- NEWS AND VIEWS
Squeezed light improves sensitivity of microscopy technique
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 594, 180-181 (2021)
doi: https://doi.org/10.1038/d41586-021-01514-w
References
Freudiger, C. W. et al. Science 322, 1857–1861 (2008).
Casacio, C. A. et al. Nature 594, 201–206 (2021).
Min, W., Freudiger, C. W., Lu, S. & Xie, X. S. Annu. Rev. Phys. Chem. 62, 507–530 (2011).
Audier, X., Heuke, S., Volz, P., Rimke, I. & Rigneault, H. APL Photon. 5, 011101 (2020).
Hopt, A. & Neher, E. Biophys. J. 80, 2029–2036 (2001).
Fischer, F. F. et al. J. Biomed. Opt. 13, 1–8 (2008).
Giovannetti, V., Lloyd, S. & Maccone, L. Science 306, 1330–1336 (2004).
Moreau, P.-A., Toninelli, E., Gregory, T. & Padgett, M. J. Nature Rev. Phys. 1, 367–380 (2019).
Aasi, J. et al. Nature Photon. 7, 613–619 (2013).
Competing Interests
The author declares no competing interests.

 Read the paper: Quantum-enhanced nonlinear microscopy
Read the paper: Quantum-enhanced nonlinear microscopy
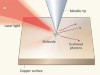 Snapshots of vibrating molecules
Snapshots of vibrating molecules
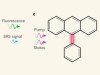 A larger palette for biological imaging
A larger palette for biological imaging