130: 496.
Massaro TF, Mohs M, Fosmire G . Effects of moderate zinc deficiency on cognitive performance in young adult rats. Physiol Behav 1982; 29: 117–121.
Boroujeni ST, Naghdi N, Shahbazi M, Farrokhi A, Bagherzadeh F, Kazemnejad A et al. The effect of severe zinc deficiency and zinc supplement on spatial learning and memory. Biol Trace Elem Res 2009; 130: 48–61.
Halas E, Eberhardt M, Diers M, Sandstead H . Learning and memory impairment in adult rats due to severe zinc deficiency during lactation. Physiol Behav 1983; 30: 371–381.
Takeda A, Tamano H, Tochigi M, Oku N . Zinc homeostasis in the hippocampus of zinc-deficient young adult rats. Neurochem Int 2005; 46: 221–225.
Halas ES, Reynolds GM, Sandstead HH . Intra-uterine nutrition and its effects on aggression. Physiol Behav 1977; 19: 653–661.
Bhatnagar S, Taneja S . Zinc and cognitive development. Br J Nutr 2001; 85: S139–S145.
Benton D . Micronutrient status, cognition and behavioral problems in childhood. Eur J Nutr 2008; 47: 38–50.
Sandstead HH . Zinc is essential for brain development and function. J Trace Elem Exp Med 2003; 16: 165–173.
Black JL, Piñero DJ, Parekh N . Zinc and cognitive development in children: perspectives from international studies. Topics Clin Nutr 2009; 24: 130–138.
Maret W, Sandstead HH . Possible roles of zinc nutriture in the fetal origins of disease. Exp Gerontol 2008; 43: 378–381.
Black RE, Allen LH, Bhutta ZA, Caulfield LE, De Onis M, Ezzati M et al. Maternal and child undernutrition: global and regional exposures and health consequences. Lancet 2008; 371: 243–260.
Sandstrom B, Sandberg AS . Inhibitory effects of isolated inositol phosphates on zinc-absorption in humans. J Trace Elem Electrolytes Health Dis 1992; 6: 99–103.
Grantham-McGregor SM, Fernald LC, Sethuraman K . Effects of health and nutrition on cognitive and behavioural development in children in the first three years of life. Part 2: infections and micronutrient deficiencies: iodine, iron, and zinc http://archive.unu.edu/unupress/food/V201e/ch08.htm. Food Nutr Bull 1999; 20: 76–99.
Sandstead HH . Causes of iron and zinc deficiencies and their effects on brain. J Nutr 2000; 130: 347S–349S.
Grantham-McGregor SM, Ani CC . The role of micronutrients in psychomotor sad cognitive development. Br Med Bull 1999; 55: 511–527.
Walker SP, Wachs TD, Gardner JM, Lozoff B, Wasserman GA, Pollitt E et al. Child development in developing countries 2 - Child development: risk factors for adverse outcomes in developing countries. Lancet 2007; 369: 145–157.
Golub M, Takeuchi P, Keen C, Gershwin M, Hendrickx A, Lonnerdal B . Modulation of behavioral performance of prepubertal monkeys by moderate dietary zinc deprivation. Am J Clin Nutr 1994; 60: 238–243.
Black MM . Zinc deficiency and cognitive development. In: Benton D editor. Lifetime Nutritional Influences on Cognition, Behaviour and Psychiatric Illness Woodhead Publishing in Food Science Technology and Nutrition. Woodhead Publ Ltd: Cambridge, 2011, pp 79–93.
Dreosti I . Zinc in brain development and function. In: Tomita H editor. Trace Elements in Clinical Medicine. Springer: Japan, 1990, pp 47–52.
Dhonukshe-Rutten RAM, Bouwman J, Brown KA, AEJM Cavelaars, Collings R, Grammatikaki E et al. EURRECA—evidence-based methodology for deriving micronutrient recommendations. Crit Rev Food Sci Nutr 2013; 53: 999–1040.
EURRECA Systematic reviews: methods and main results. Available at http://www.eurreca.org/everyone/8567/7/0/32 (accessed 8 January 2015).
