Abstract
Leukoencephalopathies are diseases with high clinical heterogeneity. In clinical work, it’s difficult for doctors to make a definite etiological diagnosis. Here, we designed a custom probe library which contains the known pathogenic genes reported to be associated with Leukoencephalopathies, and performed targeted gene capture and massively parallel sequencing (MPS) among 49 Chinese patients who has white matter damage as the main imaging changes, and made the validation by Sanger sequencing for the probands’ parents. As result, a total of 40.8% (20/49) of the patients identified pathogenic mutations, including four associated with metachromatic leukodystrophy, three associated with vanishing white matter leukoencephalopathy, three associated with mitochondrial complex I deficiency, one associated with Globoid cell leukodystrophy (or Krabbe diseases), three associated with megalencephalic leukoencephalopathy with subcortical cysts, two associated with Pelizaeus-Merzbacher disease, two associated with X-linked adrenoleukodystrophy, one associated with Zellweger syndrome and one associated with Alexander disease. Targeted capture and MPS enables to identify mutations of all classes causing leukoencephalopathy. Our study combines targeted capture and MPS technology with clinical and genetic diagnosis and highlights its usefulness for rapid and comprehensive genetic testing in the clinical setting. This method will also expand our knowledge of the genetic and clinical spectra of leukoencephalopathy.
Similar content being viewed by others
Introduction
Leukoencephalopathies are disorders that primarily affect the white matter of the central nervous system (CNS). It contains acquired leukoencephalopathy1,2,3 (leukoencephalopathy induced by ischemia, hypoxia, intoxication, infection, traumatic brain injuries, etc.), genetic leukoencephalopathy4,5,6 (such as metachromatic leukodystrophy, globoid cell leukodystrophy, X-linked adrenoleukodystrophy, etc.) In addition, it also contains some mitochondrial diseases, cerebral cortical degenerative disorders, and so on. Clinically, after considering clinical history, symptoms and brain MRI features, doctors may be able to give a diagnosis for acquired leukoencephalopathies. However, leukoencephalopathy is a disease with high clinical heterogeneity and may involve in multiple genes, it is difficult even for experienced neurologists to make definite diagnosis7,8,9. Therefore, we are in urgent need of finding an efficient, economical, and practical method for diagnosing leukoencephalopathies.
In recent years, gene sequencing technology got amazing advancement. Whole exome sequencing (WES) represents a significant breakthrough in clinical genetic as a powerful tool for etiological discovery in many kinds of disorders10. Benefited from the WES technology, a lot more pathogenic genes have been found and many types of diseases have been identified11,12,13. Innovative application of new technologies is one of the major factors driving advances in medical science, most clinical applications of next-generation sequencing (NGS) concentrate on known and potential candidate genes to generate clear reports and finally promote clinical diagnosis14,15,16,17,18. Targeted gene capture and massively parallel sequencing (MPS) have been shown to be an effective technique for genetic analysis and have already led to many exciting discoveries19,20. To make a clear or definite diagnosis for those patients with leukoencephalopathies, we designed a custom probe library containing 118 genes reported to be associated with leukoencephalopathies (Table 1).
We embarked on this study to assess the utility and effectiveness of targeted capture and MPS technology in 49 Chinese leukoencephalopathy patients. To our knowledge, this is the first study to use targeted gene capture and sequencing for leukoencephalopathies. 40.8% positive rate confirmed that the implementation of this method can accelerate diagnosis, reduce overall cost, and expand our knowledge of the genetic and clinical spectra of leukoencephalopathies.
Results
Demographic and Clinical characteristics of the total 49 patients
We summarized the clinical characteristics of the total 49 patients enrolled in this study and found 39 are male and 10 are female. The age at onset of symptoms varied from 20 days to 7 years and the average onset age was almost 1.2 years. The main neurologic complaint of these patients include developmental delay/regression (27/49, 55.1%), epilepsy (15/49, 30.6%), weakness (7/49, 14.3%), ataxia (5/49, 10.3%) and dystonia (5/49, 10.3%). The severity of the disease course is reflected in the developmental milestones achieved. Two patients have suspected familial clustering. One has been diagnosed as adrenoleukodystrophy by gene testing. His mother’s elder brother had the same clinic feature and MRI findings, and died at his age of 12. The other one has been diagnosed as mitochondrial complex I deficiency, and his elder sister has a similar brain MRI changes without significant neurological disease manifestation. The wide spectrum of MRI findings was noted in the study. Abnormality in periventricular, subcortical white matter and cerebellar hemisphere were common. Lumbar puncture and CSF analysis were performed in 19 patients. None of them had a positive result.
