- NEWS
Super-precise new CRISPR tool could tackle a plethora of genetic diseases
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 574, 464-465 (2019)
doi: https://doi.org/10.1038/d41586-019-03164-5
References
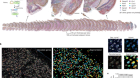
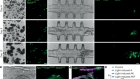
Anzalone, A. V. et al. Nature https://doi.org/10.1038/s41586-019-1711-4 (2019).
Komor, A. C. et al. Nature 533, 420-424 (2016).

 CRISPR might be the banana’s only hope against a deadly fungus
CRISPR might be the banana’s only hope against a deadly fungus
 Many cancer drugs aim at the wrong molecular targets
Many cancer drugs aim at the wrong molecular targets
 CRISPR cuts turn gels into biological watchdogs
CRISPR cuts turn gels into biological watchdogs