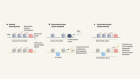
- NEWS FEATURE
The secret social lives of viruses

Illustration by Karol Banach
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 570, 290-292 (2019)
doi: https://doi.org/10.1038/d41586-019-01880-6
References
Erez, Z. et al. Nature 541, 488–493 (2017).
Turner, P. E. & Chao, L. Nature 398, 441–443 (1999).
Wang, Q. et al. Nature Microbiol. 3, 1266–1273 (2018).
Dou, C. et al. Nature Microbiol. 3, 1285–1294 (2018).
Zhen, X. et al. Protein Cell 10, 131–136 (2019).
Gallego Del Sol, F., Penadés, J. R. & Marina, A. Mol. Cell 74, 59–72.e3 (2019).
Guan, Z. et al. Cell Discov. 5, 29 (2019).
Stokar-Avihail, A., Tal, N., Erez, Z., Lopatina, A. & Sorek, R. Cell Host Microbe 25, 746–755.e5 (2019).
Silpe, J. E. & Bassler, B. L. Cell 176, 268–280.e13 (2019).
Landsberger, M. et al. Cell 174, 908–916.e12 (2018).
Borges, A. L. et al. Cell 174, 917–925.e10 (2018).
Chevallereau, A. et al. preprint at bioRxiv https://doi.org/10.1101/574418 (2019).
Domingo-Calap, P., Segredo-Otero, E., Durán-Moreno, M. & Sanjuán, R. Nature Microbiol. 4, 1006–1013 (2019).
Aguilera, E. R., Erickson, A. K., Jesudhasan, P. R., Robinson, C. M. & Pfeiffer, J. K. mBio 8, e02020-16 (2017).
Xue, K. S., Hooper, K. A., Ollodart, A. R., Dingens, A. S. & Bloom, J. D. eLife 5, e13974 (2016).
Xue, K. S., Greninger, A. L., Pérez-Osorio, A. & Bloom, J. D. mSphere 3, e00552-17 (2018).
Santiana, M. et al. Cell Host Microbe 24, 208–220.e8 (2018).
Dedrick, R. M. et al. Nature Med. 25, 730–733 (2019).
Díaz-Muñoz, S. L., Sanjuán, R. & West, S. Cell Host Microbe 22, 437–441 (2017).

 Do you speak virus? Phages caught sending chemical messages
Do you speak virus? Phages caught sending chemical messages
 Phage therapy gets revitalized
Phage therapy gets revitalized
 Modified viruses deliver death to antibiotic-resistant bacteria
Modified viruses deliver death to antibiotic-resistant bacteria
 A billion-year arms race against viruses shaped our evolution
A billion-year arms race against viruses shaped our evolution
 HIV overcomes CRISPR gene-editing attack
HIV overcomes CRISPR gene-editing attack
 CRISPR-like ‘immune’ system discovered in giant virus
CRISPR-like ‘immune’ system discovered in giant virus