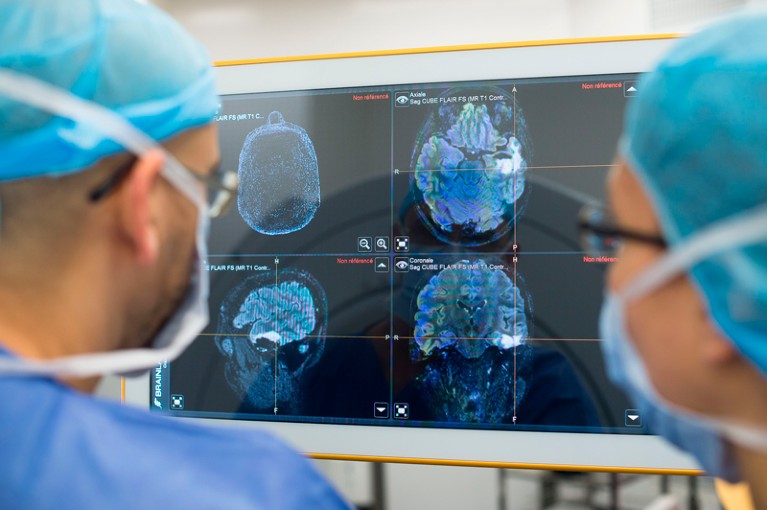
Brain tumours are among the most difficult cancers to treat.Credit: BSIP/UIG/Getty
“I knew it was serious from the sober demeanor of the medical professionals in the room.” That was how US Senator John McCain described his diagnosis of cancer in his 2018 book The Restless Wave. “And when someone, I don’t remember who, mentioned that it was the same cancer that Ted had, I got the picture.”
Ted was Edward Kennedy, McCain’s fellow senator. And the cancer was a brain tumour. Kennedy had died in 2009. McCain succumbed last August — just a year after his fateful diagnosis. That’s not unusual. Malignant tumours in the brain are among the most feared cancers, partly because they progress rapidly and are so often lethal — killing around half of patients within a year of diagnosis.
Unlike with most other cancers, survival rates for brain tumours have not improved much in recent years, even with the introduction of new treatments such as targeted therapies and immunotherapies. Better treatment for brain tumours is thus a pressing medical need, especially for some devastating childhood forms of the disease.
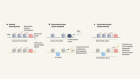
Actively personalized vaccination trial for newly diagnosed glioblastoma
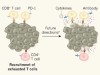
This week in Nature, two papers report some progress on that front. The positive result is that they detail the first testing in humans of a strategy involving ‘neoantigen vaccines’. Patients are given injections of a vaccine that is based on the genetic composition of their specific tumour type, and it is intended to stimulate and boost their own immune defences. This approach has previously shown promise in melanomas. It’s the latest type of immunotherapy to be tested in brain cancers, and follows trials with other methods, including viruses targeted against the cancers.
As is highlighted in an accompanying News & Views article, the vaccines managed to elicit some immune responses in people with brain tumours. But the effect was limited, and the patients still died.
The results emphasize the major anatomical and biological challenges of treating brain tumours. In particular, the brain’s delicate, complex structure is protected by the blood–brain barrier, and it is difficult to find cancer drugs that can cross this and have an impact, while limiting their toxic effects on neurons.
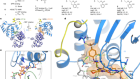
Neoantigen vaccine generates intratumoral T cell responses in phase Ib glioblastoma trial
The tumours’ sensitive location also makes it more difficult to screen and monitor them without resorting to complex surgery. They are often composed of many genetically different cells, helping them to adapt to and elude targeted therapy. And they are considered to be immunologically ‘cold’ — not recognized or responded to by the body’s immune system — which can limit the efficacy of immunotherapies.
The new vaccine studies show that supporting treatments such as the steroid dexamethasone can hamper the potency of immune responses. They also suggest that responses can be raised against both mutated and non-mutated tumour-related proteins, and show how these responses can be limited by immune-cell exhaustion.
Given the extent of the challenges involved, the slow pace of progress on brain cancer treatment should not be a reason to be discouraged. Thanks to the generosity of people who enrol in trials, and the determination of researchers and clinicians who continue to collaborate and foster treatment initiatives, we can still gain important insights. The new papers, for example, should shed light on how immune responses develop in people with brain tumours, and what can be done to boost those responses. That is one way to accelerate progress.

 Actively personalized vaccination trial for newly diagnosed glioblastoma
Actively personalized vaccination trial for newly diagnosed glioblastoma
 Neoantigen vaccine generates intratumoral T cell responses in phase Ib glioblastoma trial
Neoantigen vaccine generates intratumoral T cell responses in phase Ib glioblastoma trial
 Immune cells track hard-to-target brain tumours
Immune cells track hard-to-target brain tumours