- NEWS AND VIEWS
Brain states behind exploring and hunting revealed
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 577, 175-176 (2020)
doi: https://doi.org/10.1038/d41586-019-03811-x
References
Marques, J. C., Li, M., Schaak, D., Robson, D. N. & Li, J. M. Nature https://doi.org/10.1038/s41586-019-1858-z (2019).
Ahrens, M. B. et al. Nature 485, 471–477 (2012).
Vanwalleghem, G. C., Ahrens, M. B. & Scott, E. K. Curr. Opin. Neurobiol. 50, 136–145 (2018).
Kim, D. H. et al. Nature Methods 14, 1107–1114 (2017).
Cohen, J. D., McClure, S. M. & Yu, A. J. Phil. Trans. R. Soc. Lond. B 362, 933–942 (2007).
Flavell, S. W. et al. Cell 154, 1023–1035 (2013).
Kunst, M. et al. Neuron 103, 21–38 (2019).
Hildebrand, D. G. C. et al. Nature 545, 345–349 (2017).

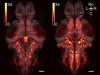 Read the paper: Internal state dynamics shape brainwide activity and foraging behaviour
Read the paper: Internal state dynamics shape brainwide activity and foraging behaviour
 Fluorescent boost for voltage sensors
Fluorescent boost for voltage sensors
 Cell-tracking pipeline reveals how motor circuits are built
Cell-tracking pipeline reveals how motor circuits are built