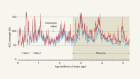
- NEWS AND VIEWS
Consistent patterns of nitrogen fixation identified in the ocean
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 566, 191-193 (2019)
doi: https://doi.org/10.1038/d41586-019-00498-y
References
Redfield, A. C. in James Johnstone Memorial Volume (ed. Daniel, R. J.) 176–192 (Univ. College Liverpool, 1934).
Carpenter, E. J. & Capone, D. G. in Nitrogen in the Marine Environment 2nd edn (eds Capone, D. G., Bronk, D. A., Mulholland, M. R. & Carpenter, E. J.) 141–198 (Elsevier, 2008).
Gruber, N. Proc. Natl Acad. Sci. USA 113, 4246–4248 (2016).
Deutsch, C., Sarmiento, J. L., Sigman, D. M., Gruber, N. & Dunne, J. P. Nature 445, 163–167 (2007).
Luo, Y.-W., Lima, I. D., Karl, D. M., Deutsch, C. A. & Doney, S. C. Biogeosciences 11, 691–708 (2014).
Knapp, A. N., Casciotti, K. L., Berelson, W. M., Prokopenko, M. G. & Capone, D. G. Proc. Natl Acad. Sci. USA 113, 4398–4403 (2016).
Bonnet, S., Caffin, M., Berthelot, H. & Moutin, T. Proc. Natl Acad. Sci. USA 114, E2800–E2801 (2017).
Wang, W.-L., Moore, J. K., Martiny, A. C. & Primeau, F. W. Nature 566, 205–211 (2019).
DeVries, T. & Primeau, F. J. Phys. Oceanogr. 41, 2381–2401 (2011).
Gruber, N. in Nitrogen in the Marine Environment 2nd edn (eds Capone, D. G., Bronk, D. A., Mulholland, M. R. & Carpenter, E. J.) 1–150 (Elsevier, 2008).
Yang, S., Gruber, N., Long, M. C. & Vogt, M. Glob. Biogeochem. Cycles 31, 1470–1487 (2017).
Kim, I.-N. et al. Science 346, 1102–1106 (2014).
Somes, C. J., Landolfi, A., Koeve, W. & Oschlies, A. Geophys. Res. Lett. 43, 4500–4509 (2016).
Moisander, P. H. et al. Science 327, 1512–1514 (2010).

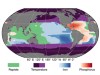 Read the paper: Convergent estimates of marine nitrogen fixation
Read the paper: Convergent estimates of marine nitrogen fixation
 A fight for scraps of ammonia
A fight for scraps of ammonia
 The depths of nitrogen recycling
The depths of nitrogen recycling