Host: Nick Petrić Howe
Welcome back to the Nature Podcast. This time: how researchers are making cells from synthetic bubbles.
Host: Shamini Bundell
And the latest from the Nature Briefing. I’m Shamini Bundell.
Host: Nick Petrić Howe
And I’m Nick Petrić Howe.
[Jingle]
Interviewer: Nick Petrić Howe
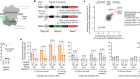
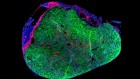
Can you make a cell – the building block of life – from scratch? This is a question that researchers have been pondering for years now, not least because artificial cells could offer some interesting opportunities. Firstly, to understand how life works full stop, but also because handcrafted cells could be used to perform all kinds of useful tasks, from detecting toxins to producing drug molecules. ‘But engineered cells already do that,’ I hear you cry, and to an extent you would be right. For example, engineered E. coli cells are already used to produce human insulin. But in theory, artificial cells could be bespoke for highly specific functions and then only carry that function out, without being bogged down in all the other clutter which comes with keeping living cells like E. coli alive and kicking. And researchers have made strides. Lots of teams have looked at elements of what cells need, from transport proteins to cell membranes to the nucleus, and tried to craft them. Other teams have crafted basic cell-type things from scratch, or they have taken existing cells and streamlined them – whittling away everything that isn’t completely necessary. And now, a team have a different approach to this, and they’ve handcrafted a single cell with seven different components of naturally occurring cells. It includes things that resemble nuclei and cytoskeletons – a potential leap forward for the field. And they’ve done it using bubbles known as coacervates that are made from polymers, but more on that in a minute. I reached out to the authors of the paper who, unfortunately, weren’t available to speak to me, but I did get Amy Yewdall, biochemist and synthetic cellsmith, on the line. Amy’s been writing an expert analysis of the paper this week in Nature, and was able to give me the inside scoop. I started by asking her, why is it so difficult to make life from scratch?
Interviewee: Amy Yewdall
So, in this scale, it is very difficult to control where everything is spatially and temporally. You want to be able to combine things in a way that makes them compatible in function. Having multiple compartments, having enzymatic reactions happen inside these compartments, spatial organisation within these compartments, these are all individual challenges that other groups have addressed but never combined into one system before.
Interviewer: Nick Petrić Howe
Yeah, well, maybe let's talk a little bit about the new paper then. So, what exactly have they done? Maybe let's start from the start. What are they beginning with and what are they adding to it to make an artificial cell?
Interviewee: Amy Yewdall
Yes, so the platform for this artificial cell is a coacervate. So, these are polymers and ATP that come together to form these liquid-liquid phase-separated droplets. And then they use this system to create membranes and internal structures. And then from this, they can add different proteins to make the hierarchical phase-separated structures, such as the DNA core, and they can generate a lot of complexity from this very basic, modular platform.
Interviewer: Nick Petrić Howe
So, they have basically almost like a little droplet made from this polymer with ATP inside. And then on top of that, they can layer all the sorts of things you might need for a cell.
Interviewee: Amy Yewdall
Yeah.
Interviewer: Nick Petrić Howe
And then they've taken parts from two different kinds of bacteria, and then put that into this artificial droplet. What's going on here?
Interviewee: Amy Yewdall
Here, they’ve chosen two different types of bacteria. One type of bacteria localise inside the droplets, and the other type templates around the outside. So, you have this kind of spatial control of where these different bacteria localise, and from this you can generate different types of membrane structures. And when you pop the bacteria, they also release their contents, like proteins et cetera, into the coacervate, and they showed that these are still functional, which is pretty cool.
Interviewer: Nick Petrić Howe
Right, so, just to make sure I've got this straight, they have these droplets, these coacervates, and the bacteria get taken up and they sit on the inside or the outside. And then by popping the bacteria, components of the bacteria end up becoming part of the synthetic cell. So, in the paper, they talk about proteins that are involved in structural and catalytic roles. For example, they showed us the amount of transcription and translation going on here inside the synthetic cells. And one thing that I thought was cool is they also stick another E. coli in there, and it actually works as a kind of mitochondria. But I guess when it comes down to it, they've got all this stuff inside and outside of the cells, but what do these synthetic cells actually do? Do they actually do anything or is this just a proof of concept?