EURRECA Best practice guidelines: nutrient intake assessment. Available at http://www.eurreca.org/everyone/8632/5/0/32 (accessed 8 January 2015).
Matthys, van 't Veer P, de Groot L, Hooper L, Cavelaars AE, Collings R et al. EURRECA's approach for estimating micronutrient requirements. Int J Vitam Nutr Res 2011; 81: 256–263.
Serra-Majem L, Pfrimer K, Doreste-Alonso J, Ribas-Barba L, Sanchez-Villegas A, Ortiz-Andrellucchi A et al. Dietary assessment methods for intakes of iron, calcium, selenium, zinc and iodine. Br J Nutr 2009; 102: S38–S55.
Nissensohn M, Sánchez-Villegas A, Fuentes Lugo D, Henríquez Sánchez P, Doreste Alonso J, Skinner AL et al. Effect of zinc intake on mental and motor development in infants: a meta-analysis. Int J Vitamin Nutr Res 2013; 83: 203–215.
EURRECA Databases and search strategies for zinc. Available at http://www.eurreca.org/everyone/8414/5/0/32 (accessed 8 January 2015).
Higgins JPT, Green S (eds). Cochrane Handbook for Systematic Reviews of Interventions, Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011. Available from (www.cochrane-handbook.org (accessed 9 April 2015).
Higgins J, Thompson SG . Quantifying heterogeneity in a meta‐analysis. Stat Med 2002; 21: 1539–1558.
Kelley K, Preacher KJ . On effect size. Psychol Methods 2012; 17: 137.
Moran VH, Skinner A-L, Medina MW, Patel S, Dykes F, Souverein OW et al. The relationship between zinc intake and serum/plasma zinc concentration in pregnant and lactating women: a systematic review with dose-response meta-analyses. J Trace Elem Med Biol 2012; 26: 74–79.
Moran VH, Stammers A-L, Medina MW, Patel S, Dykes F, Souverein OW et al. The relationship between zinc intake and serum/plasma zinc concentration in children: a systematic review and dose-response meta-analysis. Nutrients 2012; 4: 841–858.
Lowe NM, Medina MW, Stammers A-L, Patel S, Souverein OW, Dullemeijer C et al. The relationship between zinc intake and serum/plasma zinc concentration in adults: a systematic review and dose–response meta-analysis by the EURRECA Network. Br J Nutr 2012; 108: 1962–1971.
Nissensohn M, Sanchez Villegas A, Fuentes Lugo D, Henriquez Sanchez P, Doreste Alonso J, Lowe NM et al. Effect of zinc intake on serum/plasma zinc status in infants: a meta-analysis. Maternal Child Nutr 2013; 9: 285–298.
Umamaheswari K, Bhaskaran M, Krishnamurthy G, Hemamalini, Vasudevan K . Effect of iron and zinc deficiency on short term memory in children. Indian Pediatr 2011; 48: 289–293.
Tupe RP, Chiplonkar SA . Zinc supplementation improved cognitive performance and taste acuity in Indian adolescent girls. J Am Coll Nutr 2009; 28: 388–396.
Lam PK, Kritz-Silverstein D, Barrett-Connor E, Milne D, Nielsen F, Gamst A et al. Plasma trace elements and cognitive function in older men and women: the Rancho Bernardo study. J Nutr Health Aging 2008; 12: 22–27.
Ortega RM, Requejo AM, Andres P, LopezSobaler AM, Quintas ME, Redondo MR et al. Dietary intake and cognitive function in a group of elderly people. Am J Clin Nutr 1997; 66: 803–809.
Gao S, Jin Y, Unverzagt FW, Ma F, Hall KS, Murrell JR et al. Trace element levels and cognitive function in rural elderly Chinese. J Gerontol A Biol Sci Med Sci 2008; 63: 635–641.
Stoecker BJ, Abebe Y, Hubbs-Tait L, Kennedy TS, Gibson RS, Arbide I et al. Zinc status and cognitive function of pregnant women in Southern Ethiopia. Eur J Clin Nutr 2009; 63: 916–918.
