- NEWS AND VIEWS
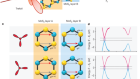
Refrigeration based on plastic crystals
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 567, 470-471 (2019)
doi: https://doi.org/10.1038/d41586-019-00974-5
References
Li, B. et al. Nature 567, 506–510 (2019).
Mañosa, L. & Planes, A. Adv. Mater. 29, 1603607 (2017).
Bermúdez-García, J. M. et al. Nature Commun. 8, 15715 (2017).
Aznar, A. et al. Nature Commun. 8, 1851 (2017).
Cazorla, C. & Errandonea, D. Nano Lett. 16, 3124–3129 (2016).
Sagotra, A. K., Errandonea, D. & Cazorla, C. Nature Commun. 8, 963 (2017).
Sagotra, A. K., Chu, D. & Cazorla, C. Nature Commun. 9, 3337 (2018).
Aznar, A, Lloveras, P. & Tamarit, J.-L. Eur. Phys. J. Spec. Top. 226, 1017–1029 (2017).
Gottschall, T. et al. Nature Mater. 17, 929–934 (2018).
Liu, S. & Cohen, R. E. J. Phys. Chem. C 120, 17274–17281 (2016).
Harada, J. et al. Nature Chem. 8, 946–952 (2016).

 Read the paper: Colossal barocaloric effects in plastic crystals
Read the paper: Colossal barocaloric effects in plastic crystals
 Optical cooling achieved by tuning thermal radiation
Optical cooling achieved by tuning thermal radiation
 Ferroelectric polymers morph into action
Ferroelectric polymers morph into action