- NEWS AND VIEWS
Yeast cells handle stress by reprogramming their metabolism
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 572, 184-185 (2019)
doi: https://doi.org/10.1038/d41586-019-02288-y
References
Olin-Sandoval, V. et al. Nature 572, 249–253 (2019).
Nielsen, J. Cell Metab. 25, 572–579 (2017).
Krüger, A. et al. EMBO Rep. 14, 1113–1119 (2013).
Qin, J. et al. Nature Commun. 6, 8224 (2015).
Eisenberg, T. et al. Nature Cell Biol. 11, 1305–1314 (2009).
Campbell, K., Vowinckel, J., Keller, M. A. & Ralser, M. Antioxid. Redox Signal. 24, 543–547 (2016).
Nielsen, J. Biotechnol. J. https://doi.org/10.1002/biot.201800421 (2019).

 Read the paper: Lysine harvesting is an antioxidant strategy and triggers underground polyamine metabolism
Read the paper: Lysine harvesting is an antioxidant strategy and triggers underground polyamine metabolism
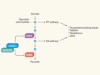 Division enzyme regulates metabolism
Division enzyme regulates metabolism
 Metabolism reprogrammed by the nitric oxide signalling molecule
Metabolism reprogrammed by the nitric oxide signalling molecule