Interviewee: Amy Yewdall
So, I think at this point, it feels more like a proof of concept – the fact that you can combine seven different features of life together into one system. They do show that there are active enzymes inside the cytosol, if you will, of these artificial cells. They show that you can have protein-making machinery inside the cell and this still functions to some extent. So, that is a pretty cool feature of artificial cells. This is something that a lot of groups have tried to get to work in their own systems, but here they show that it works in the coacervates, which is pretty neat.
Interviewer: Nick Petrić Howe
And to ask this question in a slightly different way, what's the sort of difference between what they've made and sort of a normal, naturally occurring cell?
Interviewee: Amy Yewdall
So, why we would say it's more proof of concept is because they make, for example, GFP, so very stock standard proteins, just to show that, hey, we've got some fluorescence appearing inside these cells. But in a normal cell, you can imagine that they need to produce all of these components that are found within a living system, from structural proteins to enzymes, thousands of different proteins that we are not able to achieve this at the moment in our fields.
Interviewer: Nick Petrić Howe
So, what does this sort of mean for the field? There is now this proof of concept. What can people then use this for?
Interviewee: Amy Yewdall
In general, you can imagine that if you can get these artificial cells to make any kind of protein, that you can eventually make all sorts of proteins that are involved in making cool, small molecules for biotechnology, for example, useful, commercially useful molecules, for example. You could make whatever you want, in terms of the DNA you put in, in theory.
Interviewer: Nick Petrić Howe
So, it’s really like a blank slate that we could potentially use to make sorts of useful compounds.
Interviewee: Amy Yewdall
That is the dream. That is what we'd like to do.
Interviewer: Nick Petrić Howe
And so, as someone who works in this field, what are your thoughts on this technique, maybe critically and otherwise?
Interviewee: Amy Yewdall
I think using coacervates as a template for artificial cells is pretty interesting because these are charged droplets, so you get a concentrating effect from all of these proteins that you might add to the system. So, later on in the paper, they show they can sprinkle in a bit of G-actin, and it gets localised inside these droplets, for example. So, you can add things outside the solution and they can permeate into the interphase, if you will, which is something that is more difficult to do in other systems that have the traditional artificial cells, for example, these lipid-based droplets, so lipid-based compartments where the permeability is more of an issue. But in terms of my critique for this, is that using a synthetic polymer, you have to wonder how many biological systems are actually compatible with this, right? Like if you have more sensitive proteins, for example, we don't know how they would behave in these very novel environments. But the only thing we can do is test it and try it, right? So, more work to be done, I would say.
Interviewer: Nick Petrić Howe
That was Amy Yewdall from Radboud University in the Netherlands. To learn more about these synthetic cells, check out the show notes for a link to the paper, and a News and Views article written by Amy.
Host: Shamini Bundell
Coming up, we'll be hearing about a fateful mutation that may have given our ancestors an edge over Neanderthals. Right now, though, it's time for the Research Highlights with Dan Fox.
[Jingle]
Dan Fox
One hundred and fifty million years ago, towards the end of the Jurassic period, an unknown creature dwelt in what is now Utah. But scientists may now be able to figure out its identity based on its vomit. The tiny lump of vomit was deposited into a pond and subsequently fossilised. Despite only measuring around 1 square centimetre in size, the fossil is densely packed with more than 20 undigested bones, as well as other items that may be soft tissues or part of the vomit material. Some of the bones appear to have previously belonged to a tadpole, while others were once part of prehistoric frogs, and a miniscule femur might have come from a tiny species of salamander. Given these contents and the setting in which they were found, the researchers strongly suspect that the identity of the unknown creature was an ancient fish. Read that research in full in Palaios.
