- NEWS AND VIEWS
Ancient seeds spill secrets about the evolution of flowering plants
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 594, 185-186 (2021)
doi: https://doi.org/10.1038/d41586-021-01347-7
References
Shi, G., Herrera, F., Herendeen, P. S., Clark, E. G. & Crane, P. R. Nature 594, 223–226 (2021).
Doyle, J. A. Int. J. Plant Sci. 169, 816–843 (2008).
Soltis, D. E. et al. Phylogeny and Evolution of the Angiosperms Revised & Updated Edn (Univ. Chicago Press, 2018).
Darwin, F. & Seward, A. C. (eds) More Letters of Charles Darwin Vol. II (Murray, 1903).
Endress, P. K. Ann. Bot. 107, 1465–1489 (2011).
Gasser, C. S. & Skinner, D. J. Curr. Top. Dev. Biol. 131, 373–399 (2019).
Barbacka, M. & Bóka, K. Acta Palaeobot. 40, 85–111 (2000).
Bomfleur, B. et al. Int. J. Plant Sci. 175, 1062–1075 (2014).
Taylor, T. N., Del Fueyo, G. M. & Taylor, E. L. Am. J. Bot. 81, 666–677 (1994).
Competing Interests
The author declares no competing interests.

 Read the paper: Mesozoic cupules and the origin of the angiosperm second integument
Read the paper: Mesozoic cupules and the origin of the angiosperm second integument
 Ancient plants escaped the end-Permian mass extinction
Ancient plants escaped the end-Permian mass extinction
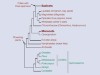 The family tree flowers
The family tree flowers