- NEWS AND VIEWS
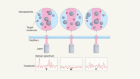
Wearable graphene sensors use ambient light to monitor health
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 576, 220-221 (2019)
doi: https://doi.org/10.1038/d41586-019-03483-7
References
Huang, Z. et al. Nature Electron. 1, 473–480 (2018).
Kim, D.-H. et al. Science 333, 838–843 (2011).
Polat, E. O. et al. Sci. Adv. 5, eaaw7846 (2019).
Fabbro, A. et al. ACS Nano 10, 615–623 (2016).
Kabiri Ameri, S. et al. ACS Nano 11, 7634–7641 (2017).
Kim, J., Kim, J. & Ko, H. Sensors 16, E46 (2015).
Mannheimer, P. D. Anesth. Analg. 105, S10–S17 (2007).
Allen, J. Physiol. Meas. 28 (3), R1–R39 (2007).
Shimazaki, T. & Hara, S. in Proc. 2015 9th Int. Symp. Med. Inform. Commun. Technol. 200–203 (IEEE, 2015).
Kuluncsics, Z., Perdiz, D., Brulay, E., Muel, B. & Sage, E. J. Photochem. Photobiol. B 49, 71–80 (1999).
Akinwande, D. et al. Nature 573, 507–518 (2019).
Huang, H. et al. Front. Chem. 7, 399 (2019).
Ferrari, A. C. et al. Nanoscale 7, 4598–4810 (2015).

 Flexible self-powered biosensors
Flexible self-powered biosensors
 Integration of optoelectronics into fibres enhances textiles
Integration of optoelectronics into fibres enhances textiles