- NEWS FEATURE
The mysterious microbes that gave rise to complex life
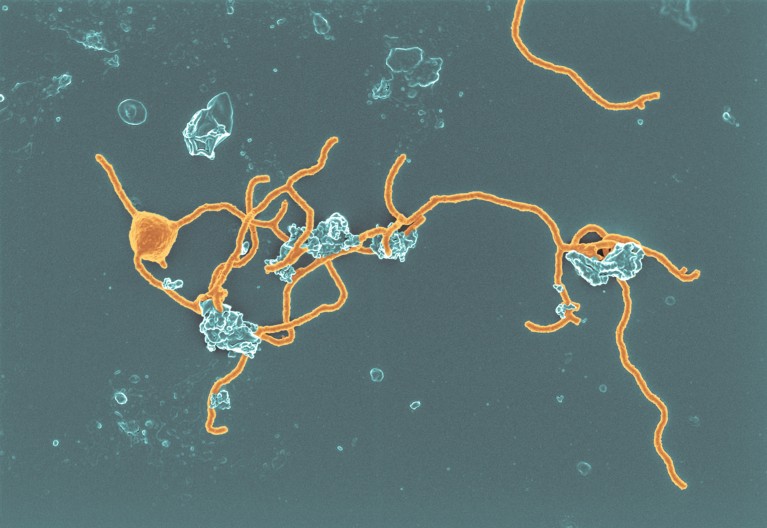
Scientists spent 12 years culturing a slow-growing, tentacled archaeon thought to be similar to the ancestor of complex cells. Credit: Hiroyuki Imachi, Masaru K. Nobu and JAMSTEC
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 593, 328-330 (2021)
doi: https://doi.org/10.1038/d41586-021-01316-0
References
Imachi, H. et al. Nature 577, 519–525 (2020).
Baum, D. A. & Baum, B. BMC Biol. 12, 76 (2014).
Liao, Y. et al. Nature Microbiol. 6, 594–605 (2021).
Nußbaum, P., Gerstner, M., Dingethal, M., Erb, C. & Albers, S. V. Preprint at bioRxiv https://doi.org/10.1101/2020.10.06.327809 (2020).
Pende, N. et al. Preprint at bioRxiv https://doi.org/10.1101/2020.10.06.328377 (2020).
Pulschen, A. A. et al. Curr. Biol. 30, 2852–2859 (2020).
Sagan, L. J. Theor. Biol. 14, 255–274 (1967).
Spang, A. et al. Nature 521, 173–179 (2015).
Gould, S. B., Garg, S. G. & Martin, W. F. Trends Microbiol. 24, 525–534 (2016).

 The trickster microbes that are shaking up the tree of life
The trickster microbes that are shaking up the tree of life
 Meet the relatives of our cellular ancestor
Meet the relatives of our cellular ancestor
 Mining the microbial dark matter
Mining the microbial dark matter
 Archaea and the origins of eukaryotes
Archaea and the origins of eukaryotes