- NEWS AND VIEWS
Airborne particles might grow fast in cities
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 581, 145-146 (2020)
doi: https://doi.org/10.1038/d41586-020-01334-4
References
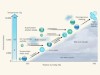
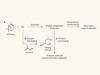
Wang, M. et al. Nature 581, 184–189 (2020).
Seinfeld, J. H. Proc. Natl Acad. Sci. USA 113, 5781–5790 (2016).
Yao, L. et al. Science 361, 278–281 (2018).
Ehn, M. et al. Nature 506, 476–479 (2014).
Kulmala, M., Kerminen, V.-M., Petäjä, T., Ding, A. J. & Wang, L. Faraday Discuss. 200, 271–288 (2017).
Chen, Y. et al. Atmos. Environ. X 5, 100052 (2020).
Van Dingenen, R. et al. Atmos. Environ. 38, 2561–2577 (2004).
Young, D. E. et al. Atmos. Chem. Phys. 15, 6351–6366 (2015).
Kirkby, J. et al. Nature 476, 429–433 (2011).
Huang, R.-J. et al. Nature 514, 218–222 (2014).

 Read the paper: Rapid growth of new atmospheric particles by nitric acid and ammonia condensation
Read the paper: Rapid growth of new atmospheric particles by nitric acid and ammonia condensation
 Sea-spray particles cause freezing in clouds
Sea-spray particles cause freezing in clouds
 Atmospheric reaction networks affecting climate are more complex than was thought
Atmospheric reaction networks affecting climate are more complex than was thought





