- NEWS FEATURE
The antibiotic paradox: why companies can’t afford to create life-saving drugs
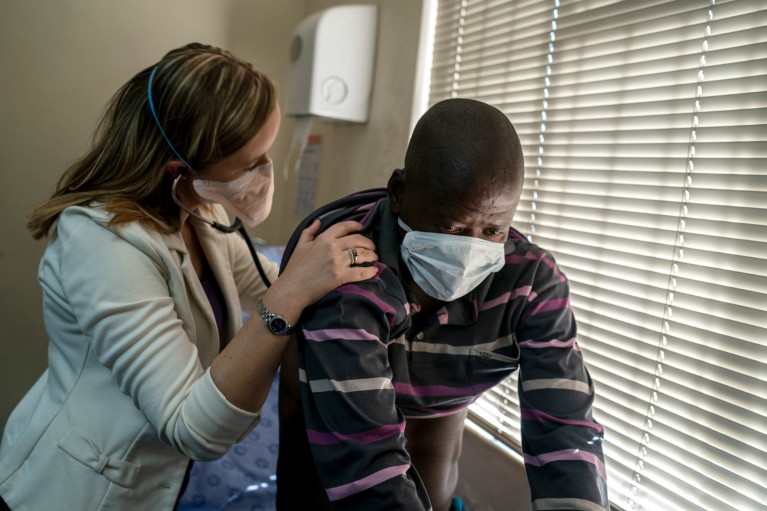
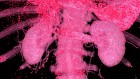
A patient in South Africa battles a strain of tuberculosis that is resistant to multiple antibiotics. Drug resistance is a growing problem with many diseases. Credit: Joao Silva/NYT/Redux/eyevine
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 584, 338-341 (2020)
doi: https://doi.org/10.1038/d41586-020-02418-x
References
DiMasi, J. A., Grabowski, H. G. & Hansen, R. W. J. Health Econ. 47, 20–33 (2016).
Shlaes, D. M. Antimicrob. Agents Chemother. 64, e02057-19 (2020).
Clancy, C. J. & Nguyen, M. H. Antimicrob. Agents Chemother. 63, e01733-19 (2019).
Clancy, C. J., Potoski, B. A., Buehrle, D. & Nguyen, M. H. Open Forum Infect. Dis. 6, ofz344 (2019).
Chen, N. et al. Lancet 395, 507–513 (2020).
Zhou, F. et al. Lancet 395, 1054–1062 (2020).
Rawson, T. A., Ming, D., Ahmad, R., Moore, L. S. P. & Holmes, A. H. Nature Rev. Microbiol. 18, 409–410 (2020).

 Powerful antibiotics discovered using AI
Powerful antibiotics discovered using AI
 Antibiotic resistance has a language problem
Antibiotic resistance has a language problem
 Antibiotics set to flood Florida’s troubled orange orchards
Antibiotics set to flood Florida’s troubled orange orchards
 When antibiotics turn toxic
When antibiotics turn toxic