- OUTLOOK
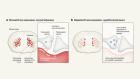
An age-old story of dementia
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 559, S2-S3 (2018)
doi: https://doi.org/10.1038/d41586-018-05718-5
This article is part of Nature Outlook: Alzheimer’s disease, an editorially independent supplement produced with the financial support of third parties. About this content.
References
Jack, C. R. Jr et al. Lancet Neurol. 12, 207–216 (2013).
Alzheimer’s Disease International. World Alzheimer Report 2015: The Global Impact of Dementia (Alzheimer’s Disease International, 2015).
Matthews, F. E. et al. Lancet 382, 1405–1412 (2013).
Chêne, G. et al. Alzheimers Dement. 11, 310–320 (2015).
Mucke, L. Nature 461, 895–897 (2009).

 Part of Nature Outlook: Alzheimer’s disease
Part of Nature Outlook: Alzheimer’s disease
 The amyloid hypothesis on trial
The amyloid hypothesis on trial
 Drawing on the brain’s resilience to fight Alzheimer’s disease
Drawing on the brain’s resilience to fight Alzheimer’s disease
 Alzheimer’s disease is getting easier to spot
Alzheimer’s disease is getting easier to spot
 The search for better animal models of Alzheimer’s disease
The search for better animal models of Alzheimer’s disease
 Ways to stop the spread of Alzheimer’s disease
Ways to stop the spread of Alzheimer’s disease
 How the evidence stacks up for preventing Alzheimer’s disease
How the evidence stacks up for preventing Alzheimer’s disease
 More from Nature Outlooks
More from Nature Outlooks