Abstract
Background:
Sarcopenia is defined as the loss of skeletal muscle mass and quality, which accelerates with aging and is associated with functional decline. Rising obesity prevalence has led to a high-risk group with both disorders. We assessed mortality risk associated with sarcopenia and sarcopenic obesity in elders.
Methods:
A subsample of 4652 subjects ⩾60 years of age was identified from the National Health and Nutrition Examination Survey III (1988–1994), a cross-sectional survey of non-institutionalized adults. National Death Index data were linked to this data set. Sarcopenia was defined using a bioelectrical impedance formula validated using magnetic resonance imaging-measured skeletal mass by Janssen et al. Cutoffs for total skeletal muscle mass adjusted for height2 were sex-specific (men: ⩽5.75 kg/m2; females ⩽10.75 kg/m2). Obesity was based on % body fat (males: ⩾27%, females: ⩾38%). Modeling assessed mortality adjusting for age, sex, ethnicity (model 1), comorbidities (hypertension, diabetes, congestive heart failure, osteoporosis, cancer, coronary artery disease and arthritis), smoking, physical activity, self-reported health (model 2) and mobility limitations (model 3).
Results:
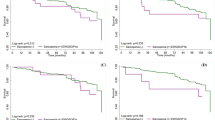
Mean age was 70.6±0.2 years and 57.2% were female. Median follow-up was 14.3 years (interquartile range: 12.5–16.1). Overall prevalence of sarcopenia was 35.4% in women and 75.5% in men, which increased with age. Prevalence of obesity was 60.8% in women and 54.4% in men. Sarcopenic obesity prevalence was 18.1% in women and 42.9% in men. There were 2782 (61.7%) deaths, of which 39.0% were cardiovascular. Women with sarcopenia and sarcopenic obesity had a higher mortality risk than those without sarcopenia or obesity after adjustment (model 2, hazard ratio (HR): 1.35 (1.05–1.74) and 1.29 (1.03–1.60)). After adjusting for mobility limitations (model 3), sarcopenia alone (HR: 1.32 ((1.04–1.69) but not sarcopenia with obesity (HR: 1.25 (0.99–1.58)) was associated with mortality. For men, the risk of death with sarcopenia and sarcopenic obesity was nonsignificant in both model-2 (HR: 0.98 (0.77–1.25), and HR: 0.99 (0.79–1.23)) and model 3 (HR: 0.98 (0.77–1.24) and HR: 0.98 (0.79–1.22)).
Conclusions:
Older women with sarcopenia have an increased all-cause mortality risk independent of obesity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Gallagher D, Visser M, Sepulveda D, Pierson RN, Harris T, Heymsfield SB . How useful is body mass index for comparison of body fatness across age, sex, and ethnic groups? Am J Epidemiol 1996; 143: 228–239.
Smalley KJ, Knerr AN, Kendrick ZV, Colliver JA, Owen OE . Reassessment of body mass indices. Am J Clin Nutr 1990; 52: 405–408.
Alley DE, Chang VW . The changing relationship of obesity and disability, 1988-2004. JAMA 2007; 298: 2020–2027.
Baumgartner RN, Wayne SJ, Waters DL, Janssen I, Gallagher D, Morley JE . Sarcopenic obesity predicts instrumental activities of daily living disability in the elderly. Obesity Res 2004; 12: 1995–2004.
Cruz-Jentoft AJ, Baeyens JP, Bauer JM, Boirie Y, Cederholm T, Landi F et al. Sarcopenia: European consensus on definition and diagnosis. Age Ageing 2010; 39: 412–423.
Flegal KM, Graubard BI, Williamson DF, Gail MH . Excess deaths associated with underweight, overweight, and obesity. JAMA 2005; 293: 1861–1867.
Gale CR, Martyn CN, Cooper C, Sayer AA Grip strength, body composition, and mortality. Int J Epidemiol 2007; 36: 228–235.
Lang IA, Llewellyn DJ, Alexander K, Melzer D . Obesity, physical function, and mortality in older adults. J Am Geriatr Soc 2008; 56: 1474–1478.
Prospective Studies C, Whitlock G, Lewington S, Sherliker P, Clarke R, Emberson J et al. Body-mass index and cause-specific mortality in 900 000 adults: collaborative analyses of 57 prospective studies. Lancet 2009; 373: 1083–1096.
Villareal DT, Banks M, Siener C, Sinacore DR, Klein S . Physical frailty and body composition in obese elderly men and women. Obes Res 2004; 12: 913–920.
Batsis JA, Barre LK, Mackenzie TA, Pratt SI, Lopez-Jimenez F, Bartels SJ . Variation in the prevalence of sarcopenia and sarcopenic obesity in older adults associated with different research definitions: dual-energy x-ray absorptiometry data from the national health and nutrition examination survey 1999-2004. J Am Geriatr Soc 2013; 61: 974–980.