[Jingle]
Dan Fox
To protect against obesity and other metabolic conditions, new research has shown that an intricate relationship between diet, gut bacteria and immune cells is key. Metabolic syndrome is a group of health conditions that collectively increase the risk of cardiovascular disease and diabetes. Now, researchers have found that in mice, certain gut bacteria helped to prevent the disorders that make up metabolic syndrome by inducing a rise in a type of immune cell. These T helper 17 cells reduce the gut’s absorption of lipids. The team found that a high-fat, high-sugar diet fuelled the growth of another bacteria which killed off the useful immune-regulating gut bacteria. But giving the mice bacterial supplements could still prevent them from developing obesity and metabolic syndrome. Read that research in full in Cell.
[Jingle]
Host: Shamini Bundell
Finally on the show, it's time for the Briefing chat, where we discuss a couple of articles that have been highlighted in the Nature Briefing. So, Nick, what's your story for this week?
Host: Nick Petrić Howe
So, I've been reading an article in Nature about research that's been trying to establish the safety of using embryos with DNA from three people.
Host: Shamini Bundell
And is there a big need to create embryos with DNA from three people?
Host: Nick Petrić Howe
Yeah, so the reason you'd want to do this is because there are these mitochondrial diseases which occur when people have mutations in the DNA of their mitochondria, and this can cause them all sorts of problems. So, it can lead to autism, it can lead to sort of muscle weakness, heart disease, liver problems. The mitochondria is very important in the cell. It's the powerhouse of the cell. So, the reason that you'd want to do this, and why it's DNA from three people, is you're trying to get healthy mitochondria that have all the DNA without bad mutations into a developing embryo.
Host: Shamini Bundell
Okay, so if someone who knows they have this mutation wants to have a baby, instead of trying to fix the mutation in the embryo, you just give the embryo a whole new set of mitochondria from someone else.
Host: Nick Petrić Howe
Yeah, basically, that's the way it works. So, there are three different main ways to do this. But the one that they were interested in in this study is where you take the nuclear DNA, so the DNA from the nucleus, from the mother and put it into an egg with healthy mitochondria, and then you fertilise that with sperm. So, that's how it ends up with DNA from three different people. And because of that, you have a healthy mitochondria without those damaging mutations in it.
Host: Shamini Bundell
I think I know the answer to why this is, but could you not use the father's mitochondria and then it wouldn't be three people?
Host: Nick Petrić Howe
So, the reason you can't do that is because sperm don't have mitochondria. So, the mitochondria only ever comes from the mother, so that's why you would do it in this way.
Host: Shamini Bundell
And so, is this new research about how to achieve that?
Host: Nick Petrić Howe
So, the new research is trying to establish whether it's safe. So, this has actually been done a few times. Someone was actually born in 2016 with DNA from three different people. They had their mitochondria replaced, as it were. And so, there have been studies in the past, but these have been in animals, and this is the first like comprehensive study in humans. And they showed that it doesn't seem to affect the development of the embryos at all. So, they looked at embryos for a week and showed that after five days, there was no difference between embryos where it has been done and where it hasn't been done.
Host: Shamini Bundell
So, if this has already been actually put into practice, why do they still need extra research sort of on the safety if it's already been done?
Host: Nick Petrić Howe
Well, in some places, you can do this technique and in other places, you can't. So, for example, here in the UK, you can do this, but in the US, it is banned. So, studies like this would help this get through the sort of regulatory checks it needs in the various places around the world to be used. And that would be really great because these sorts of mitochondrial diseases, they affect quite a lot of people. So, every 1 in 5,000 births has one of these mitochondrial errors in it. And as I said before, this can lead to all sorts of problems as someone grows up.
Host: Shamini Bundell
So, that’s obviously really good news, especially as there are people who've been born using this technique. Are there any remaining worries about it?
Host: Nick Petrić Howe
So, the one thing we don't know are the long-term impacts. Like I said, someone was born where this had been done in 2016. But they're only six years old, so we don't really know what the long-term impacts are, and they're just one person as well. There isn't really reason to think there might be. The only thing is that when this technique is done, you do end up taking a little bit of the mitochondrial DNA from the original mother who has the harmful mutations. So, that could mean that later on, you end up with some other things. But that's something we'll just have to get more data on. But the researchers interviewed for this article were very pleased with this sort of dataset. One person was quoted as saying, ‘It is unique and fabulous for its sort of high quality and depth of data.’
