- NEWS AND VIEWS
A possible non-biological reaction framework for metabolic processes on early Earth
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 569, 47-49 (2019)
doi: https://doi.org/10.1038/d41586-019-01322-3
References
Muchowska, K. B., Varma, S. J. & Moran, J. Nature 569, 104–107 (2019).
Laneuville, M., Kameya, M. & Cleaves, H. J. II Astrobiology 18, 897–914 (2018).
Fry, I. in Handbook of Astrobiology (ed. Kolb, V. M.) Ch. 3.1, 109–124 (CRC, 2019).
Pascal, R., Pross, A. & Sutherland, J. D. Open Biol. 3, 130156 (2013).
Sutherland, J. D. Nature Rev. Chem. 1, 0012 (2017).
Bruylants, G., Bartik, K. & Reisse, J. C. R. Chim. 14, 388–391 (2011).
Ruiz-Mirazo, K., Briones, C. & de la Escosura, A. Chem. Rev. 114, 285–366 (2014).
Miller, S. L. Science 117, 528–529 (1953).
Lotka, A. J. Proc. Natl Acad. Sci. USA 8, 151–154 (1922).
Pross, A. What is Life? How Chemistry Becomes Biology (Oxford Univ. Press, 2016).

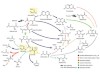 Read the paper: Synthesis and breakdown of universal metabolic precursors promoted by iron
Read the paper: Synthesis and breakdown of universal metabolic precursors promoted by iron
 The rocky road to biomolecules
The rocky road to biomolecules
 Small molecular replicators go organic
Small molecular replicators go organic