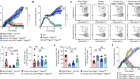
- NEWS
‘Killer’ immune cells still recognize Omicron variant
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 601, 307 (2022)
doi: https://doi.org/10.1038/d41586-022-00063-0
References
May, D. H. et al. Preprint at medRxiv https://doi.org/10.1101/2021.12.20.21267877 (2021).
Keeton, R. et al. Preprint at medRxiv https://doi.org/10.1101/2021.12.26.21268380 (2021).
Geurts van Kessel, C. H. et al. Preprint at medRxiv https://doi.org/10.1101/2021.12.27.21268416 (2021).
Liu, J. et al. Preprint at medRxiv https://doi.org/10.1101/2022.01.02.22268634 (2022).
Collie, S., Champion, J., Moultrie, H., Bekker, L.-G. & Gray, G. N. Engl. J. Med. https://doi.org/10.1056/NEJMc2119270 (2021).
Gray, G. E. et al. Preprint at medRxiv https://doi.org/10.1101/2021.12.28.21268436 (2021).

 How ‘killer’ T cells could boost COVID immunity in face of new variants
How ‘killer’ T cells could boost COVID immunity in face of new variants
 Omicron’s feeble attack on the lungs could make it less dangerous
Omicron’s feeble attack on the lungs could make it less dangerous
 Omicron overpowers key COVID antibody treatments in early tests
Omicron overpowers key COVID antibody treatments in early tests
 How severe are Omicron infections?
How severe are Omicron infections?
 Beyond Omicron: what’s next for COVID’s viral evolution
Beyond Omicron: what’s next for COVID’s viral evolution