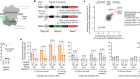
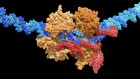
- NEWS
Landmark CRISPR trial shows promise against deadly disease
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
doi: https://doi.org/10.1038/d41586-021-01776-4
References
Gillmore, J. D. et al. N. Engl. J. Med. https://doi.org/10.1056/NEJMoa2107454 (2021).

 CRISPR gene therapy shows promise against blood diseases
CRISPR gene therapy shows promise against blood diseases
 CRISPR treatment inserted directly into the body for first time
CRISPR treatment inserted directly into the body for first time
 Quest to use CRISPR against disease gains ground
Quest to use CRISPR against disease gains ground
 First CRISPR editing trial results assuage safety concerns
First CRISPR editing trial results assuage safety concerns