Abstract
Background/Objectives:
Little is known about the long-term effects of maternal multi-micronutrient supplementation on the growth of children during early childhood. In this follow-up study, the effects of maternal supplementation with multi-micronutrients in pregnancy on postnatal child growth are examined.
Subjects/Methods:
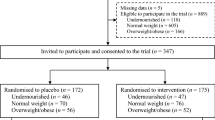
A longitudinal follow-up of a subset of newborns (n=1388) whose mothers were randomly assigned to receive the supplements of folic acid, iron–folic acid or multi-micronutrients daily during pregnancy in the original trial was conducted. Children’s weight and length were measured and assessed during monthly home visits from birth to 30 months of age.
Results:
The pooled prevalence rate of stunting over different time points during the first 30 months was 13.5, 14.9 and 12.1% for the folic acid group, iron–folic acid group and multi-micronutrient group, respectively. However, there were no significant differences in the pooled odds of stunting in children between the multi-micronutrient group and the folic acid (odds ratio (OR) 0.97, 95% confidence interval (CI): 0.74–1.26), and between the multi-micronutrient group and the iron–folic acid group (OR 0.82, 95% CI: 0.63–1.07). Similar results for the three groups were found for the occurrences of underweight and wasting in children. Furthermore, no significant differences were observed in length, weight, length-for-age, weight-for-age and weight-for-length Z-scores among the three treatment groups.
Conclusions:
Currently, available evidence is insufficient to support a greater advantage of the effect of maternal multi-micronutrient supplementation on child growth over iron–folic acid or folic acid only supplementation during the first 30 months.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Allen LH (2000). Anemia and iron deficiency: effects on pregnancy outcome. Am J Clin Nutr 71 (Suppl), 1280S–1284S.
Christian P, Khatry SK, Katz J, Pradhan EK, LeClerq S, Shrestha SR et al. (2003). Effects of alternative maternal micronutrient supplements on low birth weight in rural Nepal: double blind randomised community trial. BMJ 326, 571–576.
de Onis M, Garza C, Onyango AW, Martorell R (eds) (2006). WHO child growth standards. Acta Paediatrics 95 (Suppl 450), S1–S101.
Filmer D, Pritchett L (2001). Estimating wealth effects without expenditure data-or tears: an application to educational enrollments in states of India. Demography 38, 115–132.
Huy ND, Le Hop T, Shrimpton R, Hoa CV (2009). An effectiveness trial of multiple micronutrient supplementation during pregnancy in Vietnam: impact on birthweight and on stunting in children at around 2 years of age. Food Nutr Bull 30 (4 Suppl), S506–S516.
Jiang T, Christian P, Khatry SK, Wu L, West KP (2005). Micronutrient deficiencies in early pregnancy are common, concurrent, and vary by season among rural Nepali pregnant women. J Nutr 135, 1106–1112.
Juarez-Vazquez J, Bonizzoni E, Scotti A (2002). Iron plus folate is more effective than iron alone in the treatment of iron deficiency anemia in pregnancy: a randomized, double blind clinical trial. BJOG 109, 1009–1014.
Kawai K, Spiegelman D, Shankar AH, Fawzi WW (2011). Maternal multiple micronutrient supplemen pregnancy outcomes in developing countrie and meta-regression. Bull World Health Organ 89, 402–411.
Kontic-Vucinic O, Sulovic N, Radunovic N (2006). Micronutrients in women's reproductive health: I. Vitamins. Int J Fertil Womens Med 51, 106–115.
Li Q, Yan H, Zeng L, Cheng Y, Liang W, Dang S et al. (2009). Effects of maternal multimicronutrient supplementation on the mental development of infants in rural western China: follow-up evaluation of a double-blind, randomised, controlled trial. Pediatrics 123, 685–692.
Lindblad B, Zaman S, Malik A, Martin H, Ekström AM, Amu S et al. (2005). Folate, vitamin B12, and homocysteine levels in South Asian women with growth-retarded fetuses. Acta Obstet Gynecol Scand 84, 1055–1061.
Lingxia Z, Hong Y, Xiong G, Shaonong D, Hong X (2003). Analysis on malnutrition of children under 3 years old in 40 poor counties in the western areas of China. Chin J Public Health 19, 55–58.
Ma A, Chen X, Wang Y, Xu R, Zheng M, Li J (2004). The multiple vitamin status of Chinese pregnant women with anemia and nonanemia in the last trimester. J Nutr Sci Vitaminol (Tokyo) 50, 87–92.
Molloy AM, Kirke PN, Brody LC, Scott JM, Mills JL (2008). Effects of folate and vitamin B12 deficiencies during pregnancy on fetal, infant, and child development. Food Nutr Bull 29 (2 Suppl), S101–S111.
Mu L, Lingxia Z, Jinping W, Lihua C, Yanming L, Haili L et al. (2005). Analysis of prevalence study on maternity and child care in the experimental units of new-type rural co-medical treatment of Shaanxi province. J Xi’an Jiaotong Univ Med Sci 26, 392–394.
Pathak P, Kapil U, Kapoor SK, Saxena R, Kumar A, Gupta N et al. (2004). Prevalence of multiple micronutrient deficiencies amongst pregnant women in a rural area of Haryana. Indian J Pediatr 71, 1007–1014.