Flegal KM, Carroll MD, Kit BK, Ogden CL . Prevalence of obesity and trends in the distribution of body mass index among US adults, 1999-2010. JAMA 2012; 307: 491–497.
Lean ME, Katsarou C, McLoone P, Morrison DS . Changes in BMI and waist circumference in Scottish adults: use of repeated cross-sectional surveys to explore multiple age groups and birth-cohorts. Int J Obes (Lond) 2013; 37: 800–808.
Gregg EW, Cheng YJ, Cadwell BL, Imperatore G, Williams DE, Flegal KM et al. Secular trends in cardiovascular disease risk factors according to body mass index in US adults. JAMA 2005; 293: 1868–1874.
Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care 1997; 20: 1183–1197.
Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF 3rd, Feldman HI et al. A new equation to estimate glomerular filtration rate. Ann Intern Med 2009; 150: 604–612.
Jette AM, Branch LG . Impairment and disability in the aged. J Chronic Dis 1985; 38: 59–65.
Chumlea WC, Guo SS, Kuczmarski RJ, Flegal KM, Johnson CL, Heymsfield SB et al. Body composition estimates from NHANES III bioelectrical impedance data. Int J Obes Relat Metab Disord 2002; 26: 1596–1609.
Janssen I, Heymsfield SB, Ross R . Low relative skeletal muscle mass (sarcopenia) in older persons is associated with functional impairment and physical disability. J Am Geriatr Soc 2002; 50: 889–896.
Janssen I, Heymsfield SB, Baumgartner RN, Ross R . Estimation of skeletal muscle mass by bioelectrical impedance analysis. J Appl Physiol 2000; 89: 465–471.
Janssen I . Skeletal muscle cutpoints associated with elevated physical disability risk in older men and women. Am J Epidemiol 2004; 159: 413–421.
Lim S, Kim JH, Yoon JW, Kang SM, Choi SH, Park YJ et al. Sarcopenic obesity: prevalence and association with metabolic syndrome in the korean longitudinal study on health and aging (KLoSHA). Diabetes Care 2010; 33: 1652–1654.
Newman AB, Kupelian V, Visser M, Simonsick E, Goodpaster B, Nevitt M et al. Sarcopenia: alternative definitions and associations with lower extremity function. J Am Geriatr Soc 2003; 51: 1602–1609.
Rolland Y, Lauwers-Cances V, Cristini C, Abellan van Kan G, Janssen I, Morley JE et al. Difficulties with physical function associated with obesity, sarcopenia, and sarcopenic-obesity in community-dwelling elderly women: the EPIDOS (EPIDemiologie de l'OSteoporose) study. Am J Clin Nutr 2009; 89: 1895–1900.
Zamboni M, Mazzali G, Fantin F, Rossi A, Di Francesco V . Sarcopenic obesity: a new category of obesity in the elderly. Nutr Metab Cardiovasc Dis 2008; 18: 388–395.
Zoico E, Di Francesco V, Guralnik JM, Mazzali G, Bortolani A, Guariento S et al. Physical disability and muscular strength in relation to obesity and different body composition indexes in a sample of healthy elderly women. Int J Obes 2004; 28: 234–241.
Batsis JA, Sahakyan KR, Rodriguez-Escudero JP, Bartels SJ, Somers VK, Lopez-Jimenez F . Normal weight obesity and mortality in United States subjects >/=60 years of age (from the Third National Health and Nutrition Examination Survey). Am J Cardiol 2013; 112: 1592–1598.
Stenholm S, Sainio P, Rantanen T, Alanen E, Koskinen S . Effect of co-morbidity on the association of high body mass index with walking limitation among men and women aged 55 years and older. Aging Clin Exp Res 2007; 19: 277–283.
Visser M, Kritchevsky SB, Goodpaster BH, Newman AB, Nevitt M, Stamm E et al. Leg muscle mass and composition in relation to lower extremity performance in men and women aged 70 to 79: the health, aging and body composition study. J Am Geriatr Soc 2002; 50: 897–904.
Guralnik JM, Ferrucci L, Pieper CF, Leveille SG, Markides KS, Ostir GV et al. Lower extremity function and subsequent disability: consistency across studies, predictive models, and value of gait speed alone compared with the short physical performance battery. J Gerontol A Biol Sci Med Sci 2000; 55: M221–M231.
Rantanen T, Guralnik JM, Foley D, Masaki K, Leveille S, Curb JD et al. Midlife hand grip strength as a predictor of old age disability. JAMA 1999; 281: 558–560.
