- NEWS FEATURE
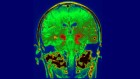
Could drugs prevent Alzheimer’s? These trials aim to find out
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 603, 216-219 (2022)
doi: https://doi.org/10.1038/d41586-022-00651-0
References
Goate, A. et al. Nature 349, 704–706 (1991).
Uhlmann, R. E. et al. Nature Neurosci. 23, 1580–1588 (2020).
Glenner, G. G. & Wong, C. W. Biochem. Biophys. Res. Commun. 122, 1131–1135 (1984).
Ikeda, S., Yanagisawa, N., Allsop, D. & Glenner, G. G. Lab. Invest. 61, 133–137 (1989).
Gordon, B. A. et al. Lancet Neurol. 17, 241–250 (2018).
Salloway, S. et al. Nature Med. 27, 1187–1196 (2021).
Donohue, M. C. et al. JAMA 317, 2305–2316 (2017).
Kapasi, A., DeCarli, C. & Schneider, J. A. Acta Neuropathol. 134, 171–186 (2017).
Cummings, J., Lee, G., Zhong, K., Fonseca, J. & Taghva, K. Alzheimers Dement. 7, e12179 (2021).

 Is ‘friendly fire’ in the brain provoking Alzheimer’s disease?
Is ‘friendly fire’ in the brain provoking Alzheimer’s disease?
 Landmark Alzheimer’s drug approval confounds research community
Landmark Alzheimer’s drug approval confounds research community
 The red-hot debate about transmissible Alzheimer’s
The red-hot debate about transmissible Alzheimer’s