Abstract
Background/Objectives:
Vascular endothelial function declines with advancing age, due in part to increased oxidative stress and inflammation, and this age-related vascular dysfunction has been identified as an independent risk factor for cardiovascular diseases. This double-blind, placebo-controlled trial investigated the effects of a dietary supplement containing β-hydroxy-β-methylbutyrate (HMB), glutamine and arginine on endothelial-dependent vasodilation of older adults.
Subjects/Methods:
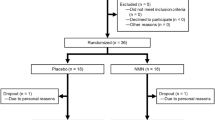
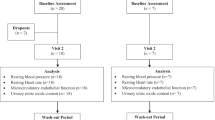
A total of 31 community-dwelling men and women aged 65–87 years were randomly assigned to two groups. The treatment group received two doses of the supplement daily (totaling 3 g HMB, 14 g glutamine and 14 g arginine) for 6 months, whereas the control group received an isocaloric placebo. At baseline and week 24, vascular endothelial function was measured by flow-mediated dilation of the brachial artery, and fasting blood samples were obtained to measure high-sensitivity C-reactive protein (hsCRP) and tumor necrosis factor-α (TNF-α).
Results:
Paired sample t-tests revealed a 27% increase in flow-mediated dilation among the treatment group (P=0.003), whereas no change was observed in the placebo group (P=0.651). Repeated-measures analysis of variance verified a significant time by group interaction (P=0.038). Although no significant changes were observed for hsCRP or TNF-α, a trend was observed for increasing hsCRP among the placebo group only (P=0.059).
Conclusions:
These results suggest that dietary supplementation of HMB, glutamine and arginine may favorably affect vascular endothelial function in older adults. Additional studies are needed to elucidate whether reduced inflammation or other mechanisms may underlie the benefits of supplementation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Lee HY, Oh BH . Aging and arterial stiffness. Circ J 2010; 74: 2257–2262.
Brandes R, Fleming I, Busse R . Endothelial aging. Cardiovasc Res 2005; 66: 286–294.
Yavuz B, Yavuz B, Sener D, Cankurtaran M, Halil M, Ulger Z et al. Advanced age is associated with endothelial dysfunction in healthy elderly subjects. Gerontology 2008; 54: 153–156.
Fitschen PJ, Wilson GJ, Wilson JM, Wilund KR . Efficacy of β-hydroxy-β-methylbutyrate supplementation in elderly and clinical populations. Nutrition 2013; 29: 29–36.
Hsieh LC, Chien SL, Huang MS, Tseng HF, Chang CK . Anti-inflammatory and anticatabolic effects of short-term beta-hydroxy-beta-methylbutyrate supplementation on chronic obstructive pulmonary disease patients in intensive care unit. Asia Pac J Clin Nutr 2006; 15: 544–550.
Townsend JR, Fragala MS, Jajtner AR, Gonzalez AM, Wells AJ, Mangine GT et al. β-Hydroxy-β-methylbutyrate (HMB)-free acid attenuates circulating TNF-α and TNFR1 expression postresistance exercise. J Appl Physiol (1985) 2013; 115: 1173–1182.
Hao Y, Jackson JR, Wang Y, Edens N, Pereira SL, Alway SE . β-Hydroxy-β-methylbutyrate reduces myonuclear apoptosis during recovery from hind limb suspension-induced muscle fiber atrophy in aged rats. Am J Physiol Regul Integr Comp Physiol 2011; 301: R701–R715.
Eley H, Russell S, Tisdale M . Attenuation of depression of muscle protein synthesis induced by lipopolysaccharide, tumor necrosis factor, and angiotensin II by beta-hydroxy-beta-methylbutyrate. Am J Physiol Endocrinol Metab 2008; 295: E1409–E1416.
Eley H, Russell S, Tisdale M . Mechanism of attenuation of muscle protein degradation induced by tumor necrosis factor-alpha and angiotensin II by beta-hydroxy-beta-methylbutyrate. Am J Physiol Endocrinol Metab 2008; 295: E1417–E1426.
Russell ST, Tisdale MJ . Mechanism of attenuation by beta-hydroxy-beta-methylbutyrate of muscle protein degradation induced by lipopolysaccharide. Mol Cell Biochem 2009; 330: 171–179.
Nissen S, Sharp R, Panton L, Vukovich M, Trappe S, Fuller JJ . beta-hydroxy-beta-methylbutyrate (HMB) supplementation in humans is safe and may decrease cardiovascular risk factors. J Nutr 2000; 130: 1937–1945.
Melis G, ter Wengel N, Boelens P, van Leeuwen P . Glutamine: recent developments in research on the clinical significance of glutamine. Curr Opin Clin Nutr Metab Care 2004; 7: 59–70.
Appleton J . Arginine: clinical potential of a semi-essential amino. Altern Med Rev 2002; 7: 512–522.
Tong B, Barbul A . Cellular and physiological effects of arginine. Mini Rev Med Chem 2004; 4: 823–832.
Williams J, Abumrad N, Barbul A . Effect of a specialized amino acid mixture on human collagen deposition. Ann Surg 2002; 236: 369–374. discussion 374-365.
Berk L, James J, Schwartz A, Hug E, Mahadevan A, Samuels M et al. A randomized, double-blind, placebo-controlled trial of a beta-hydroxyl beta-methyl butyrate, glutamine, and arginine mixture for the treatment of cancer cachexia (RTOG 0122). Support Care Cancer 2008; 16: 1179–1188.