Host: Shamini Bundell
Oh, brilliant. Well, that's fascinating. Thanks, Nick. I actually also have a sort of genetics vaguely related story this week. So, I was reading this article in Nature about a Science paper on a gene that's present in humans, but not generally in Neanderthals, that might have given sort of Homo sapiens an advantage, cognitively speaking, over Neanderthals.
Host: Nick Petrić Howe
That’s super interesting. So, if this was a gene that's present in humans and not in Neanderthals, how were they able to determine that it wasn’t present in Neanderthals? Ancient DNA is quite hard to get a hold of, I understand.
Host: Shamini Bundell
Well, so, the first fully sequenced Neanderthal genome was back in 2014. And they did compare that with certain human genomes and in that sort of original study, they found 96 amino acids that were different. Right, so, amino acids are the building blocks that then make up the proteins in our bodies. So, kind of a small number of changes, really, and you would generally expect there to be lots of sort of small cumulative changes that sort of makes species diverge. But this gene, the researchers found particularly interesting. There’s actually a couple of neuroscientists who've been looking into this, and this particular gene that was different – TKTL1 – encodes a protein from fetal brain development. So, when the fetus’s brain is first developing, this gene, which, again, a single amino acid difference, makes a different protein and changes how neurons develop.
Host: Nick Petrić Howe
Oh, right, and how does changing how neurons develop lead to sort of a cognitive advantage?
Host: Shamini Bundell
Well, okay, so they have basically done a lot of experiments to try and figure out, first off, what this gene is doing and second off, whether this could be significant to cognition. So, they theorised that this protein could be basically increasing the number of neurons in the neocortex in particular. Now, it's also important to note here that, as far as we can tell from the fossils, Neanderthal brains and human brains are kind of the same size. So, this isn't a case of humans suddenly develop giant brains, but they could have been denser brains with these more neurons, or these neurons could have made the neocortex in a larger part of the brain. And they've done a bunch of experiments on mice and ferrets trying to show that, yes, this TKTL1 gene, if you insert it, does cause this sort of proliferation of neurons. They also experimented with human fetuses and with brain organoids to, again, show that the other way around, if you insert the sort of ancestral version of the gene, they produce fewer neurons than normal. So, it seems that this small change does actually have quite a big impact.
Host: Nick Petrić Howe
And so, having more neurons and that sort of thing, could that have maybe led to us outcompeting Neanderthals back in the day?
Host: Shamini Bundell
Well, that's kind of what they're proposing, but this is not what this paper shows. So, what they haven't proven is a sort of actual cognitive difference. Did people who had this gene, were they smarter, and that's a little bit harder to prove. The ancestral form of this gene is very rare among modern humans, although I think it does exist. We don't know whether having it causes any sort of cognitive differences. A possible next step would be to create, say, mice with these two versions of the genes and try and see if there's an actual cognitive difference or behavioural difference. We don't quite know yet.
Host: Nick Petrić Howe
That's so interesting that such a small change could have had such a large impact on our history as a species. Thanks for bringing that one to the Briefing, Shamini. And listeners, for more on those stories, and for where to sign up to the Nature Briefing to get more like them direct to your inbox, check out the show notes.
Host: Shamini Bundell
And that is all for this week. As always, you can keep in touch with us on Twitter – we’re @NaturePodcast. Or why not send us an email – we’re podcast@nature.com. I’m Shamini Bundell.
Host: Nick Petrić Howe
And I’m Nick Petrić Howe. Thanks for listening.

 Read the paper: Living material assembly of bacteriogenic protocells
Read the paper: Living material assembly of bacteriogenic protocells
 Embryos with DNA from three people develop normally in first safety study
Embryos with DNA from three people develop normally in first safety study
 Did this gene give modern human brains their edge?
Did this gene give modern human brains their edge?