Pathak P, Kapil U, Yajnik CS, Kapoor SK, Dwivedi SN, Singh R (2007). Iron, folate, and vitamin B12 stores among pregnant women in a rural area of Haryana State, India. Food Nutr Bull 28, 435–438.
Schümanna K, Ettle T, Szegner B, Elsenhans B, Solomons NW (2007). On risks and benefits of iron supplementation recommendations for iron intake revisited. J Trace Elements Med Biol 21, 147–168.
Seshadri S (2001). Prevalence of micronutrient deficiency particularly of iron, zinc and folic acid in pregnant women in South East Asia. Br J Nutr 85 (Suppl 2), S87–S92.
Stewart CP, Christian P, Le Clerq SC, West KP, Khatry SK (2009a). Antenatal supplementation with folic acid + iron + zinc improves linear growth and reduces peripheral adiposity in school-age children in rural Nepal. Am J Clin Nutr 90, 132–140.
Stewart CP, Christian P, Schulze KJ, Le Clerq SC, West KP, Khatry SK (2009b). Antenatal micronutrient supplementation reduces metabolic syndrome in 6- to 8-year-old children in rural Nepal. J Nutr 139, 1575–1581.
UNICEF/UNU/WHO (1999). Composition of a Multi-Micronutrient Supplement to be Used in Pilot Programmes Among Pregnant Women in Developing Countries. UNICEF: New York, NY.
Vaidya A, Saville N, Shrestha BP, Costello AM, Manandhar DS, Osrin D (2008). Effects of antenatal multiple micronutrient supplementation on children's weight and size at 2 years of age in Nepal: follow-up of a double-blind randomised controlled trial. Lancet 371, 492–499.
Villamor E, Saathoff E, Bosch RJ, Hertzmark E, Baylin A, Manji K et al. (2005). Vitamin supplementation of HIV-infected women improves postnatal child growth. Am J Clin Nutr 81, 880–888.
WHO (1995). Physical Status: The Use and Interpretation of Anthropometry. Expert Committee Report. WHO Technical Report Series No. 854. World Health Organization: Geneva (http://whqlibdoc.who.int/trs/WHO_TRS_854.pdf).
WHO (2006). Anthro 2005 for mobile devices version 1.00 SfagadotwscGW (http://www.who.int/childgrowth/software/en/).
Zeng L, Cheng Y, Dang S, Yan H, Dibley M, Chang S et al. (2008). Impact of micronutrient supplementation during pregnancy on birth weight, duration of gestation, and perinatal mortality in rural western China: double blind cluster randomised controlled trial. BMJ 337, a2001–a2011.
Zeng F, Song YY, Li Z (2006). A research on relationship between fetal growth restriction and vitamin nutritional status in late pregnancy. Chin J Matern Child Health Res 17, 4–5.
Acknowledgements
This study was supported by United Nations Children's Fund (Grant Number YH101-H12/03) through a cooperative agreement between UNICEF and the Centers for Disease Control and Prevention, Atlanta, US, and the National Natural Science of Foundation of China (Grant Number 30271131), Beijing, China. We are grateful to all the participants and their families who participated in the trial; the field-team members; Gong Aijing and Pan Yonghe who entered data. We thank the field supervisors Li Yanqing, Kang Yijun, Xing Yuan, Fang Bo, Liang Wei-feng, Wang Bei, Duan Sheng-gang and Shen Yuan who helped in the field procedures. This study has been registered as an International Standard Randomized Controlled Trial (Number ISRCTN08850194).
This study has been registered as an International Standard Randomized Controlled Trial (No. ISRCTN08850194).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Contributors: All authors contributed to the design and implementation of the study. WW coordinated the study, trained field stuff, supervised the quality control, collected data, cleaned, analyzed and interpreted the data, and drafted the manuscript. HY designed the study and supervised the field operation, data analysis, supervised the manuscript preparation and revised the paper. LZ developed the protocol and the field procedures, made substantial contributions to the execution and supervision of the field investigation. YC contributed to the supervision of the field investigation and data management. DW analysed the data independently and reviewed and revised the paper. QL helped the data analysis. All authors contributed to the manuscript and approved the final version.
Rights and permissions
About this article
Cite this article
Wang, W., Yan, H., Zeng, L. et al. No effect of maternal micronutrient supplementation on early childhood growth in rural western China: 30 month follow-up evaluation of a double blind, cluster randomized controlled trial. Eur J Clin Nutr 66, 261–268 (2012). https://doi.org/10.1038/ejcn.2011.190
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2011.190
Keywords
This article is cited by
-
Associations of maternal serum concentration of iron-related indicators with birth outcomes in Chinese: a pilot prospective cohort study
Italian Journal of Pediatrics (2024)
-
Iron supplementation and deworming during pregnancy reduces the risk of anemia and stunting in infants less than 2 years of age: a study from Sub-Saharan Africa
BMC Pregnancy and Childbirth (2023)
-
Micro-nutrient pools and their mobility in relation to land-use system in a cold high altitude Himalayan mountainous region
Agroforestry Systems (2021)
-
Maternal antenatal multiple micronutrient supplementation for long-term health benefits in children: a systematic review and meta-analysis
BMC Medicine (2016)