May P, Barber A, D'Olimpio J, Hourihane A, Abumrad N . Reversal of cancer-related wasting using oral supplementation with a combination of beta-hydroxy-beta-methylbutyrate, arginine, and glutamine. Am J Surg 2002; 183: 471–479.
Marcora S, Lemmey A, Maddison P . Dietary treatment of rheumatoid cachexia with beta-hydroxy-beta-methylbutyrate, glutamine and arginine: a randomised controlled trial. Clin Nutr 2005; 24: 442–454.
Clark R, Feleke G, Din M, Yasmin T, Singh G, Khan F et al. Nutritional treatment for acquired immunodeficiency virus-associated wasting using beta-hydroxy beta-methylbutyrate, glutamine, and arginine: a randomized, double-blind, placebo-controlled study. JPEN J Parenter Enteral Nutr 2000; 24: 133–139.
Harnack L, Stevens M, Van Heel N, Schakel S, Dwyer JT, Himes J . A computer-based approach for assessing dietary supplement use in conjunction with dietary recalls. J Food Compost Anal 2008; 21: S78–S82.
Bianchini E, Faita F, Gemignani V, Giannoni M, Demi M . The assessment of flow-mediated dilation (FMD) of the brachial artery. Comp Cardiol 2006; 33: 509–512.
Corretti M, Anderson T, Benjamin E, Celermajer D, Charbonneau F, Creager M et al. Guidelines for the ultrasound assessment of endothelial-dependent flow-mediated vasodilation of the brachial artery: a report of the International Brachial Artery Reactivity Task Force. J Am Coll Cardiol 2002; 39: 257–265.
Seals DR, Kaplon RE, Gioscia-Ryan RA, LaRocca TJ . You're only as old as your arteries: translational strategies for preserving vascular endothelial function with aging. Physiology (Bethesda) 2014; 29: 250–264.
Singh T, Newman AB . Inflammatory markers in population studies of aging. Ageing Res Rev 2011; 10: 319–329.
Roth E . Nonnutritive effects of glutamine. J Nutr 2008; 138: 2025S–2031S.
Ligthart-Melis GC, van de Poll MC, Boelens PG, Dejong CH, Deutz NE, van Leeuwen PA . Glutamine is an important precursor for de novo synthesis of arginine in humans. Am J Clin Nutr 2008; 87: 1282–1289.
Potenza M, Nacci C, Mitolo-Chieppa D . Immunoregulatory effects of L-arginine and therapeutical implications. Curr Drug Targets Immune Endocr Metabol Disord 2001; 1: 67–77.
Preli RB, Klein KP, Herrington DM . Vascular effects of dietary L-arginine supplementation. Atherosclerosis 2002; 162: 1–15.
Bode-Böger S, Muke J, Surdacki A, Brabant G, Böger R, Frölich J . Oral L-arginine improves endothelial function in healthy individuals older than 70 years. Vasc Med 2003; 8: 77–81.
Monti LD, Casiraghi MC, Setola E, Galluccio E, Pagani MA, Quaglia L et al. L-arginine enriched biscuits improve endothelial function and glucose metabolism: a pilot study in healthy subjects and a cross-over study in subjects with impaired glucose tolerance and metabolic syndrome. Metabolism 2013; 62: 255–264.
Jahangir E, Vita J, Handy D, Holbrook M, Palmisano J, Beal R et al. The effect of L-arginine and creatine on vascular function and homocysteine metabolism. Vasc Med 2009; 14: 239–248.
Donato AJ, Gano LB, Eskurza I, Silver AE, Gates PE, Jablonski K et al. Vascular endothelial dysfunction with aging: endothelin-1 and endothelial nitric oxide synthase. Am J Physiol Heart Circ Physiol 2009; 297: H425–H432.
Chirinos JA . Arterial stiffness: basic concepts and measurement techniques. J Cardiovasc Transl Res 2012; 5: 243–255.
Barodka VM, Joshi BL, Berkowitz DE, Hogue CW, Nyhan D . Review article: implications of vascular aging. Anesth Analg 2011; 112: 1048–1060.
Acknowledgements
This work was funded by the National Center for Complementary and Alternative Medicine (F31 AT005384-01). Core laboratory support and facilities were provided by the UAB Diabetes Research Center Human Physiology Core (P30DK079626-06). Abbott Nutrition provided supplement and placebo in coded packets.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on European Journal of Clinical Nutrition website
Supplementary information
Rights and permissions
About this article
Cite this article
Ellis, A., Patterson, M., Dudenbostel, T. et al. Effects of 6-month supplementation with β-hydroxy-β-methylbutyrate, glutamine and arginine on vascular endothelial function of older adults. Eur J Clin Nutr 70, 269–273 (2016). https://doi.org/10.1038/ejcn.2015.137
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2015.137
This article is cited by
-
Evaluation of the recovery after heart surgery following preoperative supplementation with a combination of beta-hydroxy-beta-methylbutyrate, l-arginine, and l-glutamine: a double-blind randomized placebo-controlled clinical trial
Trials (2022)
-
Blood biomarkers indicate that the preclinical stages of Alzheimer's disease present overlapping molecular features
Scientific Reports (2020)
-
Effects of an L-arginine-based multi ingredient product on endothelial function in subjects with mild to moderate hypertension and hyperhomocysteinemia - a randomized, double-blind, placebo-controlled, cross-over trial
BMC Complementary and Alternative Medicine (2017)
-
Comprehensive metabolic profiling of chronic low-grade inflammation among generally healthy individuals
BMC Medicine (2017)