Twenty patients were identified pathogenic mutations in this study, and their demographic and clinical characteristics were shown in Table 2. However, more than half (29/49, 59%) of patients in our study did not reach the diagnosis.
Targeted capture and MPS sequencing results
In this study, 40.8% (20/49) exhibited pathogenic mutations, in which fifteen pathogenic variation sites have not yet been reported in HGMD. The proportion of each kind of disease diagnosed in our study is shown in Fig. 1. The most common disease diagnoses were metachromatic leukodystrophy (4/49, 8.2%), mitochondrial diseases (3/49, 6.1%), vanishing white matter disorder (3/49, 6.1%) and megalencephalic leukoencephalopathy with subcortical cysts (3/49, 6.1%). Details genetic data were summarized in Table 3.
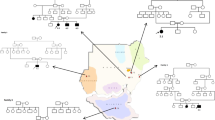
The etiology composition of Leukoencephalopathies in this cohort.
(A) Flow diagram to exhibit workflow and results in this cohort. (B) Pie chart to exhibit the etiology composition of leukoencephalopathies in this cohort. MLD: metachromatic leukodystrophy, VWM: vanishing white matter disorder, AD: Alexander disease, PMD: Pelizaeus-Merzbacher Disease, X-ALD: X-linked adrenoleukodystrophy, MLC: megalencephalic leukoencephalopathy with subcortical cysts, GLD: globoid cell leukodystrophy.
Discussion
With the widespread use of imaging examinations in nervous system diseases, finding the pathogeny of cerebral white matter lesions becomes an important clinical clue for neurologists. Because of the strong heterogeneity of hereditary leukoencephalopathy, it is difficult even for experienced doctors to make a definitive diagnosis, and a multistep process is often needed7,21. Currently, routine clinical diagnostic tests for leukodystrophy often consist of screening for genes on the basis of ethnic origin, MRI features, family history, personal history and findings from physical examinations22. In China, the problem seems more serious, with the lack of a referral system, many patients and their families wasted valuable time, finances, and medical resources seeing various doctors and getting repeat examinations in search of a correct diagnosis. Some patients who could even be cured missed the opportunity for effective treatment. However, due to the high cost of Sanger sequencing for the long list of candidate genes, more effective genetic screening methods are needed.
In recent years, targeted capture and MPS technologies have been widely used in clinical practice and have got satisfactory results15,23,24,25,26,27,28. To this end, we designed the gene panel contains 118 genes which are reported to be associated with leukoencephalopathies, not only contains genes associated with genetic leukoencephalopathy, but also mitochondrial disease, cerebral cortical degenerative disorders, etc. associated genes. Then we designed the probe library and performed this study to assess the utility and effectiveness of targeted capture and MPS in diagnosing leukoencephalopathy patients.
In our study, 40.8% (20/49) of the patients detected pathogenic mutations, which is higher than that of other commercially available chips. In our department, the positive rate of a mitochondrial disease chip is only 9.5%, and that of a metabolic disease chip is 16% (data not shown). These differences may be explained by the variety of pathogenic mutations and lack of a specific clinical phenotype associated with these disorders. Moreover, the results achieved using the leukoencephalopathy probe library may be explained by the distinctive brain MRI patterns that characterize leukoencephalopathy that was seen in most of the patients, providing a guide in the diagnostic process. In addition, patients had been thoroughly examined before the screening for leukoencephalopathy-associated genes, and other secondary causes were already excluded.