Arango-Lopera VE, Arroyo P, Gutierrez-Robledo LM, Perez-Zepeda MU, Cesari M . Mortality as an adverse outcome of sarcopenia. J Nutr Health Aging 2013; 17: 259–262.
Cesari M, Pahor M, Lauretani F, Zamboni V, Bandinelli S, Bernabei R et al. Skeletal muscle and mortality results from the InCHIANTI Study. J Gerontol A Biol Sci Med Sci 2009; 64: 377–384.
Landi F, Cruz-Jentoft AJ, Liperoti R, Russo A, Giovannini S, Tosato M et al. Sarcopenia and mortality risk in frail older persons aged 80 years and older: results from ilSIRENTE study. Age Ageing 2013; 42: 203–209.
Landi F, Liperoti R, Fusco D, Mastropaolo S, Quattrociocchi D, Proia A et al. Sarcopenia and mortality among older nursing home residents. J Am Med Dir Assoc 2012; 13: 121–126.
Newman AB, Kupelian V, Visser M, Simonsick EM, Goodpaster BH, Kritchevsky SB et al. Strength, but not muscle mass, is associated with mortality in the health, aging and body composition study cohort. J Gerontol A Biol Sci Med Sci 2006; 61: 72–77.
Stenholm S, Harkanen T, Sainio P, Heliovaara M, Koskinen S . Long-term changes in handgrip strength in men and women–accounting the effect of right censoring due to death. J Gerontol A Biol Sci Med Sci 2012; 67: 1068–1074.
Bunout D, de la Maza MP, Barrera G, Leiva L, Hirsch S . Association between sarcopenia and mortality in healthy older people. Australas J Ageing 2011; 30: 89–92.
Miller BS, Ignatoski KM, Daignault S, Lindland C, Doherty M, Gauger PG et al. Worsening central sarcopenia and increasing intra-abdominal fat correlate with decreased survival in patients with adrenocortical carcinoma. World J Surg 2012; 36: 1509–1516.
Villasenor A, Ballard-Barbash R, Baumgartner K, Baumgartner R, Bernstein L, McTiernan A et al. Prevalence and prognostic effect of sarcopenia in breast cancer survivors: the HEAL Study. J Cancer Surviv 2012; 6: 398–406.
Batsis JA, Nieto-Martinez RE, Lopez-Jimenez F . Metabolic syndrome: from global epidemiology to individualized medicine. Clin Pharmacol Ther 2007; 82: 509–524.
Deurenberg P, Deurenberg Yap M, Wang J, Lin FP, Schmidt G . The impact of body build on the relationship between body mass index and percent body fat. Int J Obes Relat Metab Disord 1999; 23: 537–542.
Deurenberg P, Deurenberg-Yap M . Differences in body-composition assumptions across ethnic groups: practical consequences. Curr Opin Clin Nutr Metab Care 2001; 4: 377–383.
Roubenoff R, Dallal GE, Wilson PW . Predicting body fatness: the body mass index vs estimation by bioelectrical impedance. Am J Public Health 1995; 85: 726–728.
Diniz Araujo ML, Coelho Cabral P, Kruze Grande de Arruda I, Siqueira Tavares Falcao AP, Silva Diniz A . Body fat assessment by bioelectrical impedance and its correlation with anthropometric indicators. Nutr Hosp 2012; 27: 1999–2005.
Acknowledgements
This project was funded by the Centers for Aging, The Dartmouth Institute and the Department of Medicine, Dartmouth-Hitchcock Medical Center.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
This work was presented in part at the American Geriatrics Society's Annual Meeting, May 2012, Seattle, WA, USA.
Rights and permissions
About this article
Cite this article
Batsis, J., Mackenzie, T., Barre, L. et al. Sarcopenia, sarcopenic obesity and mortality in older adults: results from the National Health and Nutrition Examination Survey III. Eur J Clin Nutr 68, 1001–1007 (2014). https://doi.org/10.1038/ejcn.2014.117
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2014.117
This article is cited by
-
Association of physical functional activity impairment with severity of sarcopenic obesity: findings from National Health and Nutrition Examination Survey
Scientific Reports (2024)
-
Subcutaneous adipose tissue alteration in aging process associated with thyroid hormone signaling
BMC Medical Genomics (2023)
-
The effect of sarcopenia and sarcopenic obesity on survival in gastric cancer
BMC Cancer (2023)
-
Effects of preoperative sarcopenia-related parameters on the musculoskeletal and metabolic outcomes after bariatric surgery: a one-year longitudinal study in females
Scientific Reports (2023)
-
Moracin E and M isolated from Morus alba Linné induced the skeletal muscle cell proliferation via PI3K-Akt-mTOR signaling pathway
Scientific Reports (2023)