- NEWS
- Correction 02 December 2020
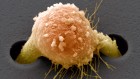
Latin America’s embrace of an unproven COVID treatment is hindering drug trials
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 586, 481-482 (2020)
doi: https://doi.org/10.1038/d41586-020-02958-2
Updates & Corrections
-
Correction 02 December 2020: This story was updated to reflect that Natal is not a state in Brazil. It is the capital city in Rio Grande do Norte, Brazil.
References
Caly, L. et al. Antivir. Res. https://doi.org/10.1016/j.antiviral.2020.104787 (2020).
Peña-Silva, R. et al. Br. J. Clin. Pharmacol. https://doi.org/10.1111/bcp.14476 (2020).
Chandler, R.E. Am. J. Trop. Med. Hyg. 98, 382–388 (2018).

 High-profile coronavirus retractions raise concerns about data oversight
High-profile coronavirus retractions raise concerns about data oversight
 Anti-parasite drugs sweep Nobel prize in medicine 2015
Anti-parasite drugs sweep Nobel prize in medicine 2015
 Evidence lags behind excitement over blood plasma as a coronavirus treatment
Evidence lags behind excitement over blood plasma as a coronavirus treatment