Among the result, one patient (case 19) was diagnosed with Zellweger syndrome with PEX6 gene compound heterozygous mutations, PEX6 gene mutation is reported to be associated with Peroxisome biogenesis disorder 4A/B29,30. The patient in our study was a 5.9-year-old girl exhibiting mental and motor retardation for 5 years, and with deterioration for 3 months (Clinical features and auxiliary examinations are included in Table 2). Brain MRI showed symmetrically increased signal intensity in T2-weighted images with gadolinium enhancement in the posterior limbs of the internal capsules (Fig. 2). There was no diffuse restriction or gadolinium enhancement in the periventricular area and deep white matter, similar to the features of X- ALD31,32. However, this is a female and the ABCD1 gene in this patient exhibited a normal sequence and gene dosage. Given the diagnostic uncertainty, targeted capture and MPS were performed. Molecular testing identified PEX6 gene compound heterozygous mutations, supporting the Zellweger spectrum disorder diagnosis in this patient. Genetic analysis showed that the two mutation sites were respectively inherited from the parents. The result showed us the effectiveness of this targeted capture and MPS method for the diagnosis of leukoencephalopathies.
Brain MRI changes of case19 and the electropherogram of Sanger sequencing of the compound mutation of PEX6 gene.
On FLAIR and T2-weighted sequences, abnormal hyperintense is seen in the splenium of corpus callosum, adjacent parieto-occipital white matter, posterior limbs of internal capsules extending to centrum oval, thalami and upper cervical spinal cord. Gadolinium enhancement is visible on T1-weighted sequences in internal capsules and anterior commissure. The child detected missense mutation on chr6:42932599(c.2735C > T) and nonsense mutation on chr6:42937459(c.1313insT), which were respectively inherited from the parents. (a) Brain MRI T1-weighted image. (b) Brain MRI T2-weighted image. (c) Brain MRI flare image. (d) Brain MRI enhanced image. (e–g) The electropherogram of Sanger sequencing of the probands (e), the father (f) and the mother (g) on chr6:42932599. (h–j) The electropherogram of Sanger sequencing of the probands (h), the father (i) and the mother (j) on chr6:42937459.
The targeted capture and MPS method can not only diagnose genetic leukoencephalopathies, but also can make the diagnosis of mitochondrial diseases with white matter abnormal as the primary imaging changes. In our study, three of our patients had pathogenic gene mutations associated with mitochondrial complex I deficiency. Our team was the first to report leukoencephalopathy associated with mitochondrial complex I deficiency due to a novel mutation in the NDUFAF1 gene (c.278A > G; c.247G > A)33. Mitochondrial complex I deficiency is the most frequent cause of respiratory chain defects in childhood, which accounts for various clinical presentations34,35. As the report, mutations have been described in 28 of these, including the 7 mitochondrial genes and 21 nuclear genes. Brain lesions caused by mitochondrial complex 1 deficiency are usually located in the brainstem, periaqueductal gray matter, the thalamus, etc. While, diffuse supratentorial leukoencephalopathy involving the deep lobar white matter may also occur in patients with mitochondrial complex 1 deficiency, especially in patients with nuclear DNA (nDNA) mutations. Some patients were available with abnormal white matter containing cysts in FLAIR sequences, and other patients may have notably hyperintense on T2 and very hypointense on T1 weighted images, suggesting cysts33,36. Therefore, containing pathogenic genes associated with mitochondrial diseases can promote the diagnosis of patients with leukoencephalopathies.
The patients in our study came from six provinces in central-south China. Therefore, the results may represent the specific disease incidence in this region. When clinicians encounter children with prominent cerebral white matter lesions that can’t be explained by a certain disease, application of leukoencephalopathy probe library gene screening may be useful. Targeted capture and MPS can detect multiple candidate genes at the same time in a fast, cost-effective way, and can facilitate clinical diagnosis. Moreover, by reaching a definitive diagnosis for children with leukoencephalopathy, we can better judge the prognosis for patients and provide genetic counseling.
In summary, our data demonstrate that the use of targeted capture and MPS technology coupled with NGS has great promise as a tool for screening leukoencephalopathy-related genes for diagnostic purposes in patients. At the same time, genetic testing results combined with detailed clinical phenotypes help us expand our knowledge of the clinical spectra of each type of leukoencephalopathy. This method enables clinicians to identify leukoencephalopathy even the clinical performance is not typical. Moreover, the entire process of targeted capture, sequencing, analysis, and parental analysis was rapid (requiring only 10 days for up to 12 patients).
