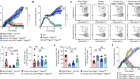
- NEWS AND VIEWS
Put to sleep by immune cells
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 562, 46-48 (2018)
doi: https://doi.org/10.1038/d41586-018-06666-w
References
Latorre, D. et al. Nature 562, 63–68 (2018).
Scammell, T. E. N. Engl. J. Med. 373, 2654–2662 (2015).
de Lecea, L. et al. Proc. Natl Acad. Sci. USA 95, 322–327 (1998).
Sakurai, T. Curr. Opin. Neurobiol. 23, 760–766 (2013).
Liblau, R. S., Vassalli, A., Seify, A. & Tafti, M. Lancet Neurol. 14, 318–328 (2015).
Dauvilliers, Y., Arnulf, I. & Mignot, E. Lancet 369, 499–511 (2007).
Dendrou, C. A., Petersen, J., Rossjohn, J. & Fugger, L. Nature Rev. Immunol. 18, 325–339 (2018).
Behalf of the European Narcolepsy Network (EU-NN). Sleep 37, 19–25 (2014)
Mignot, E. et al. Am. J. Hum. Genet. 68, 686–699 (2001).
Cvetkovic-Lopes, V. et al. J. Clin. Invest. 120, 713–719 (2010).
Ahmed, S. S. et al. Sci. Transl. Med. 7, 294ra105 (2015).
Luo, G. et al. Preprint at bioRxiv https://doi.org/10.1101/378109 (2018).
Sharon, E. et al. Nature Genet. 48, 995–1002 (2016).
Nguyen, X.-H., Saoudi, A. & Liblau, R. S. Curr. Opin. Neurol. 29, 362–371 (2016).
Sarkanen, T. O., Alakuijala, A. P. E., Dauvilliers, Y. A. & Partinen, M. M. Sleep Med. Rev. 38, 177–186 (2018).
Birnbaum, M. E. et al. Cell 157, 1073–1087 (2014).
Bernard-Valnet, R. et al. Proc. Natl Acad. Sci. USA 113, 10956–10961 (2016).
Liblau, R. S., Gonzalez-Dunia D., Wiendl, H. & Zipp, F. Trends Neurosci. 36, 315–324 (2013).
Competing Interests
R.S.L. has received a grant from GSK.

 Read the paper: T cells in narcolepsy patients target self-antigens of hypocretin neurons
Read the paper: T cells in narcolepsy patients target self-antigens of hypocretin neurons
 Synapses pruned in lupus
Synapses pruned in lupus
 Sleepy neurons?
Sleepy neurons?