- NEWS AND VIEWS
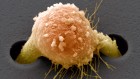
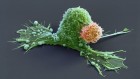
Dangerous liaisons as tumour cells form synapses with neurons
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 573, 499-501 (2019)
doi: https://doi.org/10.1038/d41586-019-02746-7
References
Venkataramani, V. et al. Nature 573, 532–538 (2019).
Venkatesh, H. S. et al. Nature 573, 539–545 (2019).
Zeng, Q. et al. Nature 573, 526–531 (2019).
Perea, G., Navarrete, M. & Araque, A. Trends Neurosci. 32, 421–431 (2009).
Larson, V. A. et al. eLife 7, e34829 (2018).
Ostrom, Q. T. et al. Neuro-oncology 15 (Suppl. 2), ii1–ii56 (2013).
Venkatesh, H. S. et al. Cell 161, 803–816 (2015).
Osswald, M. et al. Nature 528, 93–98 (2015).
Ishiuchi, S. et al. Nature Med. 8, 971–978, (2002).
Rzeski, W., Turski, L. & Ikonomidou, C. Proc. Natl Acad. Sci. USA 98, 6372–6377 (2001).
Savaskan, N. E. et al. Nature Med. 14, 629–632 (2008).
Takano, T. et al. Nature Med. 7, 1010–1015 (2001).
Gambrill, A. C. & Barria, A. Proc. Natl Acad. Sci. USA 108, 5855–5860 (2011).
Barria, A. & Malinow, R. Neuron 48, 289–301 (2005).
Li, L. & Hanahan, D. Cell 153, 86–100 (2013).

 Read the paper: Synaptic proximity enables NMDAR signalling to promote brain metastasis
Read the paper: Synaptic proximity enables NMDAR signalling to promote brain metastasis
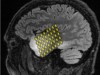 Read the paper: Electrical and synaptic integration of glioma into neural circuits
Read the paper: Electrical and synaptic integration of glioma into neural circuits
 Read the paper: Glutamatergic synaptic input to glioma cells drives brain tumour progression
Read the paper: Glutamatergic synaptic input to glioma cells drives brain tumour progression
 Drain the swamp to beat glioma
Drain the swamp to beat glioma
 Nerve cells from the brain invade prostate tumours
Nerve cells from the brain invade prostate tumours