While targeted genomic capture and MPS technology also has its limitation, it can only identify the known pathogenic mutations. With the development of gene testing technology, a lot more pathogenic mutations will be detected, so the panel should be renewed with the latest findings, and the patients with negative results of genetic testing can be re-tested using the newest panel. With the fast development of NGS sequencing, the price will be more accessible, we can choose whole exome sequencing (WES) if the targeted analysis is unrevealing, or we can directly choose the WES technology. WES will be the inevitable trend, but under the condition of most countries and before this come true, our panel with cheap price, fast testing speed and strong pertinence, still have the irreplaceable advantage. Thus, we expect that this method can serve as an inspirational starting point. This technology will enable us to conduct straightforward, comprehensive screening for more known leukoencephalopathy-related genes, and to expand and redefine the genetic and clinical spectra of leukoencephalopathies.
Methods
Patients
From December 2013 to December 2015, 49 patients (10 female and 39 male) were recruited into our cohort. All of these patients have white matter damage as the most obvious imaging characteristic. Two pediatric neurologists and one radiologist made the decisions together, according to the medical history, family history, physical examination and magnetic resonance imaging (MRI), patients with obvious ischemia, hypoxia, intoxication or infection was not enrolled in our cohort. The study design was approved by institutional review board of Xiangya Hospital of Central South University, China. And the study procedures were carried out in accordance with the requirements of regulations and procedures regarding human subject protection laws. After obtaining informed consent from all participants, we recorded the clinical features of the patients and collected blood samples from the patients and their parents via venipuncture.
Panel design
We searched the OMIM and HGMD professional databases for genes which are reported to be associated with leukoencephalopathies. A custom-based targeted Agilent SureSelect pull-down panel was designed with the SureDesign program (Agilent Technologies). This target was 0.7 Mb of sequence from the coding exons (GRCh37/hg19 human reference sequence, UCSC Genome Browser) of 118 related candidate or known genes.
Genetic testing
Genomic DNA was isolated from peripheral blood leukocytes (Promega, Beijing). Target-fragments are capture by SureDesign target enrichment kit (Agilent, Santa Clara, CA) and high throughput sequencing by HiSeq2500 sequencer (Illumina Inc, San Diego, CA) were conducted in house. Overall, 49 samples were sequenced pre lane and the mean depth is 583X.
Bioinformatic Pipeline
For the quality control, the Cutadapt and FastQC were used to remove 3′-/5′- adapters andlow-quality reads, respectively. The clean reads were mapped to the reference human genome using the BWA (Burrows–Wheeler Aligner) program with at most two mismatches. The alignment files (bam) were generated with SAM tools and the reads of low mapping quality (<Q30) were filtered out. Clonal duplicated reads that may be derived from PCR artifacts were removed using Picard Tools by default parameters. Short read alignment and annotation visualization were performed using the IGV (Integrative Genomics Viewer). The percentage of alignment of the clean read to the exome regions was obtained using our custom Perl scripts on the base of alignment files. SNVs and indels were detected by GATK (Genome Analysis ToolKit). Comprehensive annotation of all of the detected SNVs and indels were annotated by ANNOVAR, including function implication (gene region, functional effect, mRNA GenBank accession number, amino acid change, cytoband, etc.) and allele frequency in 1000 Genomes, ExAc. Damaging missense mutations were predicted by SIFT, PolyPhen-2 and MutationTaster.
Additional Information
How to cite this article: Wang, X. et al. The use of targeted genomic capture and massively parallel sequencing in diagnose of Chinese Leukoencephalopathies. Sci. Rep. 6, 35936; doi: 10.1038/srep35936 (2016).
References
Maggi, P. et al. The formation of inflammatory demyelinated lesions in cerebral white matter. Ann Neurol. 76(4), 594–608, doi: 10.1002/ana.24242 (2014).
Jelcic, I., Jelcic, I., Faigle, W., Sospedra, M. & Martin, R. Immunology of progressive multifocal leukoencephalopathy. J Neurovirol 21(6), 614–22, doi: 10.1007/s13365-014-0294-y (2015).
Kremer, S. et al. Use of Advanced Magnetic Resonance Imaging Techniques in Neuromyelitis Optica Spectrum Disorder. Jama Neurol. 72(7), 815–22, doi: 10.1001/jamaneurol (2015).
Gordon, H., Letsou, A. & Bonkowsky, J. The Leukodystrophies. Semin Neurol. 34(3), 312–20, doi: 10.1055/s-0034-1386769 (2014).
Aicardi, J. The inherited leukodystrophies: a clinical overview. J Inherit Metab Dis. 16(4), 733–43 (1993).
