- NEWS AND VIEWS
Life in a carbon dioxide world
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 592, 688-689 (2021)
doi: https://doi.org/10.1038/d41586-021-00977-1
References
Steffens, L. et al. Nature 592, 784–788 (2021).
Krebs, H. A. & Martin, A. Hans Krebs, Reminiscences and Reflections (Clarendon, 1981).
Fuchs, G. Annu. Rev. Microbiol. 65, 631–658 (2011).
Sharon, I. & Banfield, J. F. Science 342, 1057–1058 (2013).
Keller, A., Schink, B. & Müller, N. Front. Microbiol. 10, 2785 (2019).
Orsi, W. D. Nature Rev. Microbiol. 16, 671–683 (2018).
Sossi, P. A. et al. Sci. Adv. 6, eabd1387 (2020).
Sleep, N. H., Bird, D. K. & Pope, E. C. Phil. Trans. R. Soc. B 366, 2857–2869 (2011).
Preiner, M. et al. Nature Ecol. Evol. 4, 534–542 (2020).
Baross, J. A. Nature 564, 42–43 (2018).

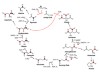 Read the paper: High CO2 levels drive the TCA cycle backwards towards autotrophy
Read the paper: High CO2 levels drive the TCA cycle backwards towards autotrophy
 Energy from thin air
Energy from thin air
 Microbes make metabolic mischief by targeting drugs
Microbes make metabolic mischief by targeting drugs