Maylor EA, Simpson EEA, Secker DL, Meunier N, Andriollo-Sanchez M, Polito A et al. Effects of zinc supplementation on cognitive function in healthy middle-aged and older adults: the ZENITH study. Br J Nutr 2006; 96: 752–760.
Murray-Kolb LE, Khatry SK, Katz J, Schaefer BA, Cole PM, Le Clerq SC et al. Preschool micronutrient supplementation effects on intellectual and motor function in school-aged nepalese children. Arch Pediatr Adolesc Med 2012; 166: 404–410.
Pongcharoen T, Ramakrishnan U, Di Girolamo AM, Winichagoon P, Flores R, Singkhornard J et al. Influence of prenatal and postnatal growth on intellectual functioning in school-aged children. Arch Pediatr Adolesc Med 2012; 166: 411–416.
Gewa CA, Weiss RE, Bwibo NO, Whaley S, Sigman M, Murphy SP et al. Dietary micronutrients are associated with higher cognitive function gains among primary school children in rural Kenya. Br J Nutr 2009; 101: 1378–1387.
Christian P, Murray-Kolb LE, Khatry SK, Katz J, Schaefer BA, Cole PM et al. Prenatal micronutrient supplementation and intellectual and motor function in early school-aged children in Nepal. JAMA 2010; 304: 2716–2723.
Caulfield LE, Putnick DL, Zavaleta N, Lazarte F, Albornoz C, Chen P et al. Maternal gestational zinc supplementation does not influence multiple aspects of child development at 54 mo of age in Peru. Am J Clin Nutr 2010; 92: 130–136.
Penland JG, Sandstead HH, Alcock NW, Dayal HH, Chen XC, Li JS et al. A preliminary report: effects of zinc and micronutrient repletion on growth and neuropsychological function of urban Chinese children. J Am Coll Nutr 1997; 16: 268–272.
Sandstead HH, Penland JG, Alcock NW, Hari H, Xue D, Chen C et al. Effects of repletion with zinc and other micronutrients on neuropsychologic performance and growth of Chinese children. Am J Clin Nutr 1998; 68: 470S–475S.
Gibson RS, Smit Vanderkooy PD, MacDonald AC, Goldman A, Ryan BA, Berry M . A growth-limiting, mild zinc-deficiency syndrome in some southern Ontario boys with low height percentiles. Am J Clin Nutr 1989; 49: 1266–1273.
Tamura T, Goldenberg RL, Ramey SL, Nelson KG, Chapman VR . Effect of zinc supplementation of pregnant women on the mental and psychomotor development of their children at 5 y of age. Am J Clin Nutr 2003; 77: 1512–1516.
Cavan KR, Gibson RS, Grazioso CF, Isalgue AM, Ruz M, Solomons NW . Growth and body composition of periurban Guatemalan children in relation to zinc status: a longitudinal zinc intervention trial. Am J Clin Nutr 1993; 57: 344–352.
Hubbs-Tait L, Kennedy TS, Droke EA, Belanger DM, Parker JR . Zinc, iron, and lead: relations to head start children's cognitive scores and teachers' ratings of behavior. J Am Diet Assoc 2007; 107: 128–133.
Kaye DB, Baron MB . The validity of the Detroit test of learning aptitude. J Psychoeduc Assess 1984; 2: 117–124.
Stanley K . Evaluation of randomized controlled trials. Circulation 2007; 115: 1819–1822.
Gogia S, Sachdev HS . Zinc supplementation for mental and motor development in children. Cochrane Database Syst Rev 2012; 12: CD007991-CD.
Brown KH, Peerson JM, Baker SK, Hess SY . Preventive zinc supplementation among infants, preschoolers, and older prepubertal children. Food Nutr Bull 2009; 30: S12–S40.
Best C, Neufingerl N, Del Rosso JM, Transler C, van den Briel T, Osendarp S . Can multi-micronutrient food fortification improve the micronutrient status, growth, health, and cognition of schoolchildren? A systematic review. Nutr Rev 2011; 69: 186–204.
Eilander A, Gera T, Sachdev HS, Transler C, van der Knaap HCM, Kok FJ et al. Multiple micronutrient supplementation for improving cognitive performance in children: systematic review of randomized controlled trials. Am J Clin Nutr 2010; 91: 115–130.