Kaye, E. M. Update on genetic disorders affecting white matter. Pediatr Neurol. 24(1), 11–24 (2001).
Bonkowsky, J. L. et al. The burden of inherited leukodystrophies in children. Neurology 75(8), 718–25, doi: 10.1212/WNL.0b013e3181eee46b (2010).
Holmes, L., Cornes, M. J., Foldi, B., Miller, F. & Dabney, K., Clinical epidemiologic characterization of orthopaedic and neurological manifestations in children with leukodystrophies. J Pediatr Orthop. 31(5), 587–93, doi: 10.1097/BPO.0b013e3182204930 (2011).
Schiffmann, R. & van der Knaap, M. S. Invited Article: An MRI-based approach to the diagnosis of white matter disorders. Neurology 72(8), 750–9, doi: 10.1212/01.wnl.0000343049.00540.c8 (2009).
Ghaoui, R. et al. Use of Whole-Exome Sequencing for Diagnosis of Limb-Girdle Muscular Dystrophy: Outcomes and Lessons Learned. Jama Neurol. 72(12), 1424–32, doi: 10.1001/jamaneurol.2015.2274 (2015).
Holst-Jensen, A. et al. Application of whole genome shotgun sequencing for detection and characterization of genetically modified organisms and derived products. Anal Bioanal Chem. 408(17), 4595–614, doi: 10.1007/s00216-016-9549-1 (2016).
Witney, A. A. et al. Clinical use of whole genome sequencing for Mycobacterium tuberculosis. Bmc Med. 14, 46, doi: 10.1186/s12916-016-0598-2 (2016).
Marvig, R. L., Sommer, L. M., Jelsbak, L., Molin, S. & Johansen, H. K. Evolutionary insight from whole-genome sequencing ofPseudomonas aeruginosafrom cystic fibrosis patients. Future Microbiol. 10(4), 599–611, doi: 10.2217/fmb.15.3 (2015).
Li, Z., Lin, Q., Huang, W. & Tzeng, C. Target Gene Capture Sequencing in Chinese Population of Sporadic Parkinson Disease. Medicine 94(20), e836, doi: 10.1097/MD.0000000000000836 (2015).
Kodera, H. et al. Targeted capture and sequencing for detection of mutations causing early onset epileptic encephalopathy. Epilepsia 54(7), 1262–9, doi: 10.1111/epi.12203 (2013).
Tekin, D. et al. A next-generation sequencing gene panel (MiamiOtoGenes) for comprehensive analysis of deafness genes. Hearing Res. 333, 179–84, doi: 10.1016/j.heares.2016.01.018 (2016).
Hildebrand, M. S. et al. A targeted resequencing gene panel for focal epilepsy. Neurology 86(17), 1605–12, doi: 10.1212/WNL.0000000000002608 (2016).
Chen, S. et al. Identification of PKD2 mutations in human preimplantation embryos in vitro using a combination of targeted next-generation sequencing and targeted haplotyping. Sci Rep-UK 6, 25488, doi: 10.1038/srep25488 (2016).
Carvill, G. L. et al. Targeted resequencing in epileptic encephalopathies identifies de novo mutations in CHD2 and SYNGAP1. Nat Genet. 45(7), 825–30, doi: 10.1038/ng.2646 (2013).
Rehm, H. L. Disease-targeted sequencing: a cornerstone in the clinic. Nat Rev Genet 14(4), 295–300, doi: 10.1038/nrg3463 (2013).
Holmes, L., Cornes, M. J., Foldi, B., Miller, F. & Dabney, K. Clinical epidemiologic characterization of orthopaedic and neurological manifestations in children with leukodystrophies. J Pediatr Orthop. 31(5), 587–9331, doi: 10.1097/BPO.0b013e3182204930 (2011).
Parikh, S. et al. A clinical approach to the diagnosis of patients with leukodystrophies and genetic leukoencephelopathies. Mol Genet Metab. 114(4), 501–15, doi: 10.1016/j.ymgme.2014.12.434 (2015).
Brownstein, Z. et al. Targeted genomic capture and massively parallel sequencing to identify genes for hereditary hearing loss in middle eastern families. Genome Biol. 12(9), R89, doi: 10.1186/gb-2011-12-9-r89 (2011).
