- NEWS AND VIEWS
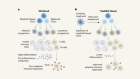
Moderation of neural excitation promotes longevity
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 574, 338-340 (2019)
doi: https://doi.org/10.1038/d41586-019-02958-x
References
Lopez-Otin, C., Blasco, M. A., Partridge, L., Serrano, M. & Kroemer, G. Cell 153, 1194–1217 (2013).
Zullo, J. M. et al. Nature 574, 359–364 (2019).
Lu, A. T. et al. Nature Commun. 8, 15353 (2017).
Camandola, S. & Mattson, M. P. EMBO J. 36, 1474–1492 (2017).
Mattson, M. P. & Arumugam, T. V. Cell Metab. 27, 1176–1199 (2018).
Ailion, M., Inoue, T., Weaver, C. I., Holdcraft, R. W. & Thomas, J. H. Proc. Natl Acad. Sci. USA 96, 7394–7397 (1999).
Apfeld, J. & Kenyon, C. Nature 402, 804–809 (1999).
Wolkow, C. A., Kimura, K. D., Lee, M. S. & Ruvkun, G. Science 290, 147–150 (2000).
Alcedo, J. & Kenyon, C. Neuron 41, 45–55 (2004).
Bishop, N. A., Lu, T. & Yankner, B. A. Nature 464, 529–535 (2010).
Schoenherr, C. J. & Anderson, D. J. Science 267, 1360–1363 (1995).
Pozzi, D. et al. EMBO J. 32, 2994–3007 (2013).
Lu, T. et al. Nature 507, 448–454 (2014).
Kenyon, C. J. Nature 464, 504–512 (2010).
Smialowska, A. & Baumeister, R. Neurodegen. Dis. 3, 227–232 (2006).
Evason, K., Huang, C., Yamben, I., Covey, D. F. & Kornfeld, K. Science 307, 258–262 (2005).
Petrascheck, M., Ye, X. & Buck, L. B. Nature 450, 553–556 (2007).
Siskova, Z. et al. Neuron 84, 1023–1033 (2014).
Ping, Y. et al. PLoS Genet. 11, e1005025 (2015).
Frazzini, V. et al. Cell Death Dis. 7, e2100 (2016).

 Read the paper: Regulation of lifespan by neural excitation and REST
Read the paper: Regulation of lifespan by neural excitation and REST
 A genetic cause of age-related decline
A genetic cause of age-related decline
 Dietary protection for genes
Dietary protection for genes