- NEWS FEATURE
- Clarification 27 March 2019
How secret conversations inside cells are transforming biology
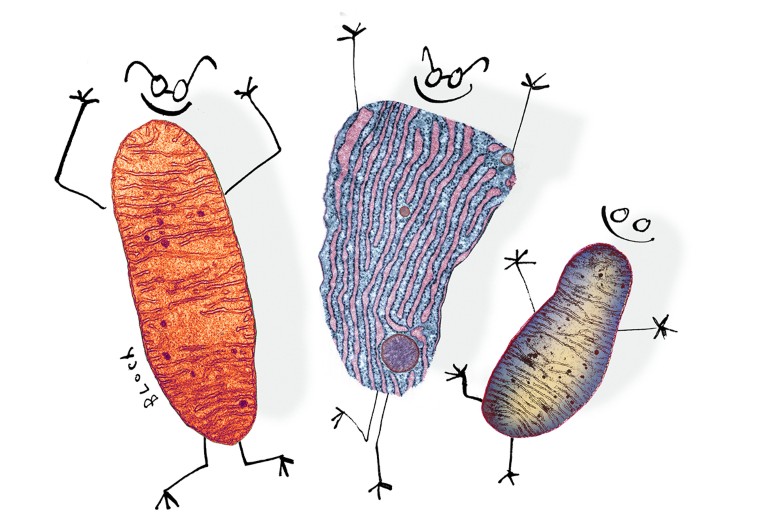
Illustration by Serge Bloch
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 567, 162-164 (2019)
doi: https://doi.org/10.1038/d41586-019-00792-9
Updates & Corrections
-
Clarification 27 March 2019: This story has been modified to make clear that the 2017 study from DeCamilli was co-led by Yale cell biologist Karin Reinisch.
References
Vance, J. E. J. Biol. Chem. 265, 7248–7256 (1990).
Valm, A. M. et al. Nature 546, 162–167 (2017).
Kornmann, B. et al. Science 325, 477–481 (2009).
Manford, A. G., Stefan, C. J., Yuan, H. L., Macgurn, J. A. & Emr, S. D. Dev. Cell 23, 1129–1140 (2012).
Hönscher, C. et al. Dev. Cell 30, 86–94 (2014).
Elbaz-Alon, Y. et al. Dev. Cell 30, 95–102 (2014).
Lackner, L. L., Ping, H., Graef, M., Murley, A. & Nunnari, J. Proc. Natl Acad. Sci. USA 110, E458–E467 (2013).
Shai, N. et al. Nature Commun. 9, 1761 (2018).
Friedman, J. R. et al. Science 334, 358–362 (2011).
Rowland, A. A., Chitwood, P. J., Phillips, M. J. & Voeltz, G. K. Cell 159, 1027–1041 (2014).
Wu, Y. et al. Proc. Natl Acad. Sci. USA 114, E4859–E4867 (2017).
Lees, J. A. et al. Science 355, eaah6171 (2017).
Arruda, A. P. et al. Nature Med. 20, 1427–1435 (2014).
Area Gómez, E. et al. Cell Death Dis. 9, 335 (2018).

 Map of body’s protein-folding machinery wins major prize
Map of body’s protein-folding machinery wins major prize