Weisschuh, N. et al. Mutation Detection in Patients with Retinal Dystrophies Using Targeted Next Generation Sequencing. Plos One 11(1), e0145951, doi: 10.1371/journal.pone.0145951 (2016).
Sakr, R. A. et al. Targeted capture massively parallel sequencing analysis of LCIS and invasive lobular cancer: Repertoire of somatic genetic alterations and clonal relationships. Mol Oncol. 10(2), 360–70, doi: 10.1016/j.molonc.2015.11.001 (2016).
Chen, X. et al. Targeted next-generation sequencing reveals multiple deleterious variants in OPLL-associated genes. Sci Rep-UK 6, 26962, doi: 10.1038/srep26962 (2016).
Liaw, H., Lee, H., Chi, C. & Tsai, C. Late infantile metachromatic leukodystrophy: Clinical manifestations of five Taiwanese patients and Genetic features in Asia. Orphanet J Rare Dis. 10, 144, doi: 10.1186/s13023-015-0363-1 (2015).
Abdel-Salam, G. M. H. et al. Megalencephalic leukoencephalopathy with cysts in twelve Egyptian patients: novel mutations in MLC1 and HEPACAM and a founder effect. Metab Brain Dis., doi: 10.1007/s11011-016-9861-7 (2016).
Stasyk, O. et al. Identification of intragenic mutations in the gene that affect peroxisome biogenesis and methylotrophic growth. Fems Yeast Res. 4(2), 141–7 (2003).
Ebberink, M. S., Kofster, J., Wanders, R. J. A. & Waterham, H. R. Spectrum ofPEX6 mutations in Zellweger syndrome spectrum patients. Hum Mutat. 31(1), E1058–70, doi: 10.1002/humu.21153 (2010).
Lee, P. R. & Raymond, G. V. Child Neurology: Zellweger syndrome. Neurology 80(20), e207–10, doi: 10.1212/WNL.0b013e3182929f8e (2013).
Tran, C., Hewson, S., Steinberg, S. J. & Mercimek-Mahmutoglu, S. Late-onset Zellweger spectrum disorder caused by PEX6 mutations mimicking X-linked adrenoleukodystrophy. Pediatr Neurol. 51(2), 262–5, doi: 10.1016/j.pediatrneurol.2014.03.020 (2014).
Wu, L. et al. Leukodystrophy associated with mitochondrial complex I deficiency due to a novel mutation in the NDUFAF1 gene. Mitochondrial DNA A DNA MappSeq Anal. 27(2), 1034–7, doi: 10.3109/19401736.2014.926543 (2016).
Grad, L. I. Mitochondrial complex I mutations in Caenorhabditis elegans produce cytochrome c oxidase deficiency, oxidative stress and vitamin-responsive lactic acidosis. Hum Mol Genet 13(3), 303–14, doi: 10.1093/hmg/ddh027 (2003).
Fassone, E. & Rahman, S. Complex I deficiency: clinical features, biochemistry and molecular genetics. J Med Genet 49(10), 668, doi: 10.1136/jmedgenet-2012-101159 (2012).
Lebre, A. S. et al. A common pattern of brain MRI imaging in mitochondrial diseases with complex I deficiency. J Med Genet 48(1), 16–23, doi: 10.1136/jmg.2010.079624 (2010).
Acknowledgements
This work was supported by the National Natural Science Foundation of China [grant number 81371434, 81370771, 31571312]; the Hunan Province Key Technology Support Program [grant number 2015SK2019, 2015JJ3151, 2011FJ3163], and Graduate Student Innovation Project of Central South University (2015zzts294). We thank the participants and clinicians who took part in the study.
Author information
Authors and Affiliations
Contributions
X.W. and F.H. discussed the results, wrote the most manuscript text; F.Y., L.W. and L.Y. clinically evaluated the patients, discussed the results; C.C. performed genetic analyses, discussed the results; J.P. conceived the study, clinically evaluated the patients, performed genetic analyses, discussed the results. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Wang, X., He, F., Yin, F. et al. The use of targeted genomic capture and massively parallel sequencing in diagnosis of Chinese Leukoencephalopathies. Sci Rep 6, 35936 (2016). https://doi.org/10.1038/srep35936
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep35936
This article is cited by
-
GALC mutations in Chinese patients with late-onset Krabbe disease: a case report
BMC Neurology (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.