- OUTLOOK
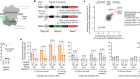
Forkhead BioTherapeutics: developing a diabetes pill
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
doi: https://doi.org/10.1038/d41586-020-01801-y
This article is part of Nature Outlook: The Spinoff Prize 2020, an editorially independent supplement produced with the financial support of third parties. About this content.
References
Bouchi, R. et al. Nature Commun. 5, 4242 (2014).
Langlet, F. et al. Cell 171, 824–835 (2017).

 The Spinoff Prize 2020
The Spinoff Prize 2020
 44 firms highlighted in The Spinoff Prize 2020
44 firms highlighted in The Spinoff Prize 2020
 CageCapture: designing a molecule to filter out pollutants
CageCapture: designing a molecule to filter out pollutants
 Caristo Diagnostics: taking a fresh look at CT scans
Caristo Diagnostics: taking a fresh look at CT scans
 EpiVario: mixing psychotherapy and small-molecule drugs
EpiVario: mixing psychotherapy and small-molecule drugs
 EraCal Therapeutics: a new drug candidate for obesity
EraCal Therapeutics: a new drug candidate for obesity
 MiWEndo Solutions: using microwave technology to improve colonoscopies
MiWEndo Solutions: using microwave technology to improve colonoscopies
 Oxford Brain Diagnostics: turning MRI into a diagnosis tool for dementia
Oxford Brain Diagnostics: turning MRI into a diagnosis tool for dementia
 PredictImmune: a blood test to tailor treatment for bowel condition
PredictImmune: a blood test to tailor treatment for bowel condition
 Scailyte: simplifying difficult diagnoses
Scailyte: simplifying difficult diagnoses
 Sibel Health: designing vital-sign sensors for delicate skin
Sibel Health: designing vital-sign sensors for delicate skin
 Softsonics: a device to take way to blood-pressure readings continuously
Softsonics: a device to take way to blood-pressure readings continuously
 Temprian Therapeutics: developing a gene-based treatment for vitiligo
Temprian Therapeutics: developing a gene-based treatment for vitiligo