- NEWS AND VIEWS
Highly reactive form of magnesium stabilized by bulky ligands
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 592, 687-688 (2021)
doi: https://doi.org/10.1038/d41586-021-01014-x
References
Rösch, B. et al. Nature 592, 717–721 (2021).
Green, S. P., Jones, C. & Stasch, A. Science 318, 1754–1757 (2007).
Arrowsmith, M. et al. Nature Chem. 8, 890–894 (2016).
Wang, G. et al. J. Am. Chem. Soc. 142, 4560–4564 (2020).
Kriek, S., Görls, H., Yu, L., Reiher, M. & Westerhausen M. J. Am. Chem. Soc. 131, 2977–2985 (2009).
Jones, C. Commun. Chem. 3, 159 (2020).
Rösch, B. et al. Chem. Commun. 56, 11402–11405 (2020).
Weetman, C. & Inoue, S. ChemCatChem 10, 4213–4228 (2018).
Rösch, B. et al. Science 371, 1125–1128 (2021).

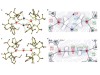 Read the paper: Strongly reducing magnesium(0) complexes
Read the paper: Strongly reducing magnesium(0) complexes
 Fleeting glimpse of an elusive element
Fleeting glimpse of an elusive element
 Peculiar boron startles again
Peculiar boron startles again