- NEWS AND VIEWS
Implanted device enables responsive bladder control
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 565, 298-300 (2019)
doi: https://doi.org/10.1038/d41586-018-07811-1
References
Birmingham, K. et al. Nature Rev. Drug Discov. 13, 399–400 (2014).
Siegel, S. W. et al. Urology 56 (Suppl. 1), 87–91 (2000).
Kavvadias, T., Huebner, M., Brucker, S. Y. & Reisenauer, C. Arch. Gynecol. Obstet. 295, 951–957 (2017).
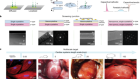
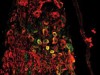
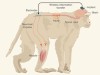
Mickle, A. D. et al. Nature 565, 361–365 (2019).
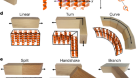
Krishnan, S. R. et al. Small 14, 1803192 (2018).
Krishnan, S. R. et al. Sci. Transl. Med. 10, eaat8437 (2018).
Chen, X. et al. Nature Commun. 9, 1690 (2018).
Nan, K. et al. Sci. Adv. 4, eaau5849 (2018).
Xu, L. et al. Nature Commun. 5, 3329 (2014).
Gustaffson, F. & Rogers, J. G. Eur. J. Heart Failure 19, 595–602 (2017).
Roche, E. T. et al. Sci. Transl. Med. 9, eaaf3925 (2017).

 Read the paper: A wireless closed-loop system for optogenetic peripheral neuromodulation
Read the paper: A wireless closed-loop system for optogenetic peripheral neuromodulation
 Neural interfaces take another step forward
Neural interfaces take another step forward
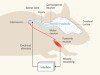 Boost for movement
Boost for movement