Nyaradi A, Li J, Hickling S, Foster J, Oddy WH . The role of nutrition in children's neurocognitive development, from pregnancy through childhood. Front Hum Neurosci 2013; 7.
Schmitt JA . Nutrition and cognition: meeting the challenge to obtain credible and evidence-based facts. Nutr Rev 2010; 68: S2–S5.
Wood RJ . Assessment of marginal zinc status in humans. J Nutr 2000; 130: 1350S–1354S.
Lowe NM, Fekete K, Decsi T . Methods of assessment of zinc status in humans: a systematic review. Am J Clin Nutr 2009; 89: 2040S–2051S.
Hambidge M . Biomarkers of trace mineral intake and status. J Nutr 2003; 133: 948S–955S.
Sandstead HH, Prasad AS, Penland JG, Beck FW, Kaplan J, Egger NG et al. Zinc deficiency in Mexican American children: influence of zinc and other micronutrients on T cells, cytokines, and antiinflammatory plasma proteins. Am J Clin Nutr [Internet] 2008 13.02.09 [cited MN 88: 1067–1073. Available from: http://www.mrw.interscience.wiley.com/cochrane/clcentral/articles/360/CN-00651360/frame.html.
Acknowledgements
The original conception of the systematic review was undertaken by the EURRECA network and coordinated by partners based at Wageningen University (WU), the Netherlands and the University of East Anglia (UEA), United Kingdom, Susan Fairweather-Tait (UEA), Lisette de Groot (WU), Pieter van’t Veer (WU), Kate Ashton (UEA), Amélie Casgrain (UEA), Adriënne Cavelaars (WU), Rachel Collings (UEA), Rosalie Dhonukshe-Rutten (WU), Esmée Doets (WU), Linda Harvey (UEA) and Lee Hooper (UEA) designed and developed the review protocol and search strategy. We thank the EURRECA Network of Excellence and to Sujata Patel, Joseph Saavedra, Nick Kenworthy, Sarah Richardson-Owen and Christine Cockburn for assistance with screening, data extraction of studies and Fiona Dykes for helpful discussions. NML, MW-M, A-LS, VHM, PQ and SD collected and analysed the data. LS-M and MN were also involved in the screening process. All authors were involved in writing the manuscript. We like to acknowledge networking support by Zn-Net COST Action TD1304, The Network for the Biology of Zinc, (http://www.cost.eu/COST_Actions/fa/Actions/TD1304). Names for PubMed indexing: Warthon-Medina, Hall Moran, Stammers, Dillon, Qualter, Nissenhohn, Serra-Majem, Lowe. This study has been supported by the EURRECA Network of Excellence (http://www.eurreca.org), which was funded by the Commission of the European Communities, specific Research, Technology and Development (RTD) Programme Quality of Life and Management of Living Resources, within the Sixth Framework Programme, contract no. 036196. Member of the Zinc-Net COST Action TD1304, http://www.cost.eu/domains_actions/fa/Actions/TD1304. This report does not necessarily reflect the Commission’s views or its future policy in this area.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Warthon-Medina, M., Moran, V., Stammers, AL. et al. Zinc intake, status and indices of cognitive function in adults and children: a systematic review and meta-analysis. Eur J Clin Nutr 69, 649–661 (2015). https://doi.org/10.1038/ejcn.2015.60
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2015.60
This article is cited by
-
Higher intake of certain nutrients among older adults is associated with better cognitive function: an analysis of NHANES 2011–2014
BMC Nutrition (2023)
-
Exposure to Heavy Metals and Allergic Outcomes in Children: a Systematic Review and Meta-analysis
Biological Trace Element Research (2022)
-
Serum Copper and Zinc Concentrations and Cognitive Impairment in Older Adults Aged 60 Years and Older
Biological Trace Element Research (2022)
-
Strategies to evaluate outcomes in long-COVID-19 and post-COVID survivors
Infectious Agents and Cancer (2021)
-
Role of Zinc in Neonatal Sepsis
Indian Journal of Pediatrics (2021)