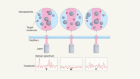
Microwaves brighten the light emitted by nanodiamonds attached to a virus.Credit: Rosanna Wan
All atoms have properties governed by the laws of quantum mechanics, but those properties are fragile. Particles don’t usually hold their quantum state long enough to be measured, unless they are in a vacuum or at ultra-cold temperatures. But a diamond, one of the hardest substances in nature, stands up to observation a little more robustly. The stiffness of its crystals can protect quantum states for milliseconds, even at room temperature — plenty of time to do something useful. Over the past 15 years, engineers have become adept at growing artificial diamonds with desirable quantum properties, opening up applications in biosensing, magnetometry and even quantum computing.
Part of Nature Outline: Quantum diamond sensors
A diamond is a collection of carbon atoms, each bonded to four other carbons to form an orderly crystalline array. But sometimes there’s a glitch in the matrix: a stray atom of another element finds its way in or a carbon atom is missing, leaving an empty space. These defects cause a diamond to sparkle in different hues, and are called colour centres.
One particularly interesting defect occurs when a carbon in the crystal is replaced by a nitrogen atom, and the adjacent carbon is missing. This defect is known as a nitrogen-vacancy (NV) centre and has its own quantum spin, which can be thought of as a rotating magnet. Diamonds are mostly made of spin-neutral carbon-12 atoms, so the NV centre’s spin is unaffected by that of its immediate neighbours. And because the diamond matrix is so stiff, the atoms don’t jostle enough at room temperature to nudge the spin into a different state.
The spin can be altered, however, by electromagnetic radiation or a magnetic field — a property that enables diamonds with NV centres to be used as sensors. The NV centre is also photoluminescent: when lit with green light it will emit a red glow. Because the spin state of the NV centre determines how strongly the diamond fluoresces, scientists can use changes in brightness to monitor changes in the centre’s spin state due to microwaves or a magnetic field. By examining which frequencies cause changes in the light, researchers can even use the diamond to measure the strength of a magnetic field. This technique is called optically detected magnetic resonance.
The ability to read spin with light has many potential applications. In biology, it can detect viruses and tumour cells in blood with a sensitivity that is hundreds to thousands of times greater than existing techniques can provide. Diamonds have also been used to image minute magnetic fields in meteorites that date back to the formation of the Solar System.
NV centres might even have applications in quantum computing. One method of making the bits for quantum computers — qubits — relies on superconductors, which must be cooled to near absolute zero. But it is hard to scale the number of qubits up to the millions that will be needed for useful programs without raising their temperature and destroying their quantum properties. Because NV centres retain their spin at room temperature and communicate their state with light, they might play a part in quantum-computer designs.

 Part of Nature Outline: Quantum diamond sensors
Part of Nature Outline: Quantum diamond sensors
 How quantum diamonds work: from imaging magnetic fields to detecting viruses
How quantum diamonds work: from imaging magnetic fields to detecting viruses
 Video: The quantum world of diamonds
Video: The quantum world of diamonds